ALDEHYDES KETONES ALKANALS ALKANONES 1 ALDEHYDES KETONES ALKANALS






- Slides: 21

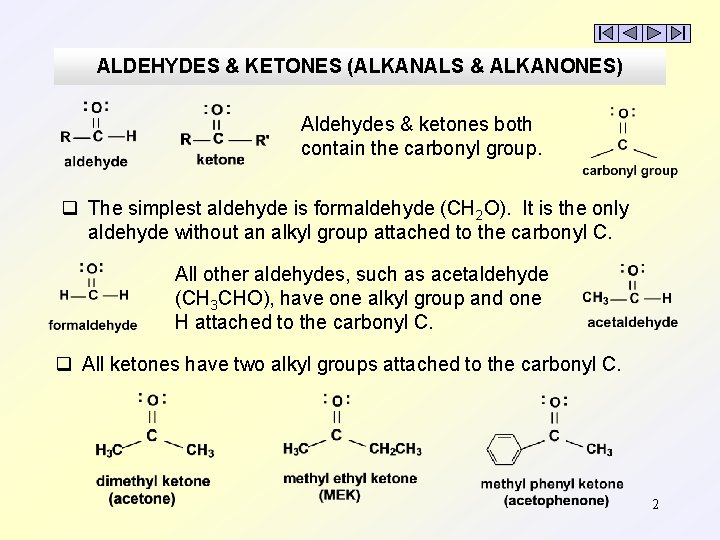
ALDEHYDES & KETONES (ALKANALS & ALKANONES) 1

ALDEHYDES & KETONES (ALKANALS & ALKANONES) Aldehydes & ketones both contain the carbonyl group. q The simplest aldehyde is formaldehyde (CH 2 O). It is the only aldehyde without an alkyl group attached to the carbonyl C. All other aldehydes, such as acetaldehyde (CH 3 CHO), have one alkyl group and one H attached to the carbonyl C. q All ketones have two alkyl groups attached to the carbonyl C. 2

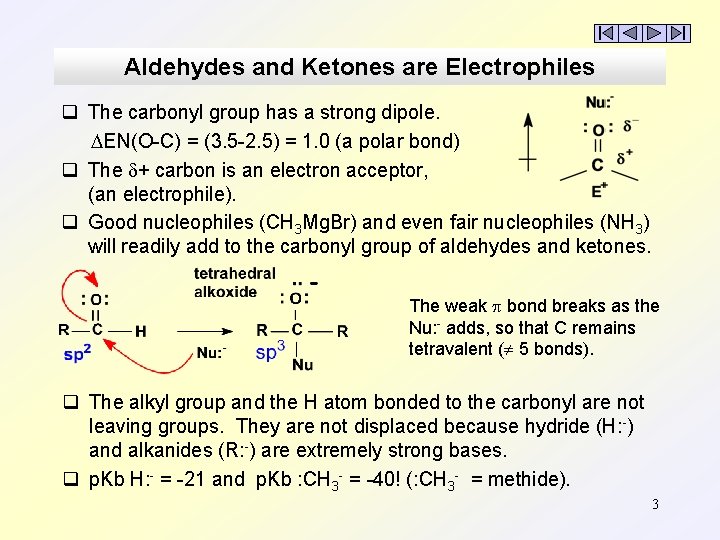
Aldehydes and Ketones are Electrophiles q The carbonyl group has a strong dipole. DEN(O-C) = (3. 5 -2. 5) = 1. 0 (a polar bond) q The + carbon is an electron acceptor, (an electrophile). q Good nucleophiles (CH 3 Mg. Br) and even fair nucleophiles (NH 3) will readily add to the carbonyl group of aldehydes and ketones. The weak p bond breaks as the Nu: - adds, so that C remains tetravalent ( 5 bonds). q The alkyl group and the H atom bonded to the carbonyl are not leaving groups. They are not displaced because hydride (H: -) and alkanides (R: -) are extremely strong bases. q p. Kb H: - = -21 and p. Kb : CH 3 - = -40! (: CH 3 - = methide). 3

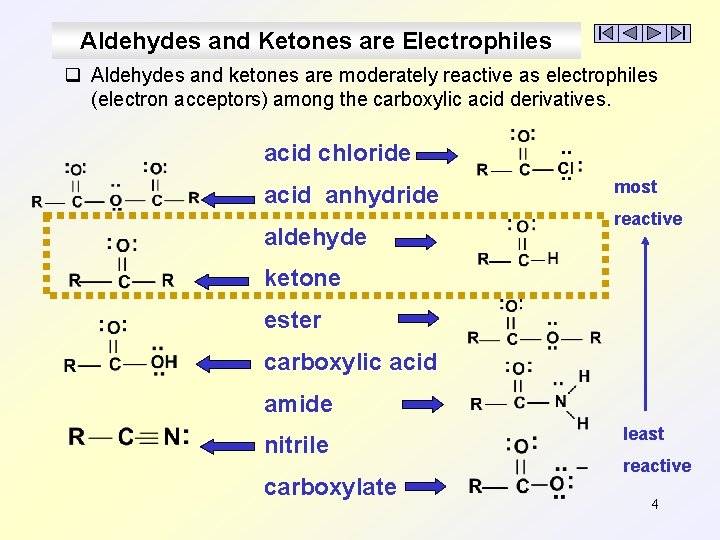
Aldehydes and Ketones are Electrophiles q Aldehydes and ketones are moderately reactive as electrophiles (electron acceptors) among the carboxylic acid derivatives. acid chloride acid anhydride aldehyde most reactive ketone ester carboxylic acid amide nitrile carboxylate least reactive 4

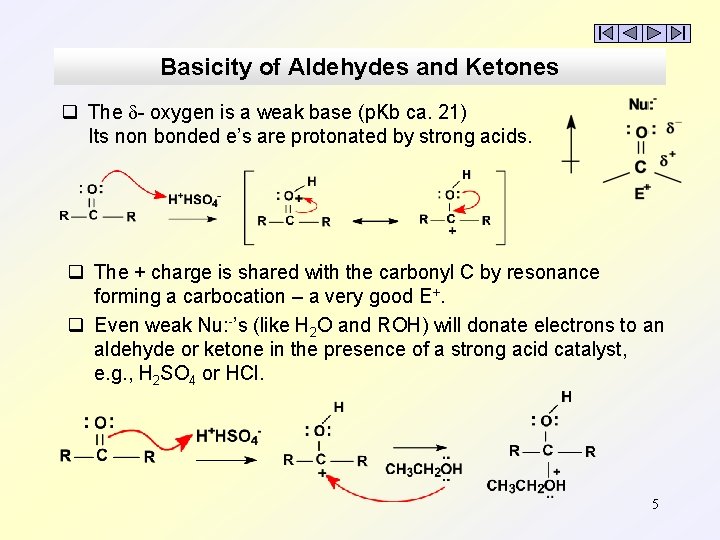
Basicity of Aldehydes and Ketones q The - oxygen is a weak base (p. Kb ca. 21) Its non bonded e’s are protonated by strong acids. q The + charge is shared with the carbonyl C by resonance forming a carbocation – a very good E+. q Even weak Nu: -’s (like H 2 O and ROH) will donate electrons to an aldehyde or ketone in the presence of a strong acid catalyst, e. g. , H 2 SO 4 or HCl. 5

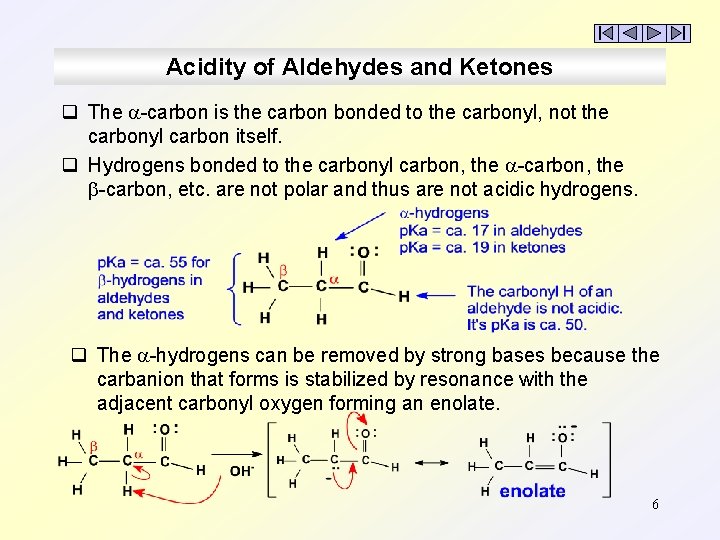
Acidity of Aldehydes and Ketones q The -carbon is the carbon bonded to the carbonyl, not the carbonyl carbon itself. q Hydrogens bonded to the carbonyl carbon, the -carbon, etc. are not polar and thus are not acidic hydrogens. q The -hydrogens can be removed by strong bases because the carbanion that forms is stabilized by resonance with the adjacent carbonyl oxygen forming an enolate. 6

Boiling Points and Solubility of Aldehydes and Ketones q The carbonyl group is strongly polar but does not produce hydrogen bonding (It has no polar hydrogens). As a result, the boiling points of aldehydes and ketones are higher than the nonpolar hydrocarbons and the alkyl halides but lower than those of alcohols. q Formaldehyde is a gas at room temperature (b. p. = -21 C) but heavier aldehydes are liquids. Acetone, the simplest ketone, is a liquid at room temperature (b. p. = 56 C). q Lower molecular weight aldehydes and ketones are water soluble. Acetone, formaldehyde and acetaldehyde are miscible in water. 7

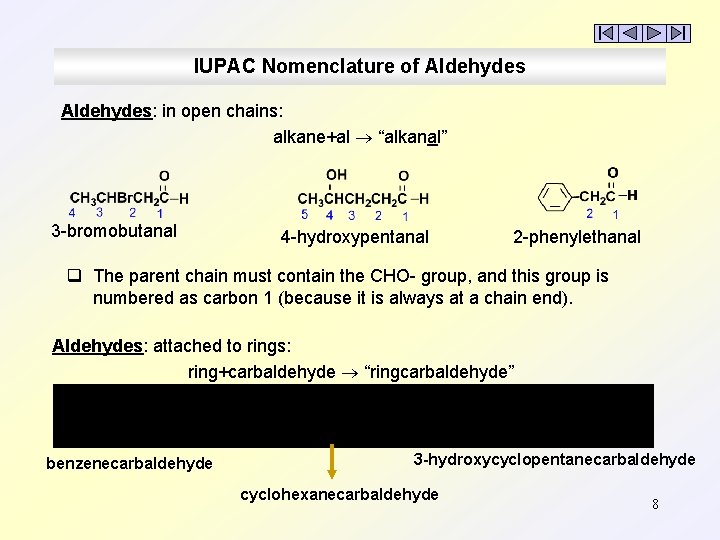
IUPAC Nomenclature of Aldehydes: in open chains: alkane+al “alkanal” 3 -bromobutanal 4 -hydroxypentanal 2 -phenylethanal q The parent chain must contain the CHO- group, and this group is numbered as carbon 1 (because it is always at a chain end). Aldehydes: attached to rings: ring+carbaldehyde “ringcarbaldehyde” benzenecarbaldehyde 3 -hydroxycyclopentanecarbaldehyde cyclohexanecarbaldehyde 8

9

Common Names of Aldehydes q In the common system, aldehydes are named from the common names of the corresponding carboxylic acid. q The ‘ic acid’ ending is replaced with ‘aldehyde’. formic acid acetic acid propionic acid butyric acid valeric acid caproic acid formaldehyde acetaldehyde propionaldehyde butyraldehyde valeraldehyde caproaldehyde q Substituents locations are given using Greek letters ( , , , . ) beginning with the carbon next to the carbonyl carbon, the -carbon. -bromobutyraldehyde -hydroxyvaleraldehyde -phenylacetaldehyde 10

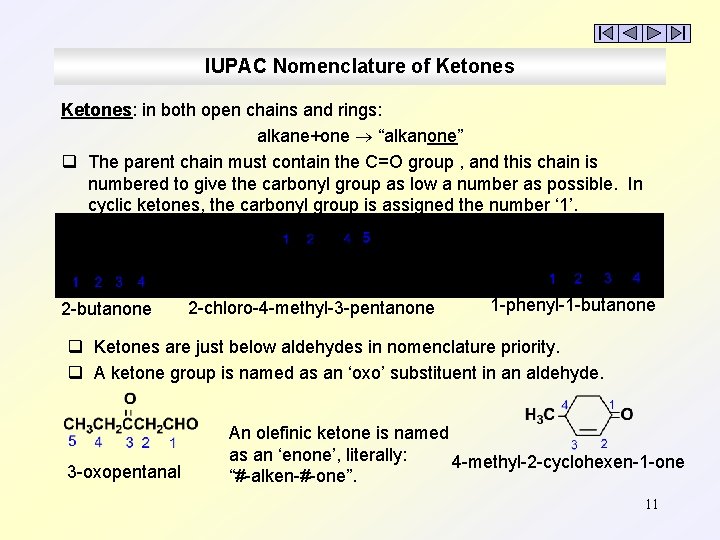
IUPAC Nomenclature of Ketones: in both open chains and rings: alkane+one “alkanone” q The parent chain must contain the C=O group , and this chain is numbered to give the carbonyl group as low a number as possible. In cyclic ketones, the carbonyl group is assigned the number ‘ 1’. 2 -butanone 2 -chloro-4 -methyl-3 -pentanone 1 -phenyl-1 -butanone q Ketones are just below aldehydes in nomenclature priority. q A ketone group is named as an ‘oxo’ substituent in an aldehyde. 3 -oxopentanal An olefinic ketone is named as an ‘enone’, literally: 4 -methyl-2 -cyclohexen-1 -one “#-alken-#-one”. 11

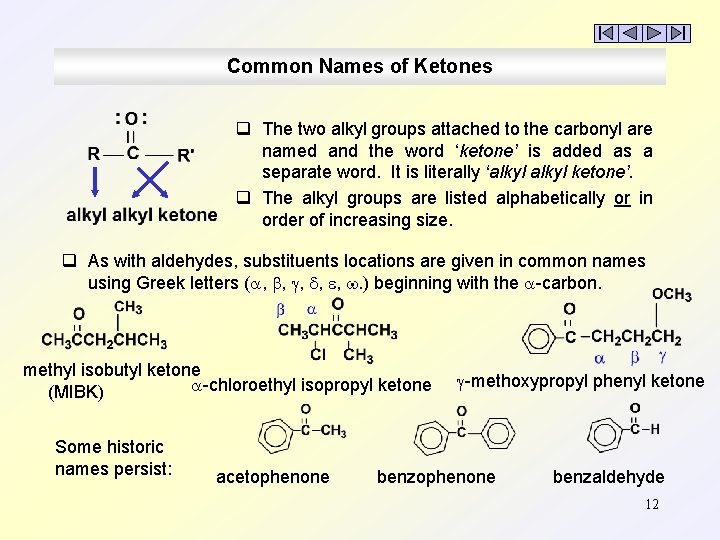
Common Names of Ketones q The two alkyl groups attached to the carbonyl are named and the word ‘ketone’ is added as a separate word. It is literally ‘alkyl ketone’. q The alkyl groups are listed alphabetically or in order of increasing size. q As with aldehydes, substituents locations are given in common names using Greek letters ( , , , . ) beginning with the -carbon. methyl isobutyl ketone -chloroethyl isopropyl ketone (MIBK) Some historic names persist: acetophenone -methoxypropyl phenyl ketone benzophenone benzaldehyde 12

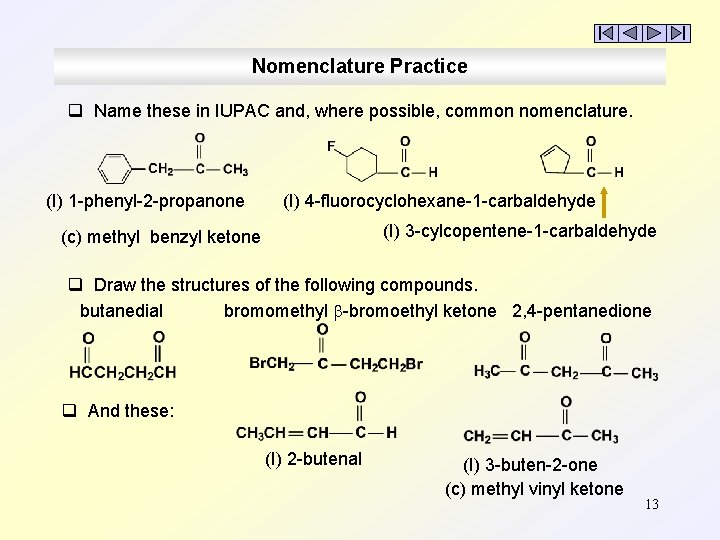
Nomenclature Practice q Name these in IUPAC and, where possible, common nomenclature. (I) 1 -phenyl-2 -propanone (I) 4 -fluorocyclohexane-1 -carbaldehyde (I) 3 -cylcopentene-1 -carbaldehyde (c) methyl benzyl ketone q Draw the structures of the following compounds. butanedial bromomethyl -bromoethyl ketone 2, 4 -pentanedione q And these: (I) 2 -butenal (I) 3 -buten-2 -one (c) methyl vinyl ketone 13

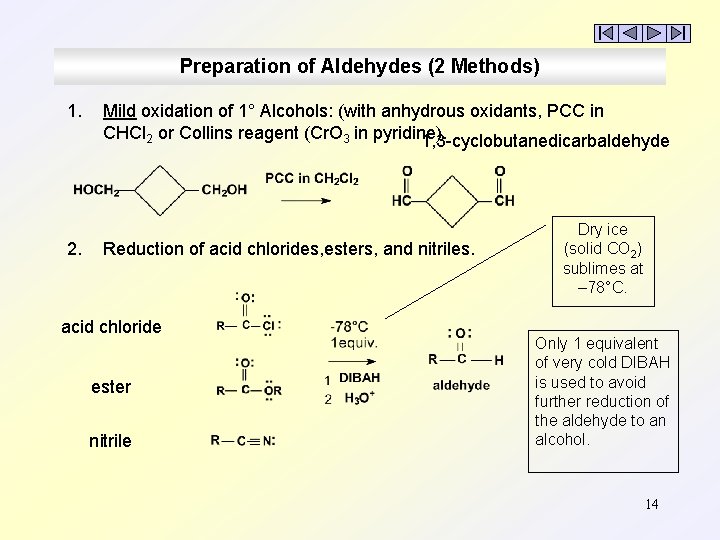
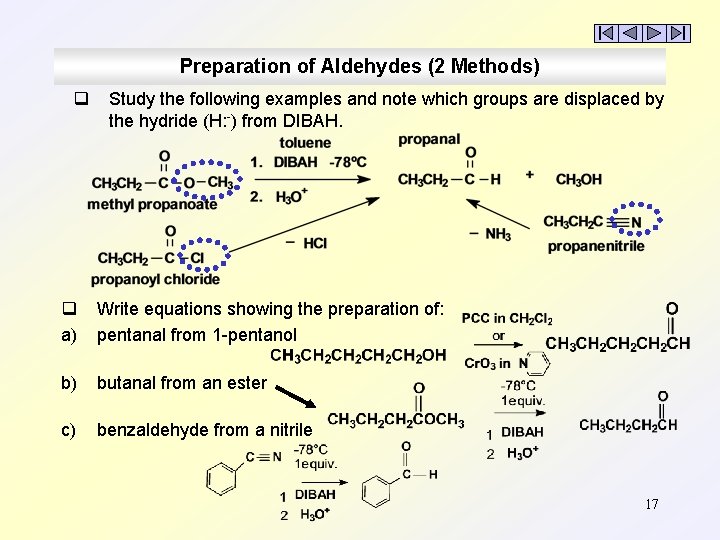
Preparation of Aldehydes (2 Methods) 1. 2. Mild oxidation of 1° Alcohols: (with anhydrous oxidants, PCC in CHCl 2 or Collins reagent (Cr. O 3 in pyridine). 1, 3 -cyclobutanedicarbaldehyde Reduction of acid chlorides, esters, and nitriles. acid chloride ester nitrile Dry ice (solid CO 2) sublimes at – 78°C. Only 1 equivalent of very cold DIBAH is used to avoid further reduction of the aldehyde to an alcohol. 14

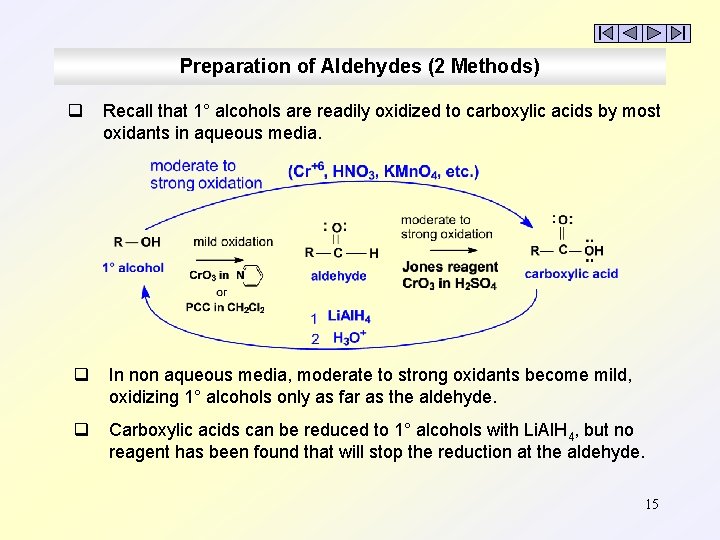
Preparation of Aldehydes (2 Methods) q Recall that 1° alcohols are readily oxidized to carboxylic acids by most oxidants in aqueous media. q In non aqueous media, moderate to strong oxidants become mild, oxidizing 1° alcohols only as far as the aldehyde. q Carboxylic acids can be reduced to 1° alcohols with Li. Al. H 4, but no reagent has been found that will stop the reduction at the aldehyde. 15

Preparation of Aldehydes (2 Methods) q q q Carboxylic acids are difficult to reduce and any reducing agent strong enough to reduce them, e. g. , Li. Al. H 4, will not stop at the aldehyde but always produces the 1° alcohol. Several ‘derivatives’ of carboxylic acids can be reduced to aldehydes under carefully controlled conditions. Acid chlorides, esters, and nitriles are reduced to aldehydes using very cold conditions (-78°C) and only 1 equivalent of a mild reducing agent, ‘diisobutylaluminum hydride’ = DIBAH (usually in toluene). DIBAH is weaker than Li. Al. H 4. DIBAH is neutral; Li. Al. H 4 is ionic. DIBAH is similar to Al. H 3 but is hindered by its bulky isobutyl groups. Only one mole of H: - is released per mole of DIBAH. 16

Preparation of Aldehydes (2 Methods) q Study the following examples and note which groups are displaced by the hydride (H: -) from DIBAH. q a) Write equations showing the preparation of: pentanal from 1 -pentanol b) butanal from an ester c) benzaldehyde from a nitrile 17

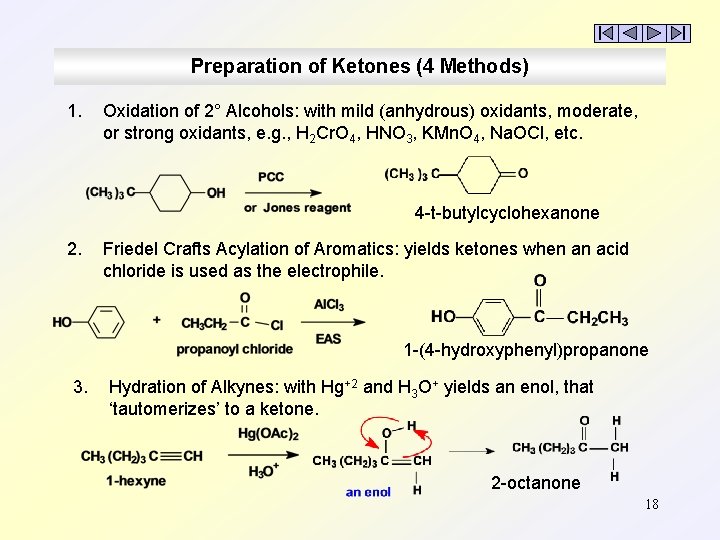
Preparation of Ketones (4 Methods) 1. Oxidation of 2° Alcohols: with mild (anhydrous) oxidants, moderate, or strong oxidants, e. g. , H 2 Cr. O 4, HNO 3, KMn. O 4, Na. OCl, etc. 4 -t-butylcyclohexanone 2. Friedel Crafts Acylation of Aromatics: yields ketones when an acid chloride is used as the electrophile. 1 -(4 -hydroxyphenyl)propanone 3. Hydration of Alkynes: with Hg+2 and H 3 O+ yields an enol, that ‘tautomerizes’ to a ketone. 2 -octanone 18

Preparation of Ketones (4 Methods) 4. q q Acid Chlorides + Lithium Dialkyl Copper (Gilman Reagent): produces ketones. The reaction is unique to these two reagents and the mechanism is uncertain. As with DIBAH for aldehyde reductions, a low temperature (78 C) solvent (ether) is used to prevent further alkyl addition to the ketone to form an alcohol. (Acid chlorides are very good electrophiles). Carboxylic acids, esters, anhydrides and amides are not reduced by diorganocopper reagents. They are not as reactive as acid chlorides. 2 -heptanone q Recall that a stronger reducing reagent, such as a Grignard (RMg. Br) will also reduce an acid chloride to a ketone, but reduction cannot be stopped here. The ketone is further reduced to an alcohol. 19

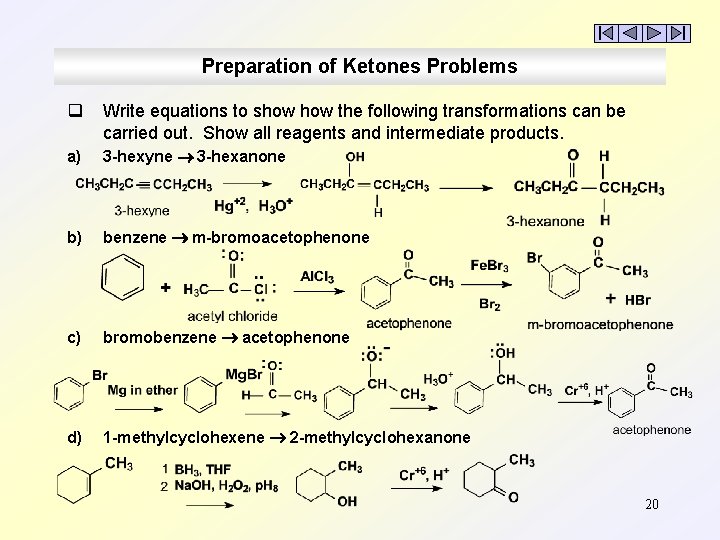
Preparation of Ketones Problems q Write equations to show the following transformations can be carried out. Show all reagents and intermediate products. a) 3 -hexyne 3 -hexanone b) benzene m-bromoacetophenone c) bromobenzene acetophenone d) 1 -methylcyclohexene 2 -methylcyclohexanone 20

Preparation of Ketones Problems q Recall the effects of substituents on aromatic rings. They affect both the reactivity of aromatics and the position at which Electrophilic Aromatic Substitution (EAS) will occur. 21