Gases Kinetic Molecular Theory of Gases A gas

































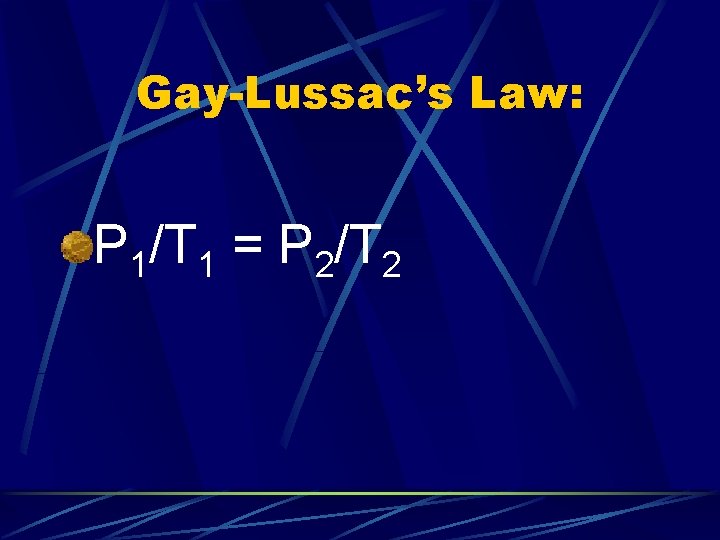

- Slides: 35

Gases Kinetic Molecular Theory of Gases

A gas consists of small particles (atoms/molecules) that move randomly with rapid velocities Further Information They move faster when heated.

The attractive forces between particles of a gas can be neglected Do you think this is accurate? Why would this be important for calculations?

The actual volume occupied by a gas molecule is extremely small compared to the volume that gas occupies. Is this true in the real world? Why would this be helpful with calculations?

The average kinetic energy of a gas molecule is proportional to Kelvin temperature What is kinetic energy? Why Kelvin temperature and not Celsius or Fahrenheit? What does proportional mean?

Gas particles are in constant motion, moving rapidly in straight paths. * Is this true? What do we know about their motion? Why would the real situation make the calculations more difficult?

Ideal Gases An imaginary gas that perfectly fits all the assumptions of the kinetic molecular theory (KMT).

Expansion Gases do not have definite shape or volume. The expand to any container they are enclosed in. A gas in a 1 L container is then put into a 2 L container. How much volume does it have now?

Fluidity In an ideal gas, the gas particles glide past each other. This feature allows gases to be referred to as fluids just like liquids.

Low Density of a gas substance is only about 1/1000 of the same substance in liquid or solid state. Why is this true?

Compressibility This is a crowding effect of gases when the volume is decreased

Diffusion Spontaneous (does not require energy) mixing of particles of two substances caused by their random motion

Properties of a Gas Units of Measure

Pressure is not the same as force. Pressure is a force over an area. Example: psi = Pounds per in 2

Measuring Pressure A barometer measures atmospheric pressure.

Units of Pressure k. Pa, atm, mm of Hg, torr Helpful Conversions 1 atm = 760 mm Hg 1 atm = 760 torr 1 mm Hg = 1 torr 1 atm = 101. 325 k. Pa

Volume L, m. L or cm 3 Helpful conversions 1000 m. L = 1 L l 1 m. L = 1 cm 3 l

Temperature 0 C or K Helpful conversions: 0 C = K – 273 K = 0 C + 273

moles Number of moles = n If you are given grams, how would you convert to moles?

Standard Temperature and Pressure (STP) Standard Temperature is 00 C or 273 K Standard Pressure is 101. 3 k. Pa or 1 atm

Boyle’s Law: Pressure and volume are inversely proportional P 1 V 1 = P 2 V 2

Charles’ Law: Temperature and Volume are directly proportional V 1 / T 1 = V 2 / T 2

Gay-Lussac’s Law: Pressure and Temperature are directly proportional P 1/T 1 = P 2/T 2

Combined Gas Law P 1 V 1 = P 2 V 2 T 1 T 2 If you remember this law, hold constant the other variables not used and you have all the gas laws we’ve used so far.

Molar Volume 1 mole = 22. 4 L of a gas at STP Now, we can convert between moles and grams; moles and molecules/atoms; and moles and volume (L)

Dalton’s Law of Partial Pressure The total pressure is equal to the sum of the partial pressures

Avagadro’s Law: V 1 / n 1 = V 2/n 2 Where n = number of moles How do you convert grams to moles?

Ideal Gas Law PV=n. RT P = Pressure (k. Pa) V = volume (L) n = number of moles R = 8. 31 k. Pa x L / moles x K T = temperature (K) You must use these units for the R constant to be correct.

Name the Law! You will be given a series of laws and asked to name the law or you will be given the name and be asked to come up with the formula!

PV =n. RT Ideal Gas Law

V 1 / T 1 = V 2 / T 2 Charles’ Law

Boyle’s Law P 1 V 1 = P 2 V 2

Combined Gas Law P 1 V 1 = P 2 V 2 T 1 T 2
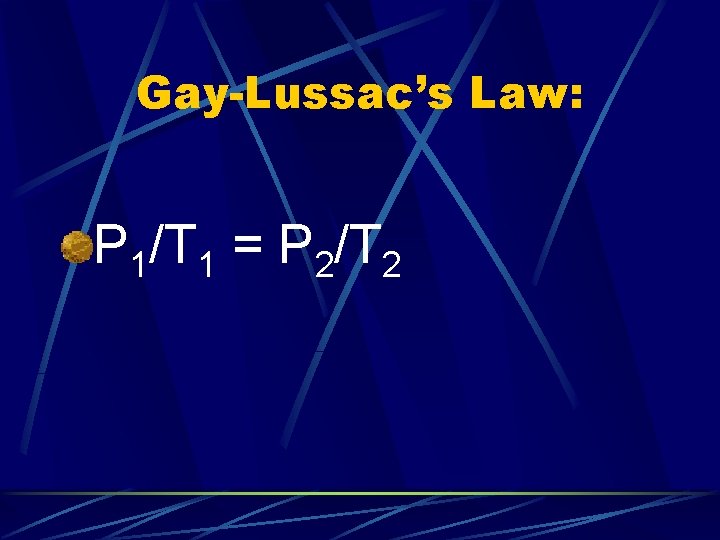
Gay-Lussac’s Law: P 1/T 1 = P 2/T 2

Avagadro’s Law: V 1 / n 1 = V 2/n 2