Bonding Theories Valence Bond Theory Molecular Orbital Theory

Bonding Theories: Valence Bond Theory Molecular Orbital Theory Tro: Chemistry: A Molecular Approach, 2/e Copyright 2011 Pearson Education, Inc.

Lewis Theory: Electron Groups and Molecular Shapes There are Regions of electrons in an atom: • Some from placing shared pairs of valence electrons between bonding nuclei; • Other from placing lone pairs valence electrons on a single nuclei Regions of electron groups should repel each other (VSEPR) and determines the molecular shape and molecular polarity Tro: Chemistry: A Molecular Approach, 2/e 2 Copyright 2011 Pearson Education, Inc.
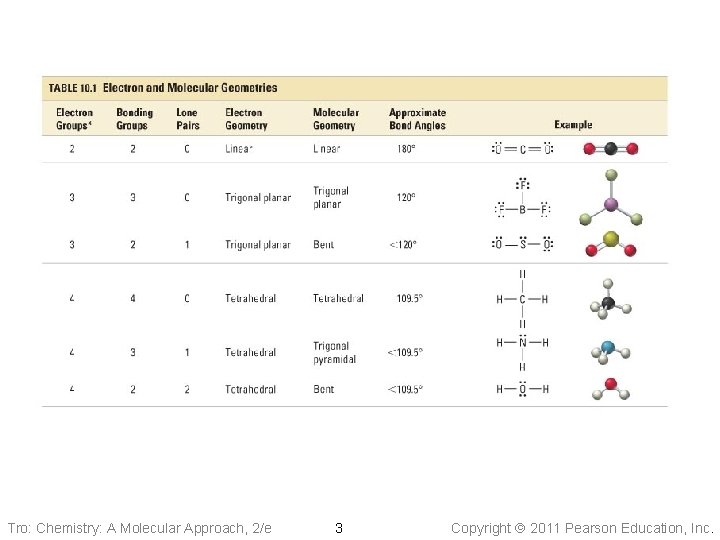
Tro: Chemistry: A Molecular Approach, 2/e 3 Copyright 2011 Pearson Education, Inc.

Problems with Lewis Theory • Useful predicting trends, but not quantitative e. g. bond angles, bond strength, bond length • Can not give one correct structure for many molecules where resonance is important • Often does not predict the correct magnetic behavior of molecules ü e. g. O 2 is paramagnetic, though the Lewis structure predicts it is diamagnetic Tro: Chemistry: A Molecular Approach, 2/e 4 Copyright 2011 Pearson Education, Inc.

Valence Bond Theory. I Linus Pauling et al: • Chemical bonds form when the orbitals (wave functions of electrons in the atoms) on those atoms interact (overlap) • The kind of interaction depends on whether the orbitals align along the axis between the nuclei, or outside the axis Tro: Chemistry: A Molecular Approach, 2/e 5 Copyright 2011 Pearson Education, Inc.
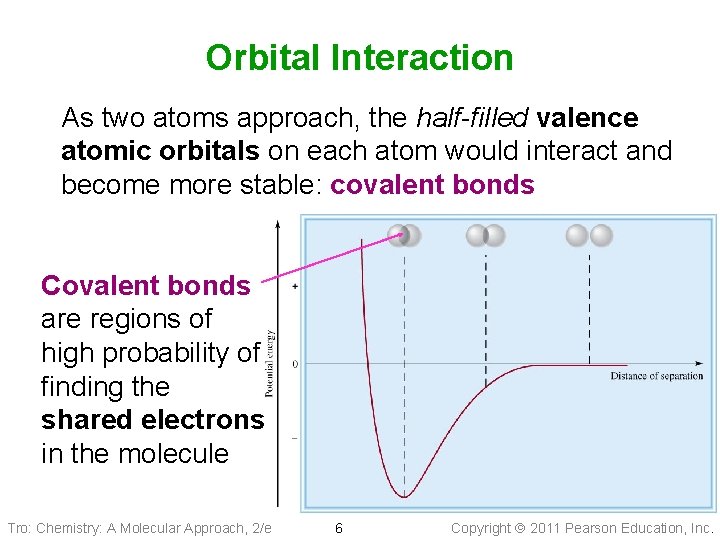
Orbital Interaction As two atoms approach, the half-filled valence atomic orbitals on each atom would interact and become more stable: covalent bonds Covalent bonds are regions of high probability of finding the shared electrons in the molecule Tro: Chemistry: A Molecular Approach, 2/e 6 Copyright 2011 Pearson Education, Inc.
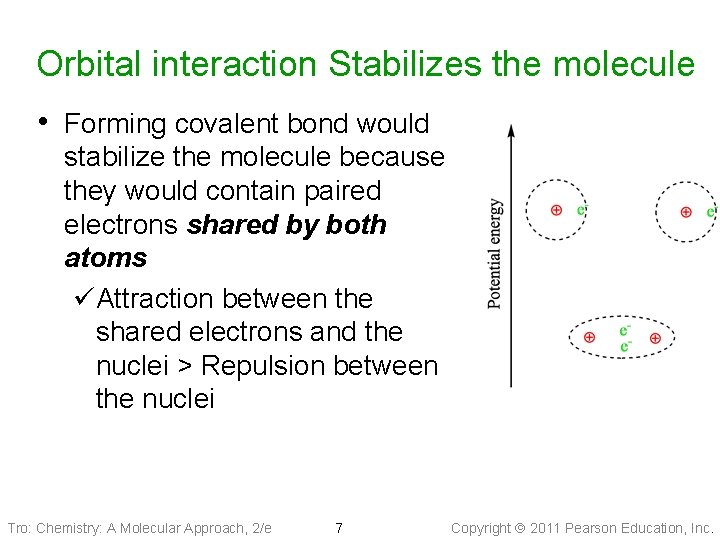
Orbital interaction Stabilizes the molecule • Forming covalent bond would stabilize the molecule because they would contain paired electrons shared by both atoms üAttraction between the shared electrons and the nuclei > Repulsion between the nuclei Tro: Chemistry: A Molecular Approach, 2/e 7 Copyright 2011 Pearson Education, Inc.
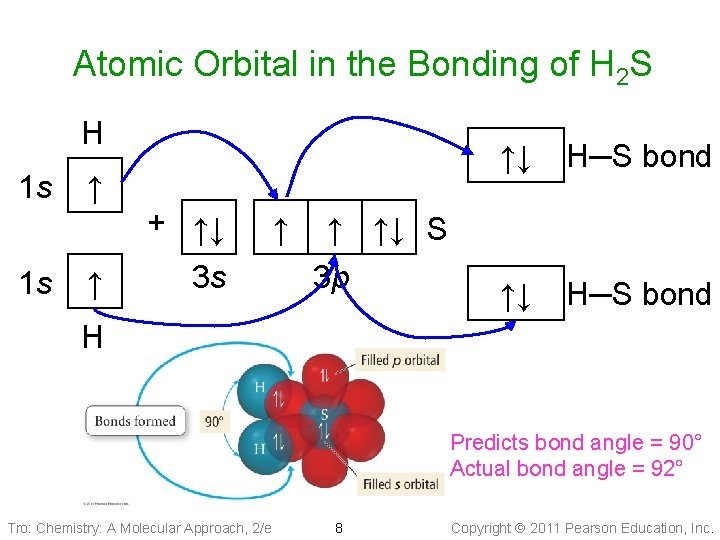
Atomic Orbital in the Bonding of H 2 S H 1 s ↑ + ↑↓ 1 s ↑ 3 s ↑ ↑↓ H─S bond ↑ ↑↓ S 3 p H Predicts bond angle = 90° Actual bond angle = 92° Tro: Chemistry: A Molecular Approach, 2/e 8 Copyright 2011 Pearson Education, Inc.
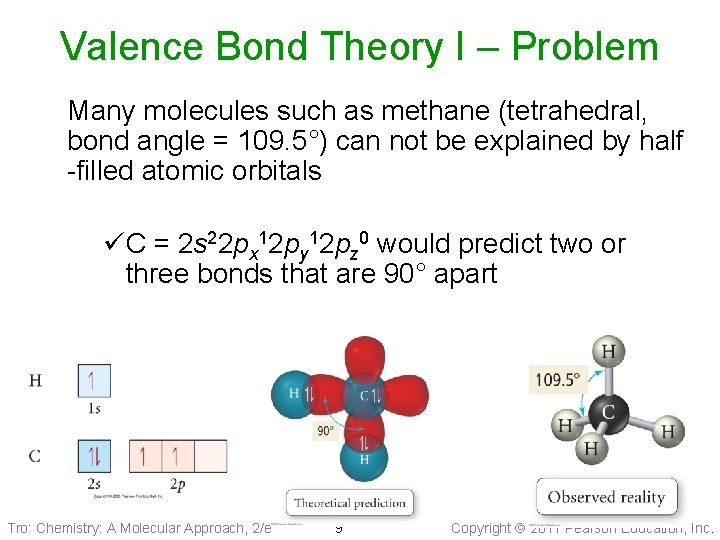
Valence Bond Theory I – Problem Many molecules such as methane (tetrahedral, bond angle = 109. 5°) can not be explained by half -filled atomic orbitals üC = 2 s 22 px 12 py 12 pz 0 would predict two or three bonds that are 90° apart Tro: Chemistry: A Molecular Approach, 2/e 9 Copyright 2011 Pearson Education, Inc.

Valence Bond Theory II: Orbital Hybridization 1. When forming covalent bonds, each valence electron can reside in either standard s, p, d, and f orbitals, or in hybridized orbitals of these atomic orbitals. 2. Covalent bond forms via half-filled atomic orbitals interacting, ONE pair of electrons (↑↓) in the new bonding orbital 3. Shape of the molecule determined by the geometry of the interacting orbitals Tro: Chemistry: A Molecular Approach, 2/e 10 Copyright 2011 Pearson Education, Inc.

Hybridization + = • Some atoms hybridize their orbitals to • maximize bonding Hybridization: mixing different types of orbitals in the valence shell to make a new set of degenerate (equal energy) orbitals ü 1 x s orbital + 2 x p orbital = 3 x sp 2 hybrid orbital • Same type of atom can have different types of hybridization ü C = sp, sp 2, sp 3 Tro: Chemistry: A Molecular Approach, 2/e 11 Copyright 2011 Pearson Education, Inc.

Hybrid Orbitals • The number of standard atomic orbitals combined = the number of hybrid orbitals formed ücombining a 2 s with a 2 p gives 2 x sp hybrid orbitals üH cannot hybridize!! Øits valence shell only has one orbital • The number and type of standard atomic orbitals combined determines the shape of the hybrid orbitals Tro: Chemistry: A Molecular Approach, 2/e 12 Copyright 2011 Pearson Education, Inc.
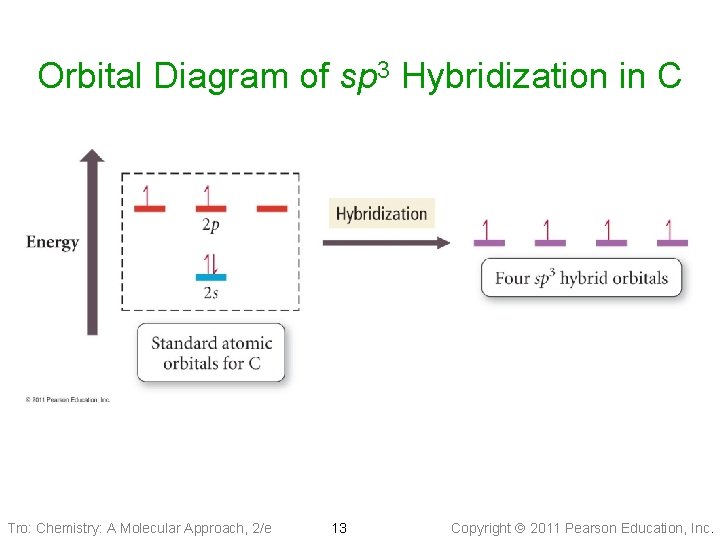
Orbital Diagram of sp 3 Hybridization in C Tro: Chemistry: A Molecular Approach, 2/e 13 Copyright 2011 Pearson Education, Inc.
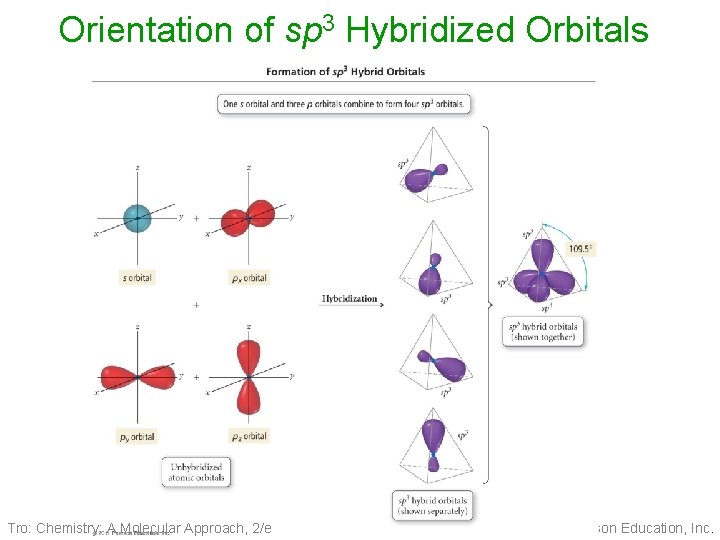
Orientation of sp 3 Hybridized Orbitals Tro: Chemistry: A Molecular Approach, 2/e 14 Copyright 2011 Pearson Education, Inc.
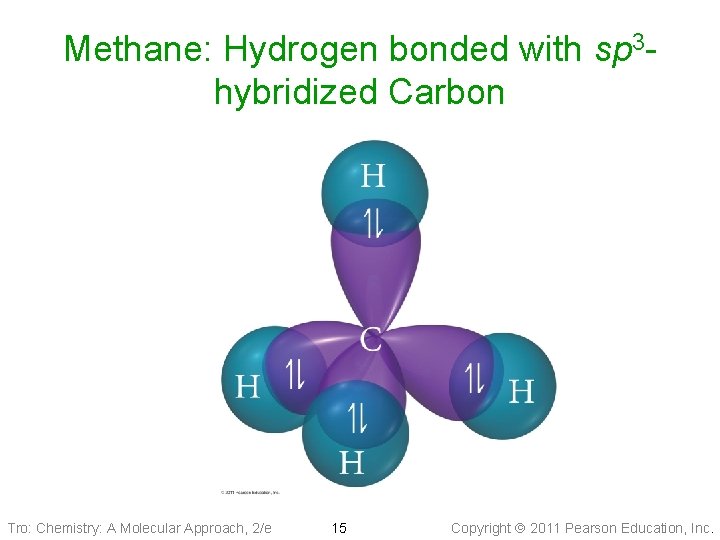
Methane: Hydrogen bonded with sp 3 hybridized Carbon Tro: Chemistry: A Molecular Approach, 2/e 15 Copyright 2011 Pearson Education, Inc.
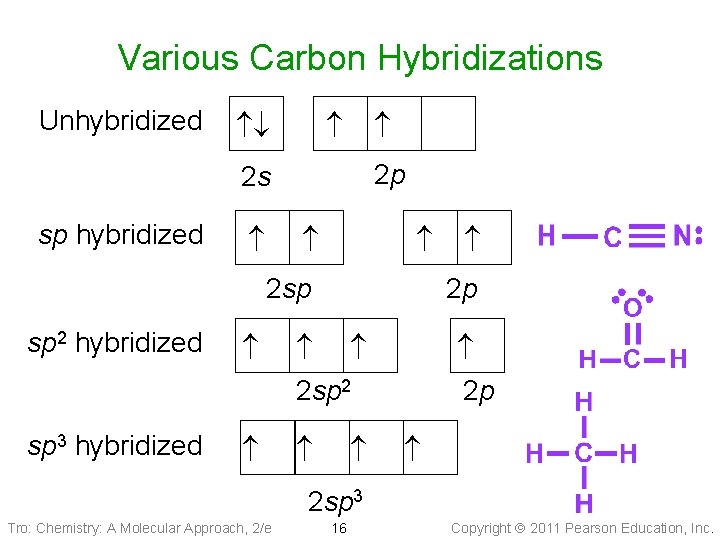
Various Carbon Hybridizations Unhybridized 2 p 2 s sp hybridized 2 sp 2 p sp 2 hybridized 2 sp 2 sp 3 hybridized 2 p 2 sp 3 Tro: Chemistry: A Molecular Approach, 2/e 16 Copyright 2011 Pearson Education, Inc.
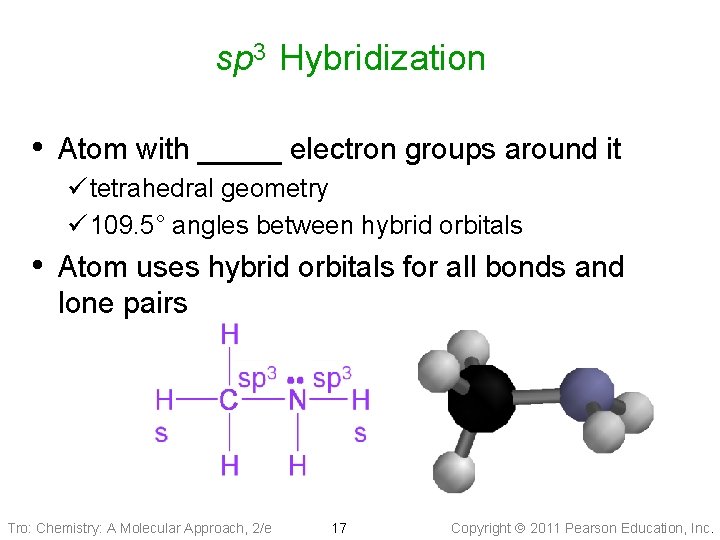
sp 3 Hybridization • Atom with _____ electron groups around it ü tetrahedral geometry ü 109. 5° angles between hybrid orbitals • Atom uses hybrid orbitals for all bonds and lone pairs Tro: Chemistry: A Molecular Approach, 2/e 17 Copyright 2011 Pearson Education, Inc.
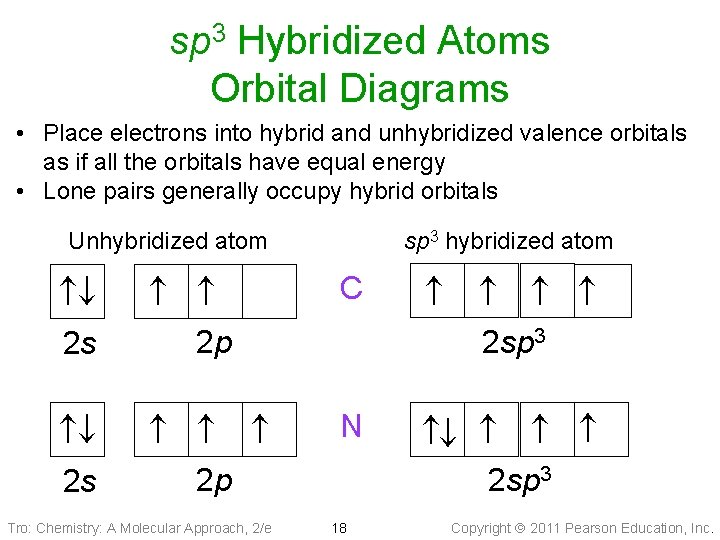
sp 3 Hybridized Atoms Orbital Diagrams • Place electrons into hybrid and unhybridized valence orbitals as if all the orbitals have equal energy • Lone pairs generally occupy hybrid orbitals Unhybridized atom 2 s 2 p C 2 p N Tro: Chemistry: A Molecular Approach, 2/e 18 2 sp 3 sp 3 hybridized atom 2 sp 3 Copyright 2011 Pearson Education, Inc.

Bonding with Valence Bond Theory According to valence bond theory, bonding takes place between atoms when their atomic or hybrid orbitals interact (“overlap”). To interact, • The orbitals must either be aligned along the axis between the atoms. or • The orbitals must be parallel to each other and perpendicular to the interatomic axis Tro: Chemistry: A Molecular Approach, 2/e 19 Copyright 2011 Pearson Education, Inc.
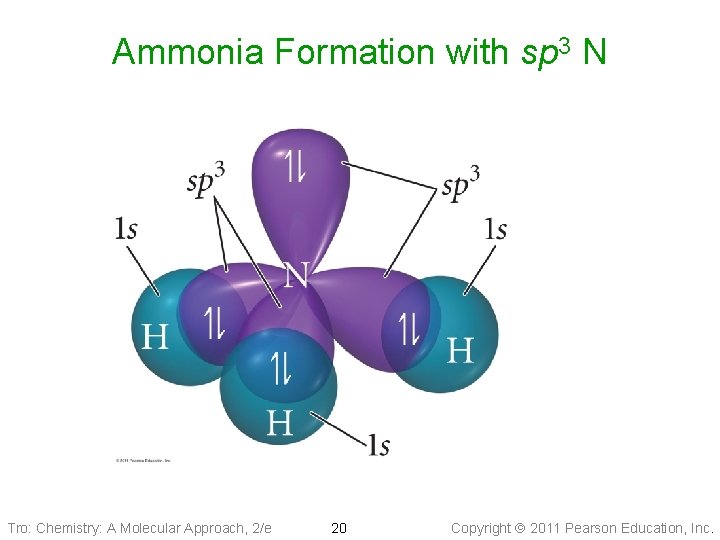
Ammonia Formation with sp 3 N Tro: Chemistry: A Molecular Approach, 2/e 20 Copyright 2011 Pearson Education, Inc.
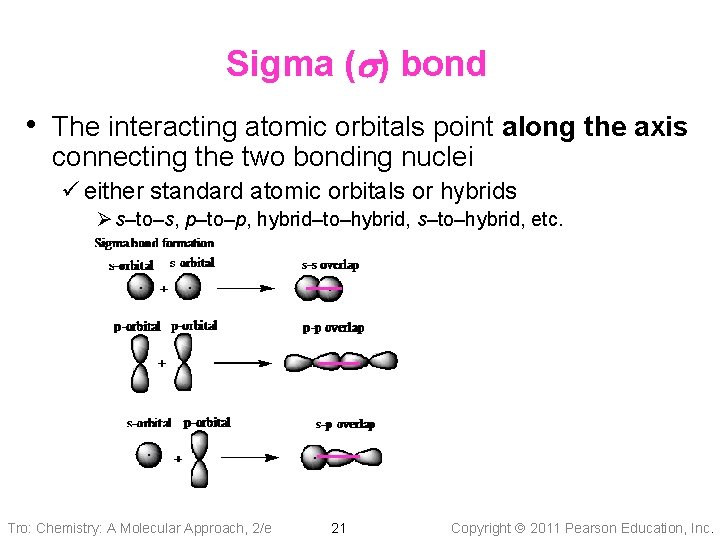
Sigma (s) bond • The interacting atomic orbitals point along the axis connecting the two bonding nuclei ü either standard atomic orbitals or hybrids Ø s–to–s, p–to–p, hybrid–to–hybrid, s–to–hybrid, etc. Tro: Chemistry: A Molecular Approach, 2/e 21 Copyright 2011 Pearson Education, Inc.

pi (p) bond • Definition: The bonding atomic orbitals are parallel to each other and perpendicular to the axis connecting the two bonding nuclei ü between unhybridized parallel p orbitals • Double bond = one s bond + one p bond • Triple bond = one s bond + two p bonds • Strength of orbital overlap: s bonds > p bonds Tro: Chemistry: A Molecular Approach, 2/e 22 Copyright 2011 Pearson Education, Inc.
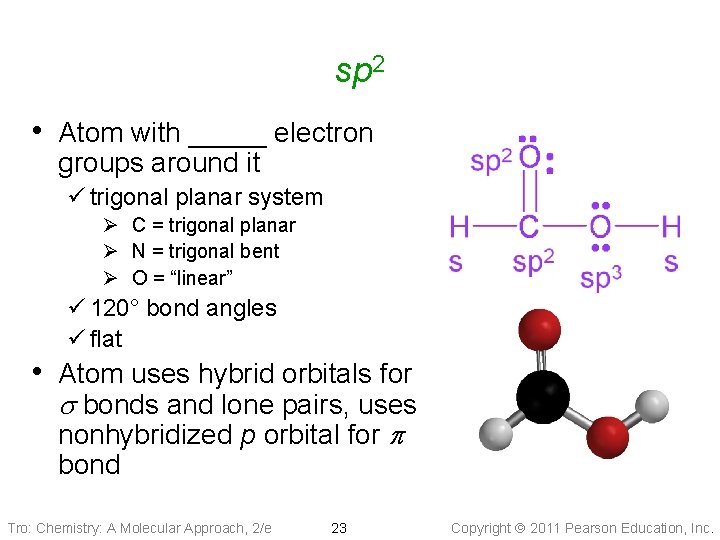
sp 2 • Atom with _____ electron groups around it ü trigonal planar system Ø C = trigonal planar Ø N = trigonal bent Ø O = “linear” ü 120° bond angles ü flat • Atom uses hybrid orbitals for s bonds and lone pairs, uses nonhybridized p orbital for p bond Tro: Chemistry: A Molecular Approach, 2/e 23 Copyright 2011 Pearson Education, Inc.
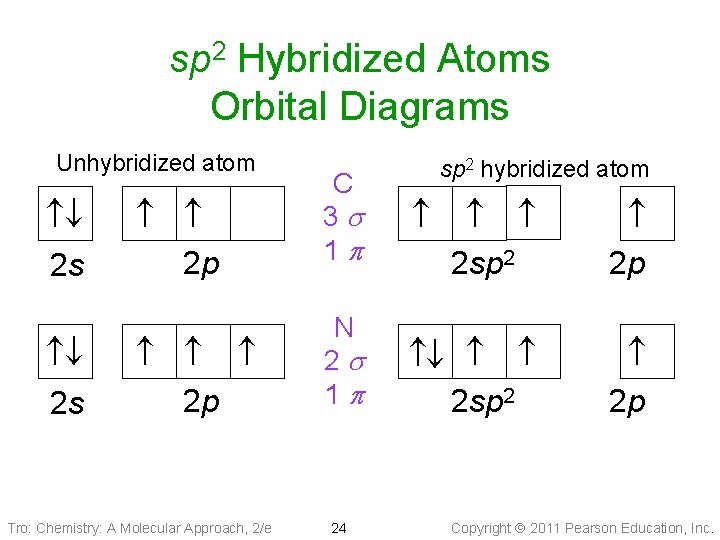
sp 2 Hybridized Atoms Orbital Diagrams 2 s 2 p Tro: Chemistry: A Molecular Approach, 2/e sp 2 hybridized atom C 3 s 1 p N 2 s 1 p 24 Unhybridized atom 2 sp 2 2 p Copyright 2011 Pearson Education, Inc.
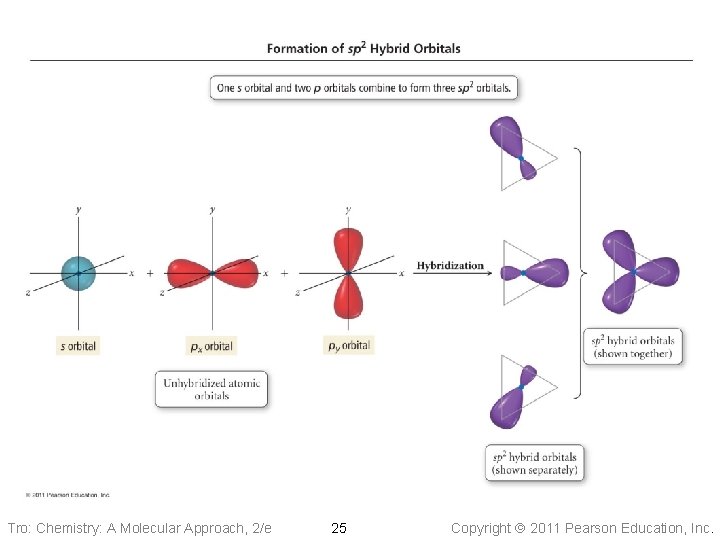
Tro: Chemistry: A Molecular Approach, 2/e 25 Copyright 2011 Pearson Education, Inc.
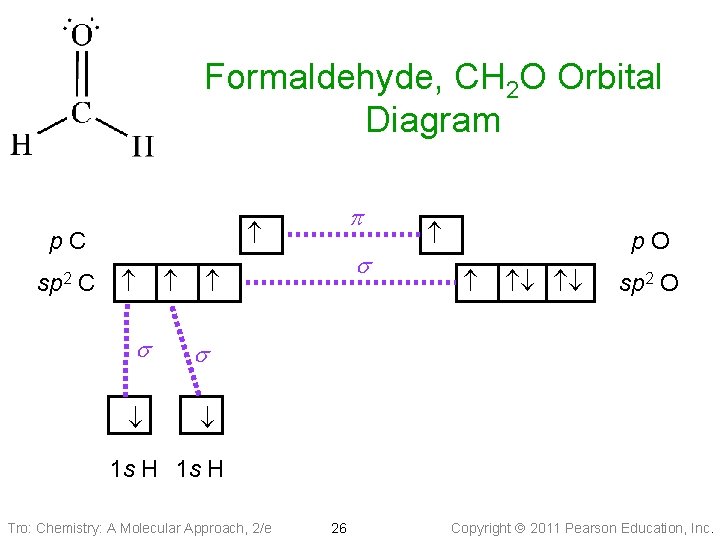
Formaldehyde, CH 2 O Orbital Diagram p. C s s s C sp 2 p p. O sp 2 O 1 s H Tro: Chemistry: A Molecular Approach, 2/e 26 Copyright 2011 Pearson Education, Inc.
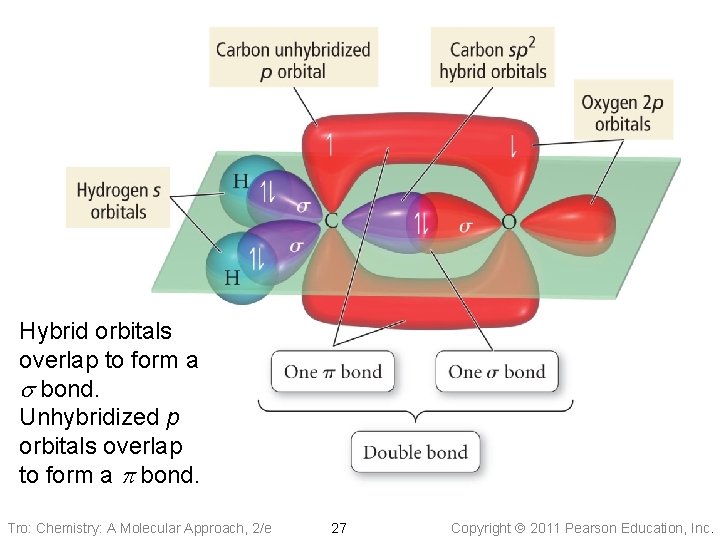
Hybrid orbitals overlap to form a s bond. Unhybridized p orbitals overlap to form a p bond. Tro: Chemistry: A Molecular Approach, 2/e 27 Copyright 2011 Pearson Education, Inc.
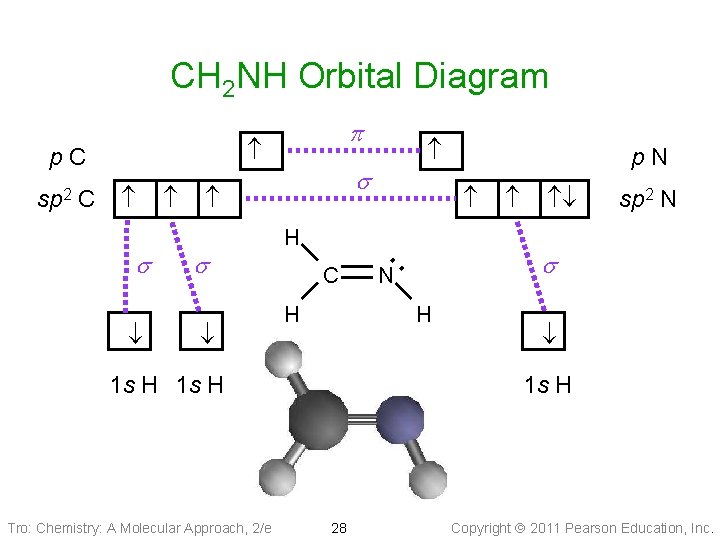
CH 2 NH Orbital Diagram s s s C H p. N ・・ N sp 2 N s H 1 s H Tro: Chemistry: A Molecular Approach, 2/e p. C sp 2 p 1 s H 28 Copyright 2011 Pearson Education, Inc.
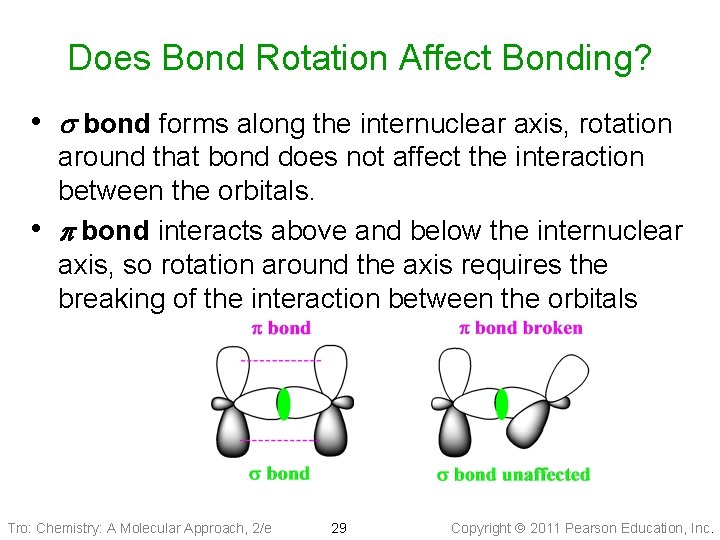
Does Bond Rotation Affect Bonding? • s bond forms along the internuclear axis, rotation • around that bond does not affect the interaction between the orbitals. p bond interacts above and below the internuclear axis, so rotation around the axis requires the breaking of the interaction between the orbitals Tro: Chemistry: A Molecular Approach, 2/e 29 Copyright 2011 Pearson Education, Inc.
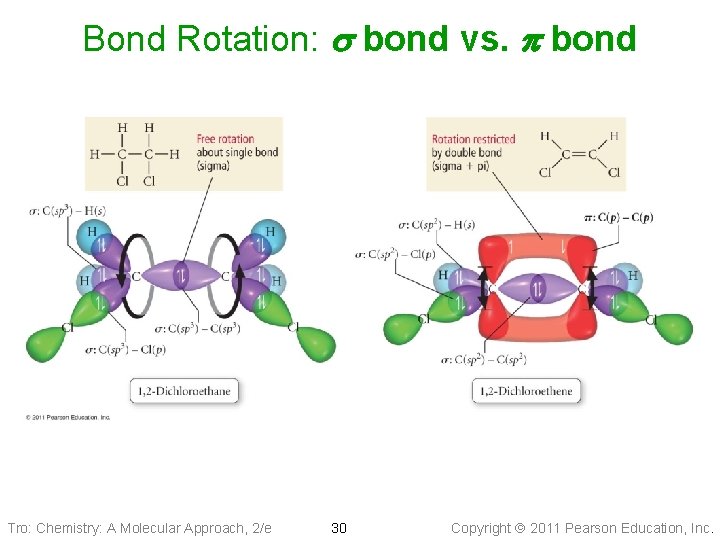
Bond Rotation: s bond vs. p bond Tro: Chemistry: A Molecular Approach, 2/e 30 Copyright 2011 Pearson Education, Inc.
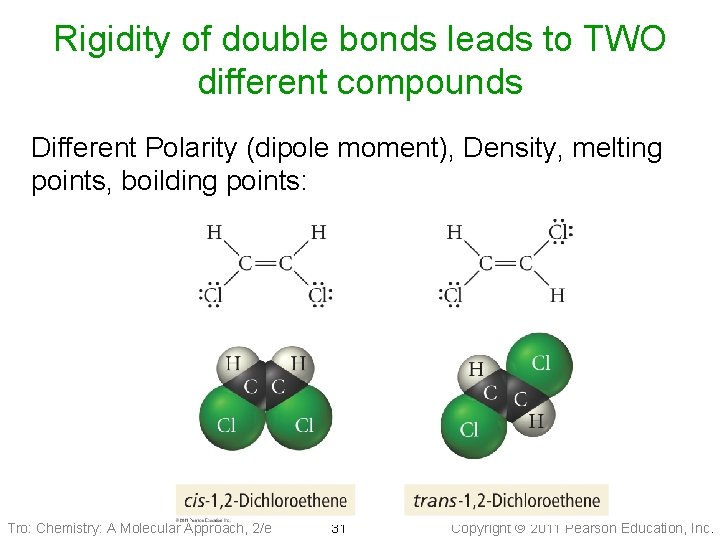
Rigidity of double bonds leads to TWO different compounds Different Polarity (dipole moment), Density, melting points, boilding points: Tro: Chemistry: A Molecular Approach, 2/e 31 Copyright 2011 Pearson Education, Inc.
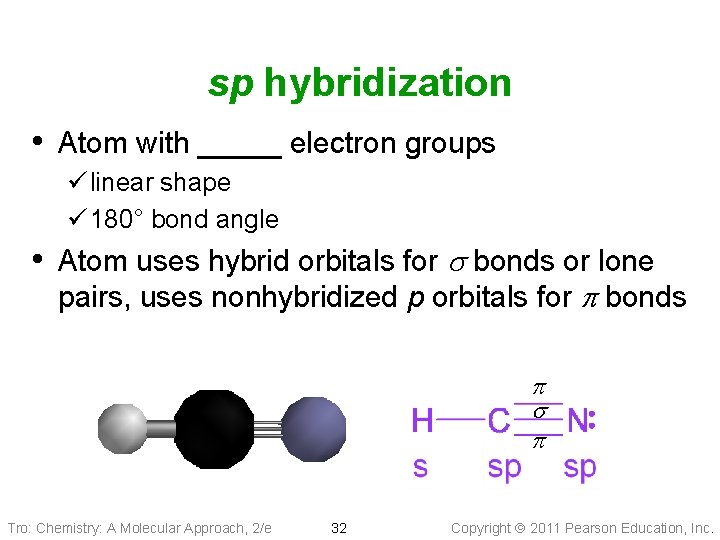
sp hybridization • Atom with _____ electron groups ü linear shape ü 180° bond angle • Atom uses hybrid orbitals for s bonds or lone pairs, uses nonhybridized p orbitals for p bonds p Tro: Chemistry: A Molecular Approach, 2/e 32 Copyright 2011 Pearson Education, Inc.
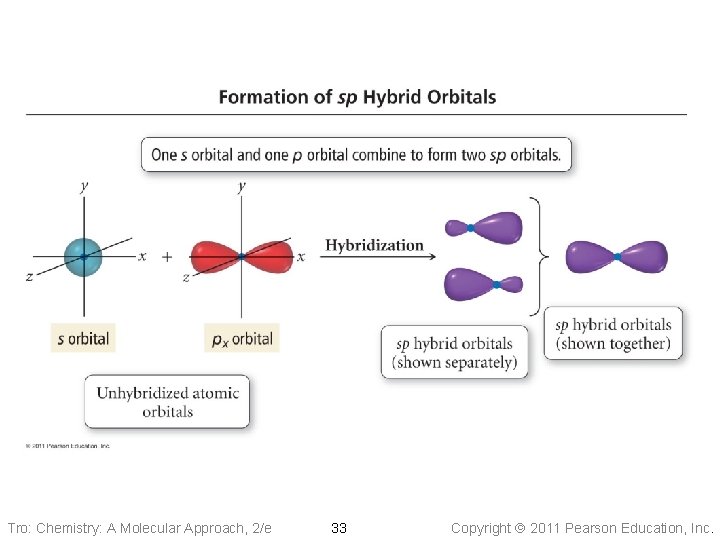
Tro: Chemistry: A Molecular Approach, 2/e 33 Copyright 2011 Pearson Education, Inc.
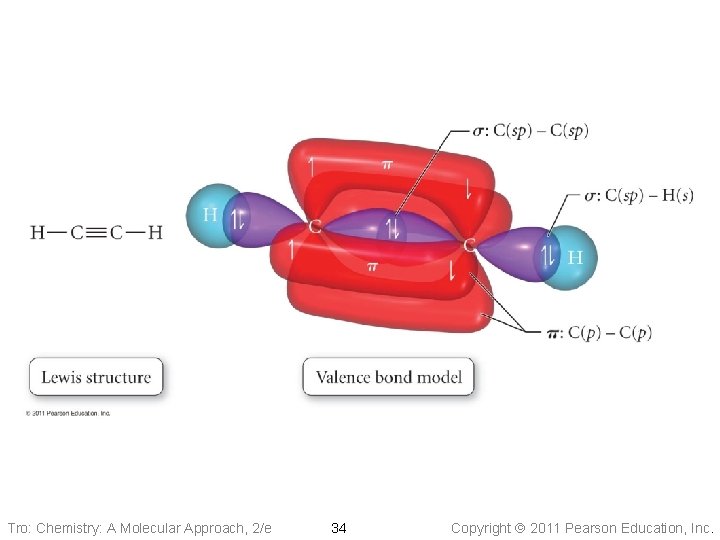
Tro: Chemistry: A Molecular Approach, 2/e 34 Copyright 2011 Pearson Education, Inc.
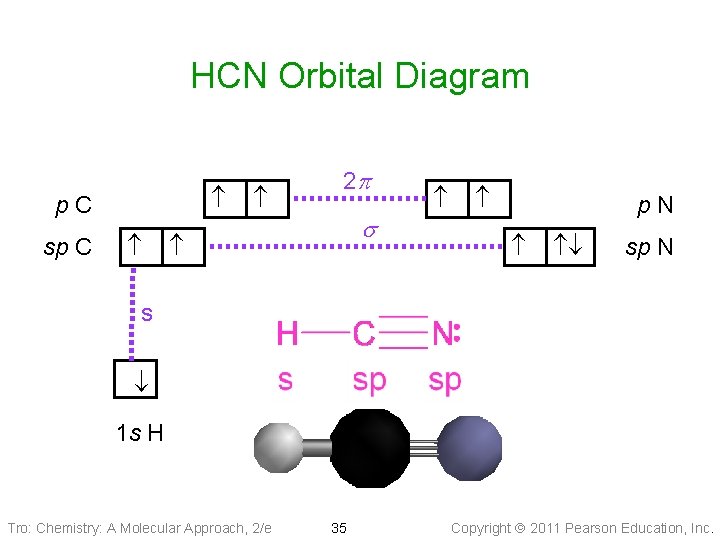
HCN Orbital Diagram p. C sp C 2 p s p. N sp N s 1 s H Tro: Chemistry: A Molecular Approach, 2/e 35 Copyright 2011 Pearson Education, Inc.
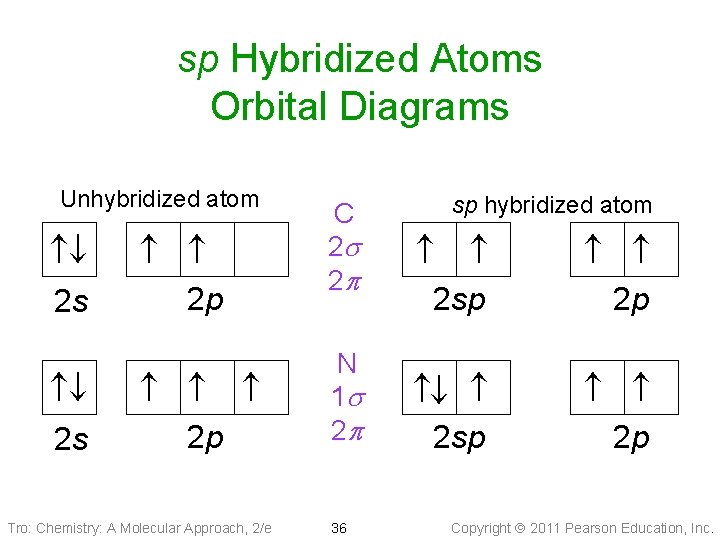
sp Hybridized Atoms Orbital Diagrams 2 s 2 p 2 p Tro: Chemistry: A Molecular Approach, 2/e C 2 s 2 p N 1 s 2 p 36 sp hybridized atom 2 sp Unhybridized atom 2 sp 2 p 2 p Copyright 2011 Pearson Education, Inc.
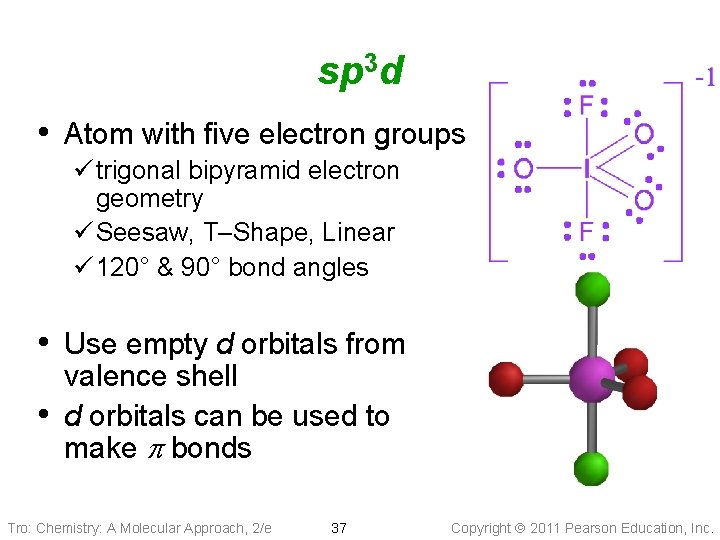
sp 3 d • Atom with five electron groups ü trigonal bipyramid electron geometry ü Seesaw, T–Shape, Linear ü 120° & 90° bond angles • Use empty d orbitals from • valence shell d orbitals can be used to make p bonds Tro: Chemistry: A Molecular Approach, 2/e 37 Copyright 2011 Pearson Education, Inc.
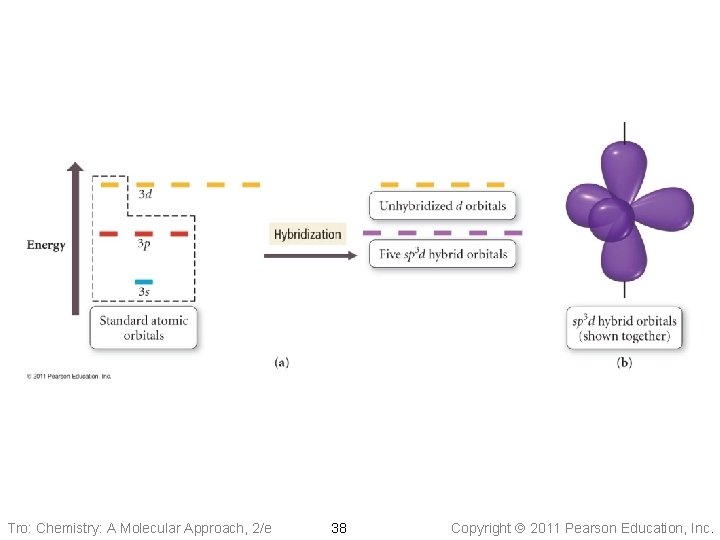
Tro: Chemistry: A Molecular Approach, 2/e 38 Copyright 2011 Pearson Education, Inc.
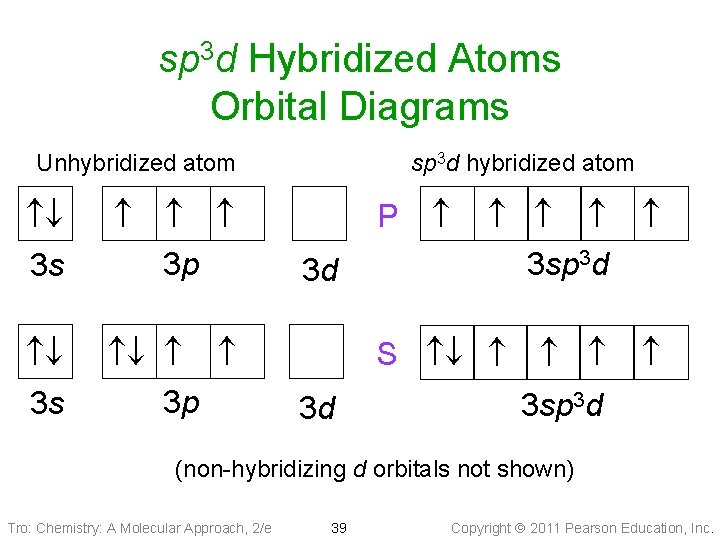
sp 3 d Hybridized Atoms Orbital Diagrams Unhybridized atom 3 s 3 p sp 3 d hybridized atom P 3 d 3 sp 3 d S 3 d 3 sp 3 d (non-hybridizing d orbitals not shown) Tro: Chemistry: A Molecular Approach, 2/e 39 Copyright 2011 Pearson Education, Inc.
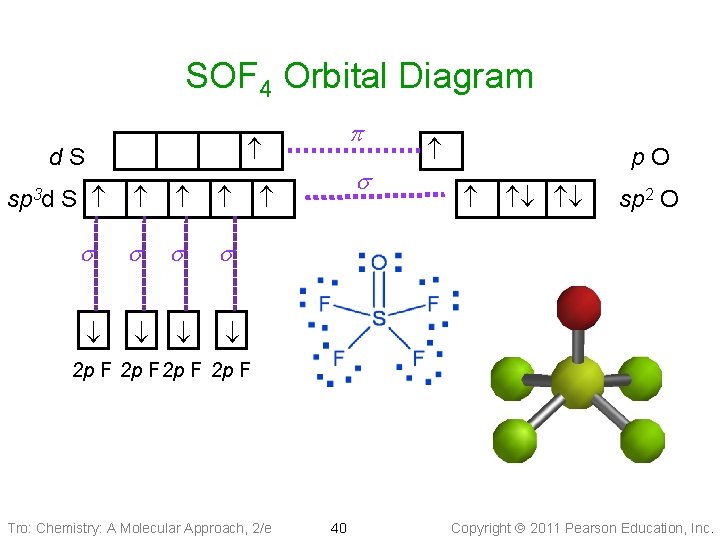
SOF 4 Orbital Diagram d. S S s s sp 3 d p s p. O sp 2 O 2 p F Tro: Chemistry: A Molecular Approach, 2/e 40 Copyright 2011 Pearson Education, Inc.

sp 3 d 2 • Atom with six electron groups around it ü octahedral electron geometry ü Square Pyramid, Square Planar ü 90° bond angles • Use empty d orbitals from • valence shell to form hybrid d orbitals can be used to make p bonds Tro: Chemistry: A Molecular Approach, 2/e 41 Copyright 2011 Pearson Education, Inc.
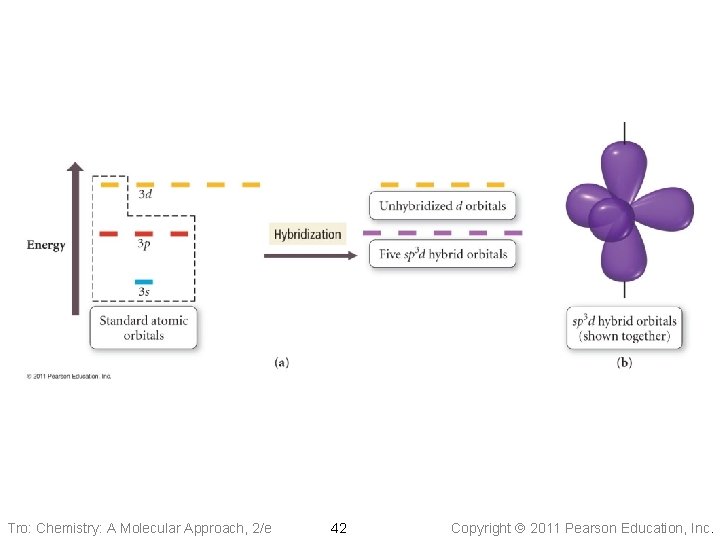
Tro: Chemistry: A Molecular Approach, 2/e 42 Copyright 2011 Pearson Education, Inc.
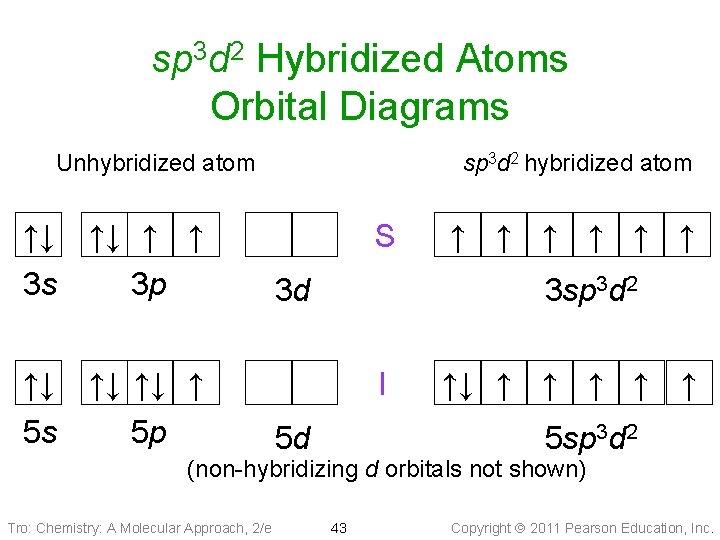
sp 3 d 2 Hybridized Atoms Orbital Diagrams Unhybridized atom ↑↓ ↑↓ ↑ ↑ 3 s 3 p ↑↓ ↑↓ ↑↓ ↑ 5 s 5 p sp 3 d 2 hybridized atom S 3 d ↑ ↑ ↑ 3 sp 3 d 2 I 5 d ↑↓ ↑ ↑ ↑ 5 sp 3 d 2 (non-hybridizing d orbitals not shown) Tro: Chemistry: A Molecular Approach, 2/e 43 Copyright 2011 Pearson Education, Inc.

Tro: Chemistry: A Molecular Approach, 2/e 44 Copyright 2011 Pearson Education, Inc.

Predicting Hybridization and Bonding Scheme 1. 2. 3. 4. 5. Start by drawing the Lewis structure Use VSEPR Theory to predict the electron group geometry around each central atom Select the hybridization scheme based on the electron group geometry Sketch the atomic and hybrid orbitals on the atoms in the molecule, showing overlap of the appropriate orbitals Label the bonds as s or p Tro: Chemistry: A Molecular Approach, 2/e 45 Copyright 2011 Pearson Education, Inc.
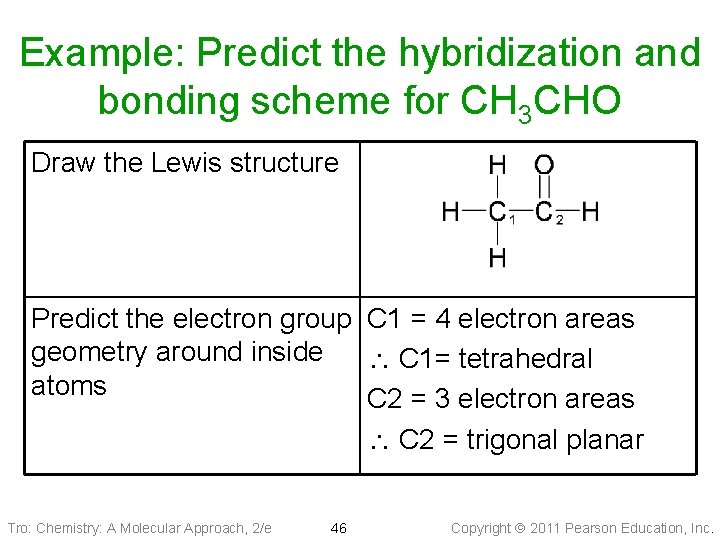
Example: Predict the hybridization and bonding scheme for CH 3 CHO Draw the Lewis structure Predict the electron group C 1 = 4 electron areas geometry around inside C 1= tetrahedral atoms C 2 = 3 electron areas C 2 = trigonal planar Tro: Chemistry: A Molecular Approach, 2/e 46 Copyright 2011 Pearson Education, Inc.
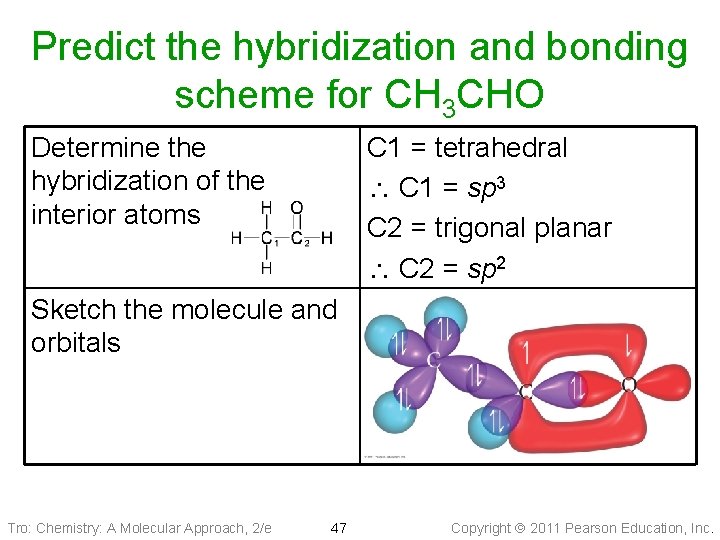
Predict the hybridization and bonding scheme for CH 3 CHO Determine the hybridization of the interior atoms C 1 = tetrahedral C 1 = sp 3 C 2 = trigonal planar C 2 = sp 2 Sketch the molecule and orbitals Tro: Chemistry: A Molecular Approach, 2/e 47 Copyright 2011 Pearson Education, Inc.
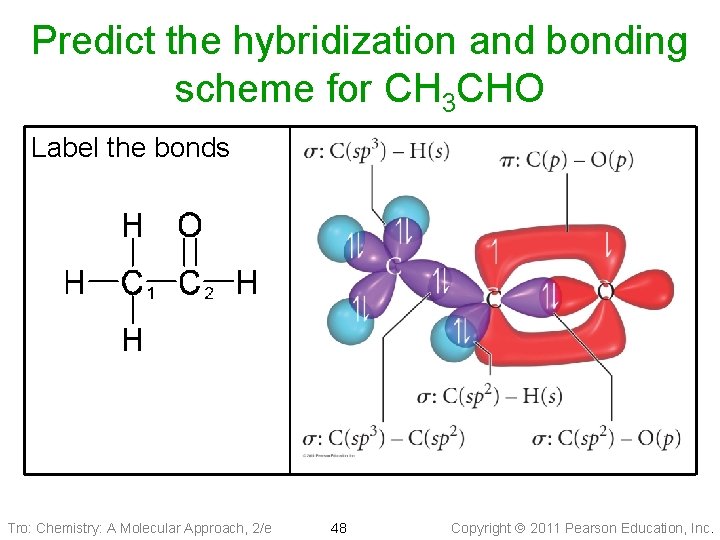
Predict the hybridization and bonding scheme for CH 3 CHO Label the bonds Tro: Chemistry: A Molecular Approach, 2/e 48 Copyright 2011 Pearson Education, Inc.
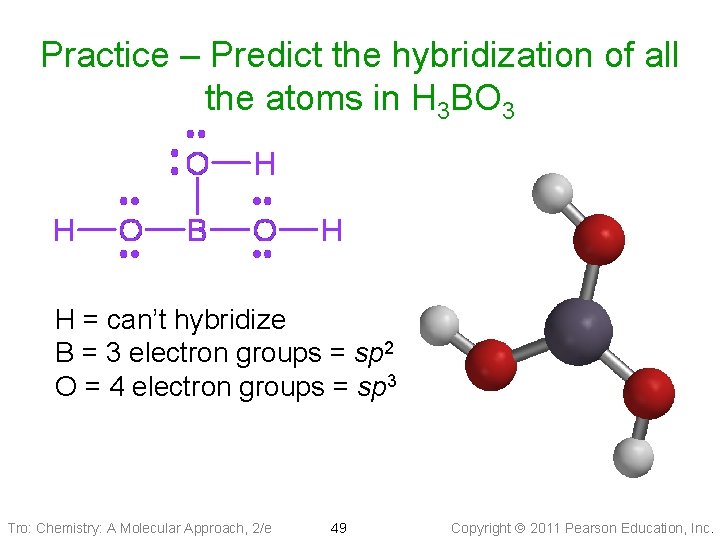
Practice – Predict the hybridization of all the atoms in H 3 BO 3 H = can’t hybridize B = 3 electron groups = sp 2 O = 4 electron groups = sp 3 Tro: Chemistry: A Molecular Approach, 2/e 49 Copyright 2011 Pearson Education, Inc.
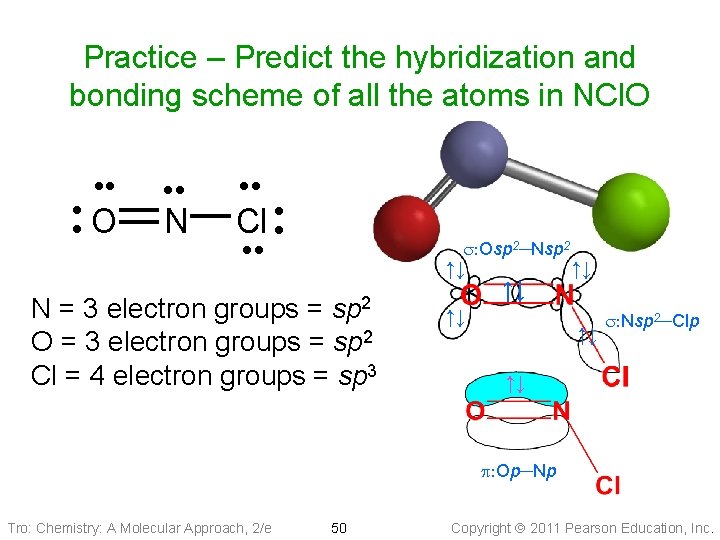
Practice – Predict the hybridization and bonding scheme of all the atoms in NCl. O • • • O • • • N • • Cl • • s: Osp 2─Nsp 2 ↑↓ sp 2 N = 3 electron groups = O = 3 electron groups = sp 2 Cl = 4 electron groups = sp 3 ↑↓ ↑↓ s: Nsp 2─Clp ↑↓ p: Op─Np Tro: Chemistry: A Molecular Approach, 2/e 50 Copyright 2011 Pearson Education, Inc.

Problems with Valence Bond Theory • VB theory predicts many properties better than Lewis theory ü bonding schemes, bond strengths, bond lengths, bond rigidity • However, VB theory doesn’t predict many other properties ü magnetic behavior of O 2 • In addition, VB theory treats the electrons as localized in orbitals on the atoms in the molecule Tro: Chemistry: A Molecular Approach, 2/e 51 Copyright 2011 Pearson Education, Inc.

Molecular Orbital Theory • In MO theory, solving Schrödinger’s • wave equation to calculate a set of molecular orbitals In this treatment, the electrons belong to the whole molecule – so the orbitals belong to the whole molecule ü delocalization Tro: Chemistry: A Molecular Approach, 2/e 52 Copyright 2011 Pearson Education, Inc.

LCAO • The simplest guess starts with the atomic orbitals of the atoms adding together to make molecular orbitals – this is called the Linear Combination of Atomic Orbitals method ü weighted sum • Because the orbitals are wave functions, the waves can combine either constructively or destructively Tro: Chemistry: A Molecular Approach, 2/e 53 Copyright 2011 Pearson Education, Inc.

Molecular Orbitals • Wave functions combine constructively forms molecular orbital _____ energy than the original atomic orbitals Bonding Molecular Orbital ü s, p ü most of the electron density between the nuclei • Wave functions combine destructively forms molecular orbital ______ energy than the original atomic orbitals Antibonding Molecular Orbital ü s*, p* ü most of the electron density outside the nuclei ü nodes between nuclei Tro: Chemistry: A Molecular Approach, 2/e 54 Copyright 2011 Pearson Education, Inc.
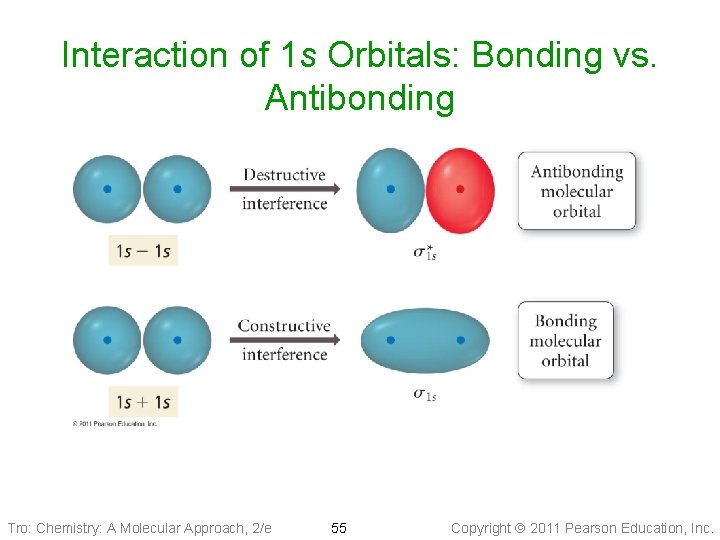
Interaction of 1 s Orbitals: Bonding vs. Antibonding Tro: Chemistry: A Molecular Approach, 2/e 55 Copyright 2011 Pearson Education, Inc.

Molecular Orbital Theory • Electrons in bonding MOs are stabilizing ü lower energy than the atomic orbitals • Electrons in antibonding MOs are destabilizing ü higher in energy than atomic orbitals ü electron density located outside the internuclear axis ü electrons in antibonding orbitals cancel stability gained by electrons in bonding orbitals Tro: Chemistry: A Molecular Approach, 2/e 56 Copyright 2011 Pearson Education, Inc.
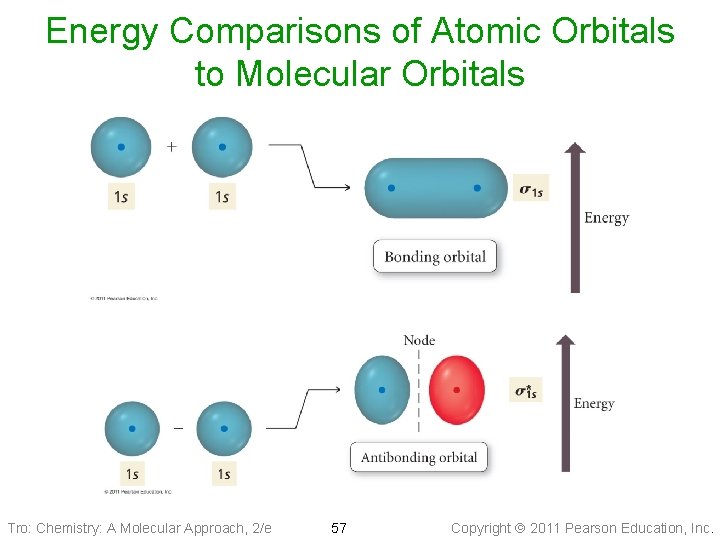
Energy Comparisons of Atomic Orbitals to Molecular Orbitals Tro: Chemistry: A Molecular Approach, 2/e 57 Copyright 2011 Pearson Education, Inc.

MO and Properties • Bond Order = difference between number of electrons in bonding and antibonding orbitals ü only need to consider valence electrons ü may be a fraction ü higher bond order = stronger and shorter bonds ü if bond order = 0, then bond is unstable compared to individual atoms and no bond will form • Paramagnetic: unpaired electrons in MO ü if all electrons paired it is diamagnetic Tro: Chemistry: A Molecular Approach, 2/e 58 Copyright 2011 Pearson Education, Inc.
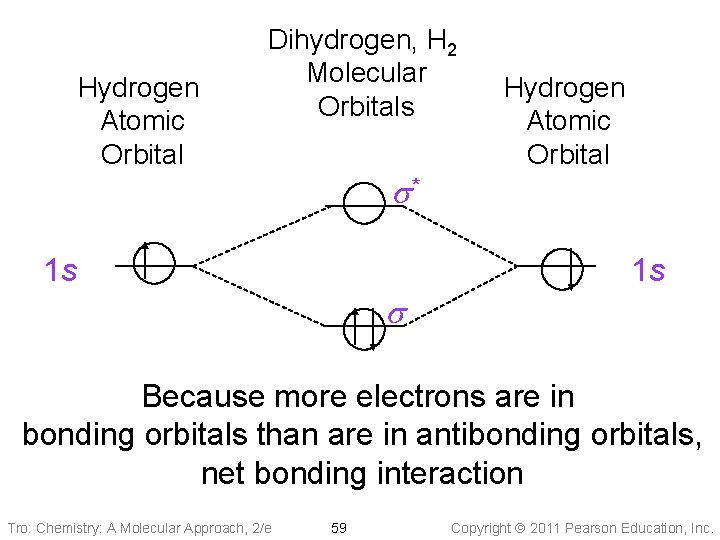
Hydrogen Atomic Orbital Dihydrogen, H 2 Molecular Orbitals s* 1 s Hydrogen Atomic Orbital 1 s s Because more electrons are in bonding orbitals than are in antibonding orbitals, net bonding interaction Tro: Chemistry: A Molecular Approach, 2/e 59 Copyright 2011 Pearson Education, Inc.
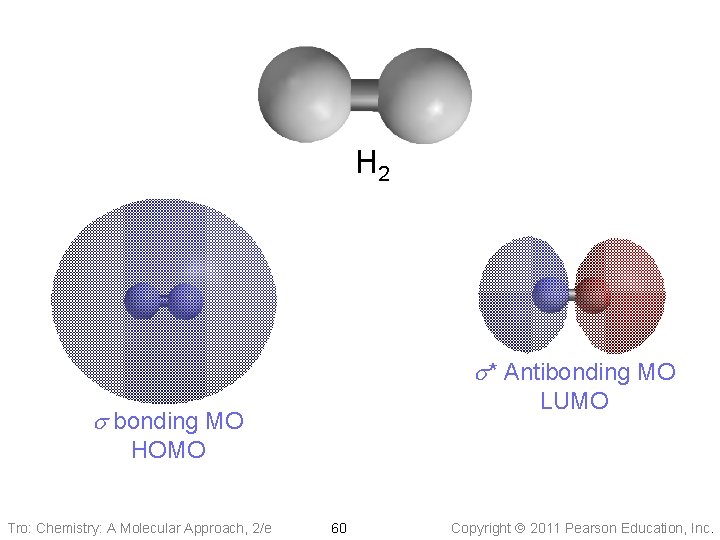
H 2 s* Antibonding MO LUMO s bonding MO HOMO Tro: Chemistry: A Molecular Approach, 2/e 60 Copyright 2011 Pearson Education, Inc.
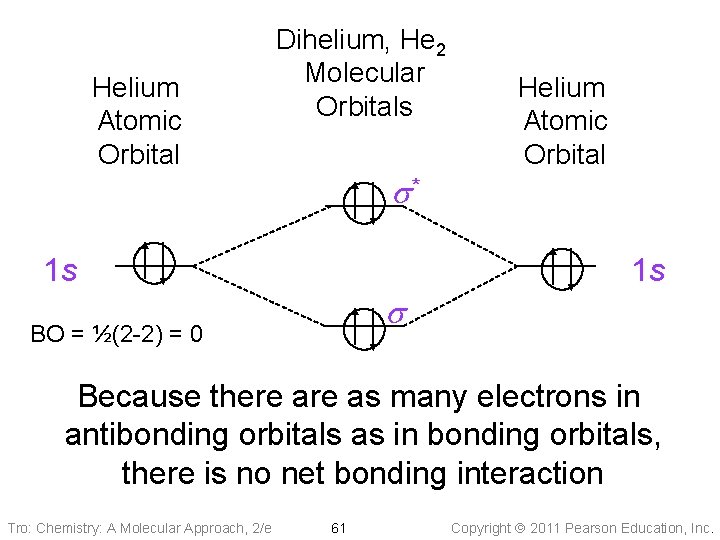
Helium Atomic Orbital Dihelium, He 2 Molecular Orbitals s* 1 s Helium Atomic Orbital 1 s s BO = ½(2 -2) = 0 Because there as many electrons in antibonding orbitals as in bonding orbitals, there is no net bonding interaction Tro: Chemistry: A Molecular Approach, 2/e 61 Copyright 2011 Pearson Education, Inc.
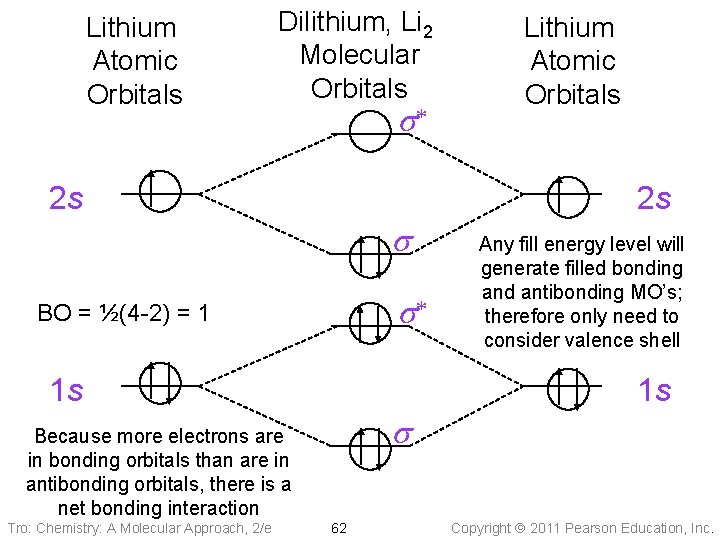
Lithium Atomic Orbitals Dilithium, Li 2 Molecular Orbitals s* 2 s 2 s s s* BO = ½(4 -2) = 1 1 s Any fill energy level will generate filled bonding and antibonding MO’s; therefore only need to consider valence shell 1 s s Because more electrons are in bonding orbitals than are in antibonding orbitals, there is a net bonding interaction Tro: Chemistry: A Molecular Approach, 2/e Lithium Atomic Orbitals 62 Copyright 2011 Pearson Education, Inc.
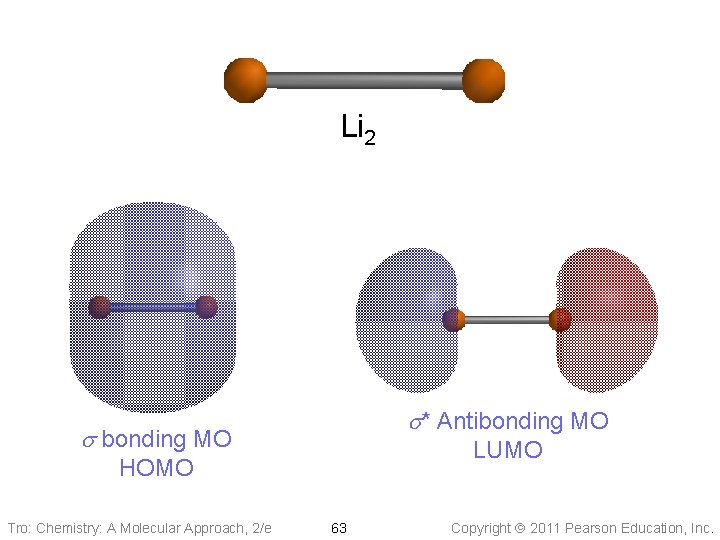
Li 2 s* Antibonding MO s bonding MO LUMO HOMO Tro: Chemistry: A Molecular Approach, 2/e 63 Copyright 2011 Pearson Education, Inc.
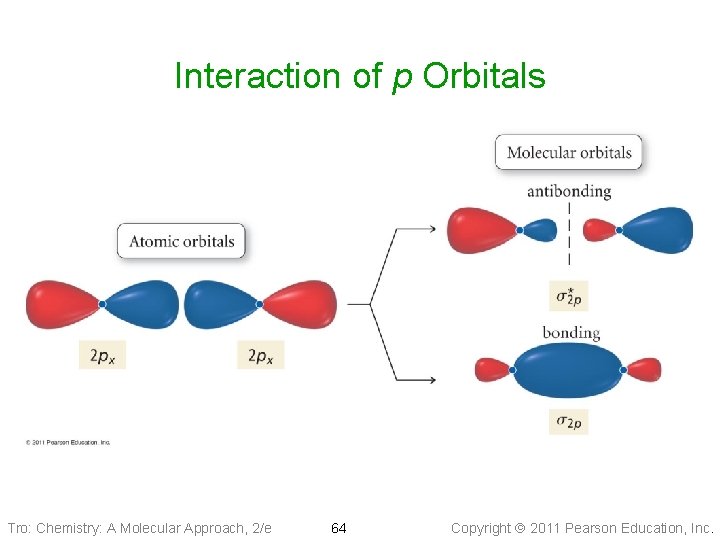
Interaction of p Orbitals Tro: Chemistry: A Molecular Approach, 2/e 64 Copyright 2011 Pearson Education, Inc.
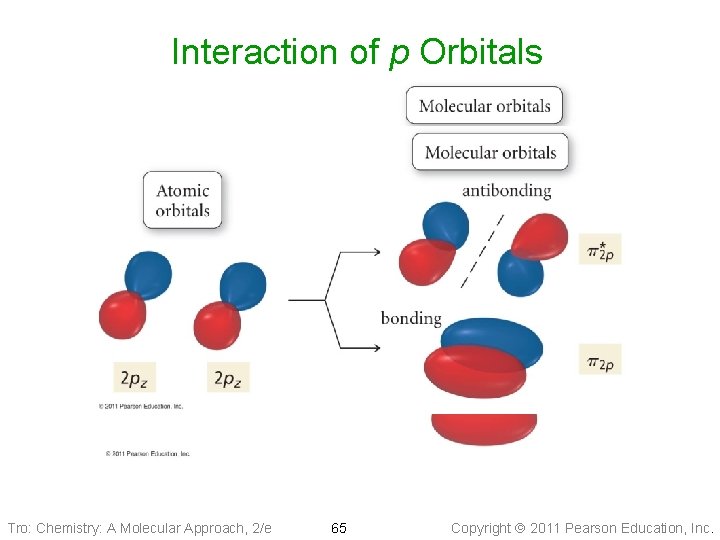
Interaction of p Orbitals Tro: Chemistry: A Molecular Approach, 2/e 65 Copyright 2011 Pearson Education, Inc.
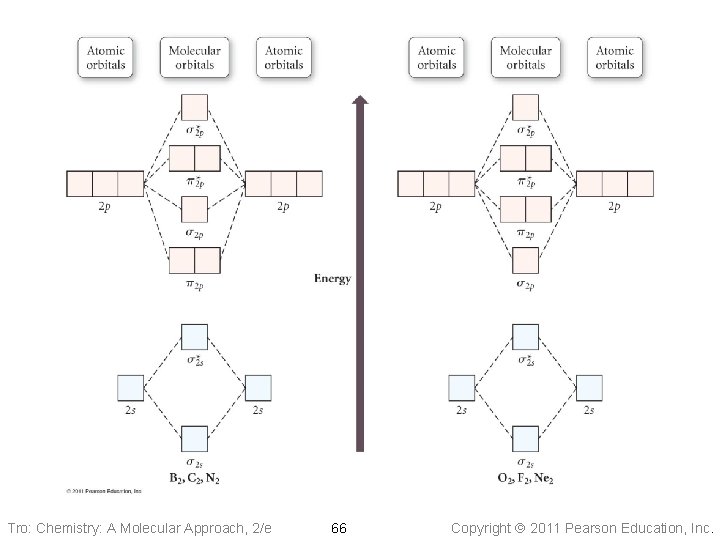
Tro: Chemistry: A Molecular Approach, 2/e 66 Copyright 2011 Pearson Education, Inc.

O 2 • Dioxygen is paramagnetic • Paramagnetic material has unpaired electrons • Neither Lewis theory nor valence bond theory predict this result Tro: Chemistry: A Molecular Approach, 2/e 67 Copyright 2011 Pearson Education, Inc.

O 2 as Described by Lewis and VB Theory Tro: Chemistry: A Molecular Approach, 2/e 68 Copyright 2011 Pearson Education, Inc.
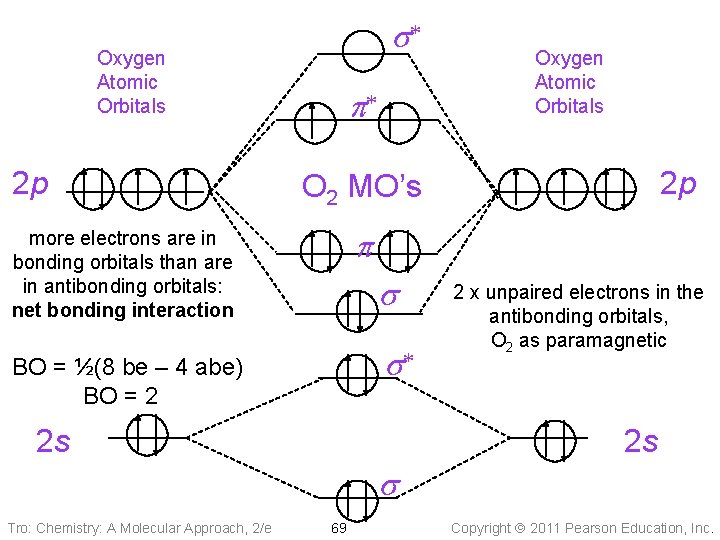
s* Oxygen Atomic Orbitals 2 p p* Oxygen Atomic Orbitals 2 p O 2 MO’s p more electrons are in bonding orbitals than are in antibonding orbitals: net bonding interaction s s* BO = ½(8 be – 4 abe) BO = 2 2 s 2 x unpaired electrons in the antibonding orbitals, O 2 as paramagnetic 2 s s Tro: Chemistry: A Molecular Approach, 2/e 69 Copyright 2011 Pearson Education, Inc.
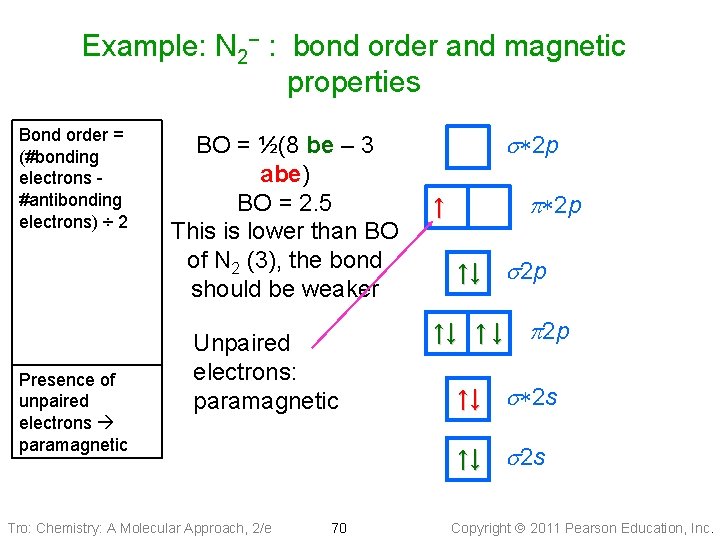
Example: N 2− : bond order and magnetic properties Bond order = (#bonding electrons #antibonding electrons) ÷ 2 Presence of unpaired electrons paramagnetic BO = ½(8 be – 3 abe) BO = 2. 5 This is lower than BO of N 2 (3), the bond should be weaker Unpaired electrons: paramagnetic Tro: Chemistry: A Molecular Approach, 2/e s*2 p p*2 p ↑ ↑↓ s 2 p ↑↓ ↑ ↓ p 2 p ↑↓ s*2 s ↑↓ s 2 s 70 Copyright 2011 Pearson Education, Inc.

Heteronuclear Diatomic Molecules & Ions • When the combining atomic orbitals are • identical and equal energy, the contribution of each atomic orbital to the molecular orbital is equal When the combining atomic orbitals are different types and energies, the atomic orbital closest in energy to the molecular orbital contributes more to the molecular orbital Tro: Chemistry: A Molecular Approach, 2/e 71 Copyright 2011 Pearson Education, Inc.

Heteronuclear Diatomic Molecules & Ions • The more electronegative an atom is, the • • • lower in energy are its orbitals Lower energy atomic orbitals contribute more to the bonding MOs Higher energy atomic orbitals contribute more to the antibonding MOs Nonbonding MOs remain localized on the atom donating its atomic orbitals Tro: Chemistry: A Molecular Approach, 2/e 72 Copyright 2011 Pearson Education, Inc.
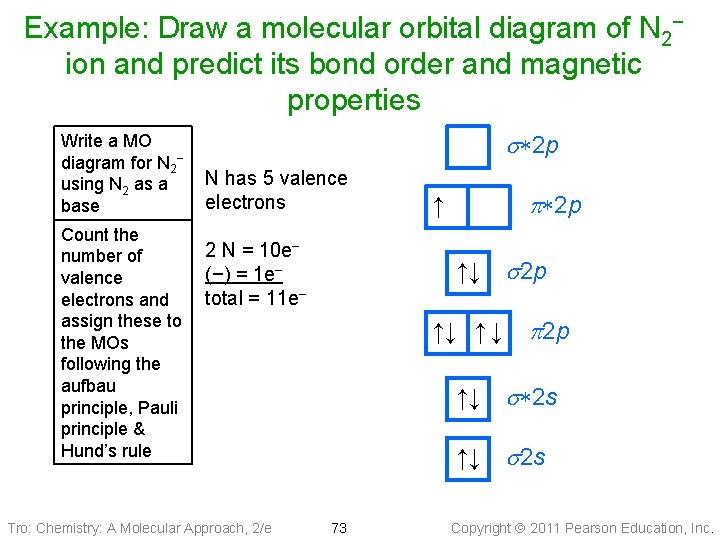
Example: Draw a molecular orbital diagram of N 2− ion and predict its bond order and magnetic properties Write a MO diagram for N 2− using N 2 as a base Count the number of valence electrons and assign these to the MOs following the aufbau principle, Pauli principle & Hund’s rule s*2 p N has 5 valence electrons 2 N = 10 e− (−) = 1 e− total = 11 e− Tro: Chemistry: A Molecular Approach, 2/e p*2 p ↑ ↑↓ s 2 p ↑↓ ↑ ↓ ↑↓ p 2 p s*2 s ↑↓ s 2 s 73 Copyright 2011 Pearson Education, Inc.
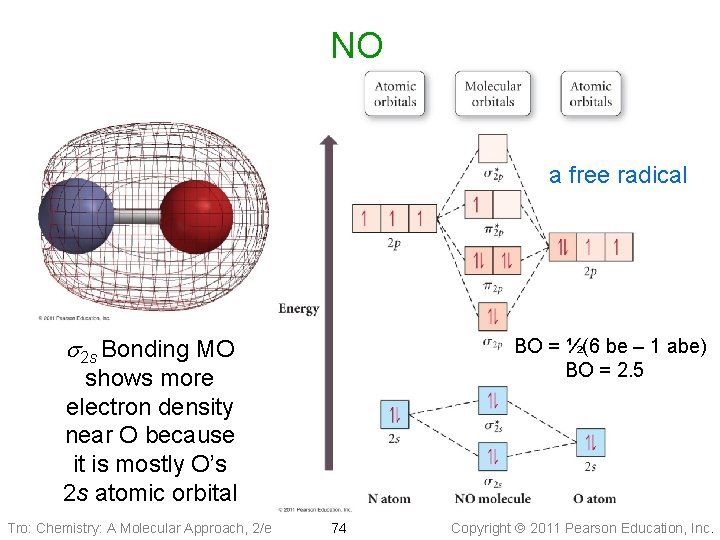
NO a free radical s 2 s Bonding MO BO = ½(6 be – 1 abe) BO = 2. 5 shows more electron density near O because it is mostly O’s 2 s atomic orbital Tro: Chemistry: A Molecular Approach, 2/e 74 Copyright 2011 Pearson Education, Inc.

Practice – Draw a molecular orbital diagram of C 2+ and predict its bond order and magnetic properties Tro: Chemistry: A Molecular Approach, 2/e 75 Copyright 2011 Pearson Education, Inc.
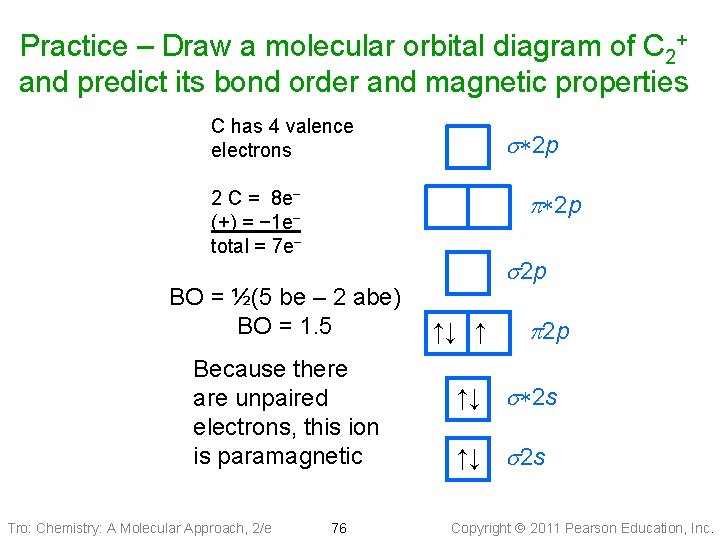
Practice – Draw a molecular orbital diagram of C 2+ and predict its bond order and magnetic properties C has 4 valence electrons s*2 p 2 C = 8 e− (+) = − 1 e− total = 7 e− p*2 p BO = ½(5 be – 2 abe) BO = 1. 5 Because there are unpaired electrons, this ion is paramagnetic Tro: Chemistry: A Molecular Approach, 2/e 76 s 2 p ↑↓ ↑ ↑↓ p 2 p s*2 s ↑↓ s 2 s Copyright 2011 Pearson Education, Inc.
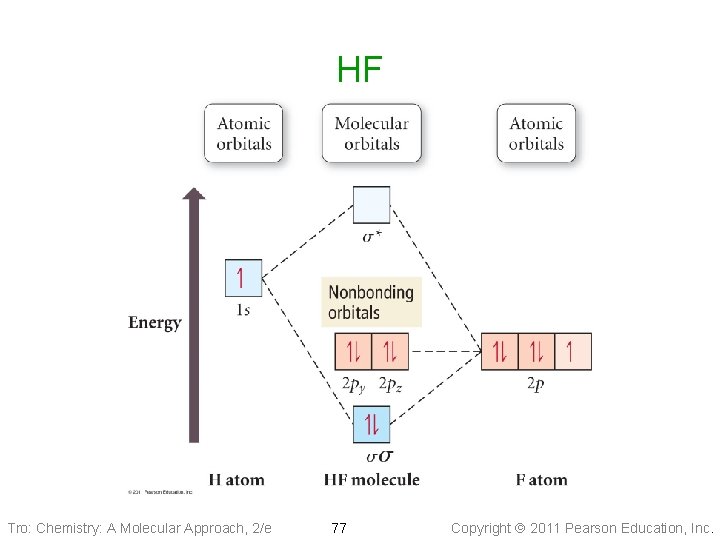
HF Tro: Chemistry: A Molecular Approach, 2/e 77 Copyright 2011 Pearson Education, Inc.

Polyatomic Molecules • When many atoms are combined together, the • atomic orbitals of all the atoms are combined to make a set of molecular orbitals, which are delocalized over the entire molecule Gives results that better match real molecule properties than either Lewis or valence bond theories Tro: Chemistry: A Molecular Approach, 2/e 78 Copyright 2011 Pearson Education, Inc.
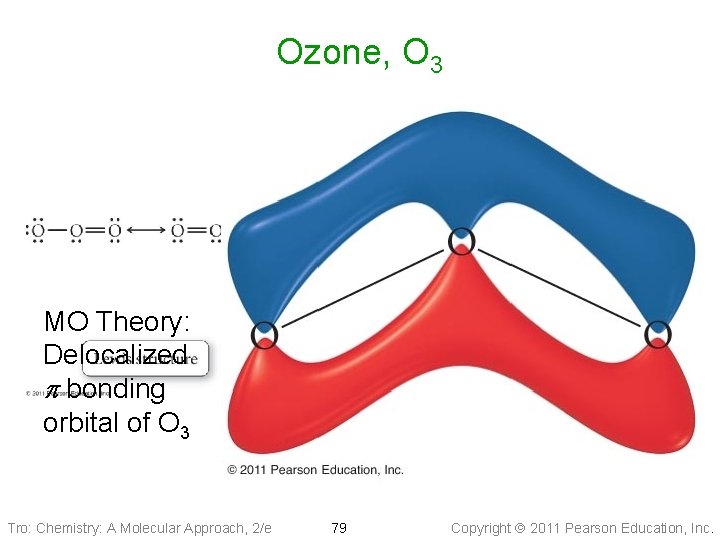
Ozone, O 3 MO Theory: Delocalized p bonding orbital of O 3 Tro: Chemistry: A Molecular Approach, 2/e 79 Copyright 2011 Pearson Education, Inc.
- Slides: 79