Kinetic Theory Kinetic Theory Kinetic refers to movement










































- Slides: 45

Kinetic Theory

Kinetic Theory • “Kinetic” refers to movement

Kinetic Theory • “Kinetic” refers to movement • “Theory, ” a unifying idea that is useful for understanding a wide variety of observations

Kinetic Theory • The kinetic theory states that submicroscopic particles of all matter are in constant, random motion. • The Kinetic Theory is a unifying concept that uses the idea of moving particles to explain gases, liquids, solids, phase changes and the effect of temperature and pressure on these.

Kinetic Theory 3 Assumptions necessary to use theory:

Kinetic Theory 3 Assumptions necessary to use theory: • All matter is made of particles (atoms, ions or molecules)

Kinetic Theory 3 Assumptions necessary to use theory: • All matter is made of particles (atoms, ions or molecules) • The particles of matter are in constant motion

Kinetic Theory 3 Assumptions necessary to use theory: • All matter is made of particles (atoms, ions or molecules) • The particles of matter are in constant motion • All collisions are perfectly elastic (no energy is lost)

Solid

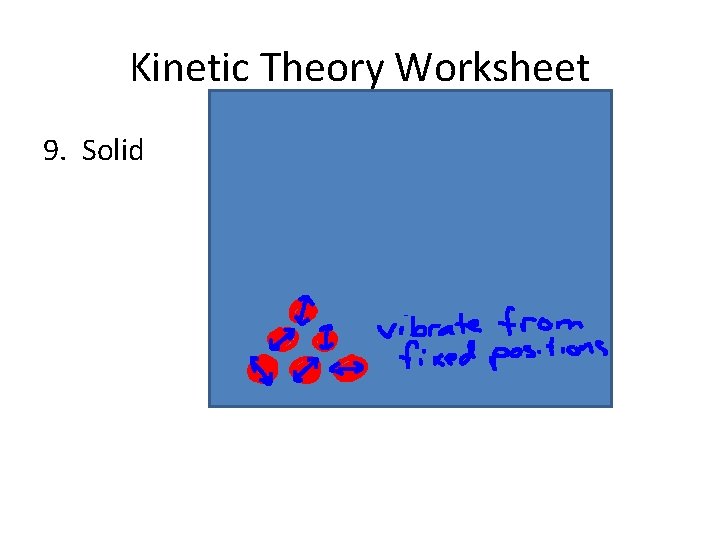
Solid • Solids have their own shape and volume regardless of their containers.

Solid • Solids have their own shape and volume regardless of their containers. • The particles in a solid vibrate but don’t have enough energy to move past one another

Solid • Solids have their own shape and volume regardless of their containers. • The particles in a solid vibrate but don’t have enough energy to move past one another • But, if you heat a solid (giving it more energy) it will ______ and become a ….

Liquid

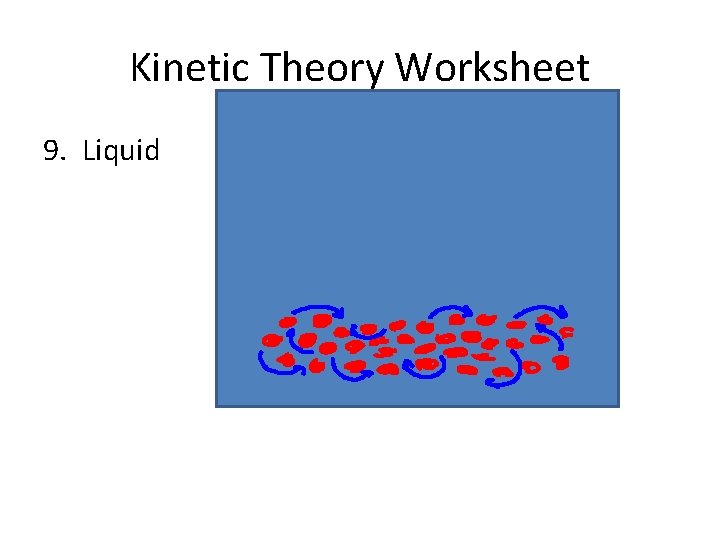
Liquid • The particles in a liquid have enough energy to flow past one another, and thus take the shape of whatever container they are in.

Liquid • The particles in a liquid have enough energy to flow past one another, and thus take the shape of whatever container they are in. • But they don’t have enough energy to break the attractive forces between them so they remain in contact and maintain their own volume.

Liquid • The particles in a liquid have enough energy to flow past one another, and thus take the shape of whatever container they are in. • But they don’t have enough energy to break the attractive forces between them so they remain in contact and maintain their own volume. • If you heat a liquid (giving it more energy) it will _____, and become a …

Gas

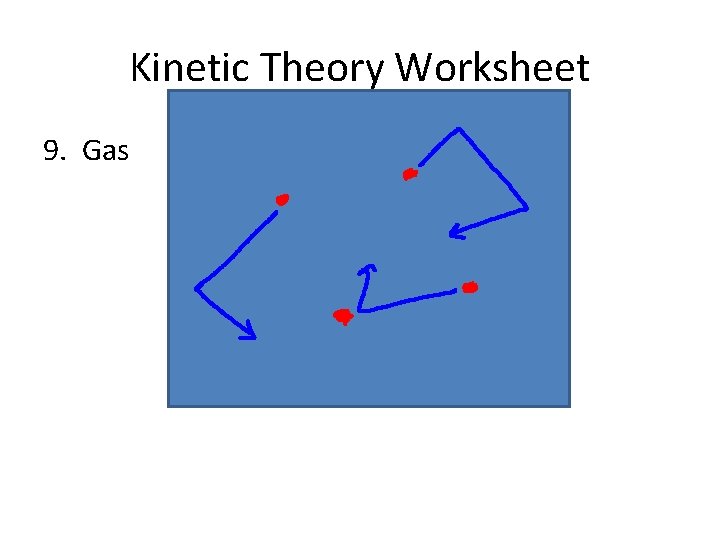
Gas • The particles of a gas have enough energy to overwhelm any attractive forces between them. If they collide, they bounce off without losing energy.

Gas • The particles of a gas have enough energy to overwhelm any attractive forces between them. If they collide, they bounce off without losing energy. • Gases take the shape and volume of their containers.

Gas • The particles of a gas have enough energy to overwhelm any attractive forces between them. If they collide, they bounce off without losing energy. • Gases take the shape and volume of their containers. • If you cool a gas, it _____ and becomes a…

Liquid • If you cool a liquid, the particles will lose energy and will _____ to become a …

Solid • If you cool a solid, it will lose energy and vibrate more and more slowly until a temperature is reached at which no movement occurs.

Solid • If you cool a solid, it will lose energy and vibrate more and more slowly until a temperature is reached at which no movement occurs. • This temperature is called ABSOLUTE ZERO, and is theoretically the coldest possible temperature.

Temperature • We will soon be defining temperature as a measure of the average kinetic energy of a substance.

Simulation • (hit escape then double click the link below)

Kinetic Theory Worksheet 1. What is the main idea of the kinetic theory?

Kinetic Theory Worksheet 1. What is the main idea of the kinetic theory? (the movement of particles causes many of the properties of matter that we observe)

Kinetic Theory Worksheet 2. What are three assumptions one must make to use theory?

Kinetic Theory Worksheet 2. What are three assumptions one must make to use theory? Matter is made of particles Particles are in constant motion Collisions are elastic

Kinetic Theory Worksheet 3. What are three most common states of matter on Earth?

Kinetic Theory Worksheet 3. What are three most common states of matter on Earth? Solid, liquid, gas

Kinetic Theory Worksheet 4. In which state(s) are the particles separated by much empty space?

Kinetic Theory Worksheet 4. In which state(s) are the particles separated by much empty space? gas

Kinetic Theory Worksheet 5. In which state(s) are the particles in contact with one another?

Kinetic Theory Worksheet 5. In which state(s) are the particles in contact with one another? Solid and liquid

Kinetic Theory Worksheet 6. In which state(s) are the particles in contact with one another but have enough kinetic energy to flow past one another?

Kinetic Theory Worksheet 6. In which state(s) are the particles in contact with one another but have enough kinetic energy to flow past one another? liquid

Kinetic Theory Worksheet 7. In which state(s) are the particles in contact with one another but don’t have enough kinetic energy to move past one another, but instead vibrate from their fixed positions?

Kinetic Theory Worksheet 7. In which state(s) are the particles in contact with one another but don’t have enough kinetic energy to move past one another, but instead vibrate from their fixed positions? solid

Kinetic Theory Worksheet 8. Which state would be most compressible? Why?

Kinetic Theory Worksheet 8. Which state would be most compressible? Why? Gas. Because there is empty space around the particles.

Kinetic Theory Worksheet 9. Gas

Kinetic Theory Worksheet 9. Liquid

Kinetic Theory Worksheet 9. Solid
