States of Matter A Matter of Kinetic Energy












- Slides: 21

States of Matter A Matter of Kinetic Energy

Types of States of Matter • • • Solid Liquid Gas Plasma Beam BEC, or Bose-Einstein Condensate – Zero State of Matter – Most Dense

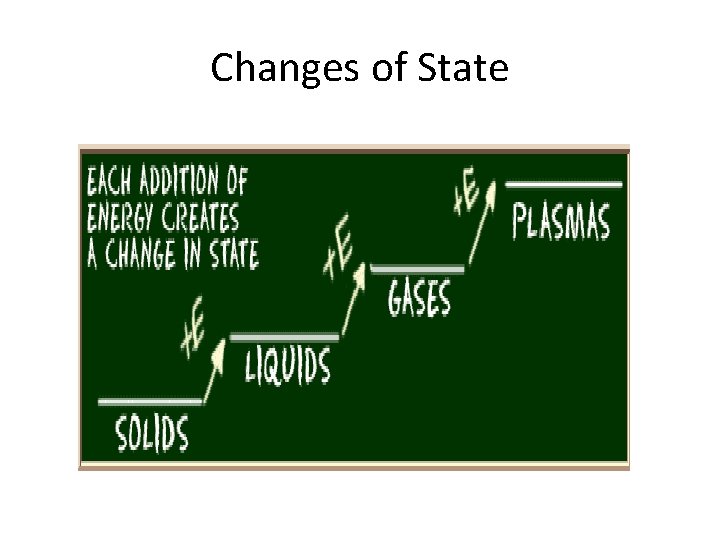
Changes of State

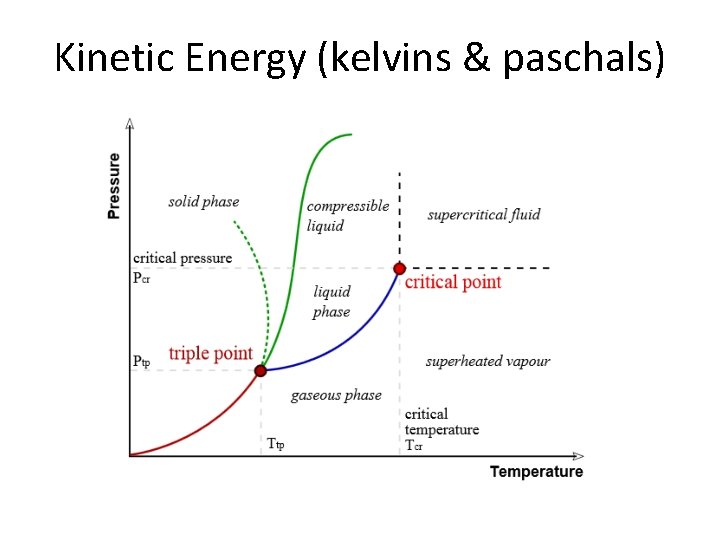
Kinetic Energy (kelvins & paschals)

chemwiki. ucdavis. edu/Physical_Chemistry/Physical_Properties_ of_Matter/Supercritical_Fluids • Supercritical fluids are useful in science today – extraction of floral fragrance – the process of creating decaffeinated coffee – food science and functional food ingredients – pharmaceuticals, cosmetics, polymers, powders, bio- and functional materials – nano-systems, natural products, biotechnology, fossil & biofuels, microelectronics & environment (Bottini 133).

www. engineeringtoolbox. com/vapor-steam-d_609 • Superheated Vapor • When the temperature is higher than the boiling point @ a given pressure. • Vapor cannot exist in contact with the fluid, nor contain fluid particles. • Increase in pressure or decrease in temperature will not, within limits, condensate out liquid particles in the vapor. • Highly superheated vapors are gases that approximately follow the general gas law.

Critical Temp & Pressure • Critical Temperature – The temperature at which only gas exists, regardless of its pressure • Critical Pressure – The lowest pressure at which liquids exist at critical temperature • Critical Point – The intersection of critical temperature & pressure

Kinetic-Molecular Theory of Gases • Ideal gas = hypothetical gas perfectly aligns with all kinetic-molecular theory assumptions • Five Assumptions – Distance between molecules dwarfs actual size – All collisions are perfectly elastic – Particles are in continuous, rapid, random motion – Particles have NO attraction to each other – Temperature = average kinetic energy of particles

Nature of Gases • Ideal vs. Real – Real approaches ideal @ low pressure/ high temp • • • Expansion – molecules fill entire space Fluidity – no intermolecular attractions Density - ~ 10 -3 of liquid or solid state Compressibility – 100 X more molecules Diffusion & Effusion – Spontaneous mixing via random motion – Passing through tiny opening

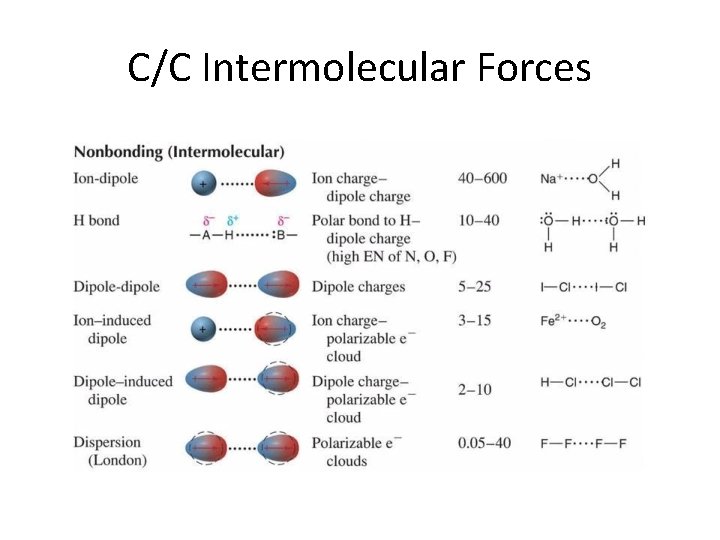
C/C Intermolecular Forces

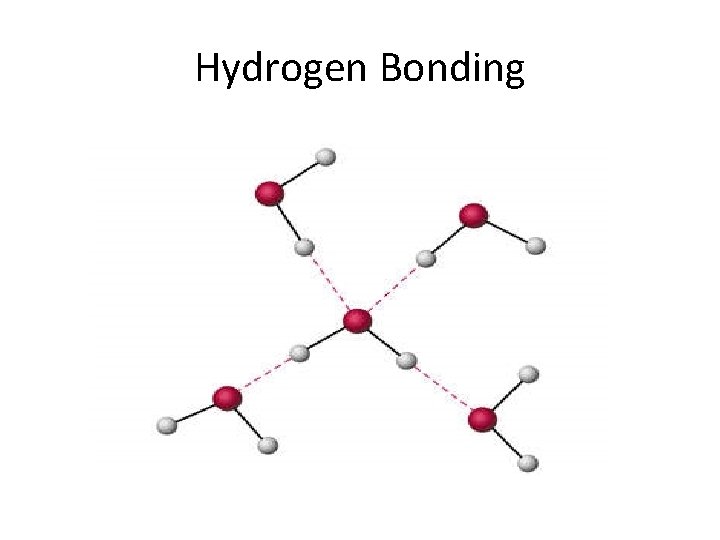
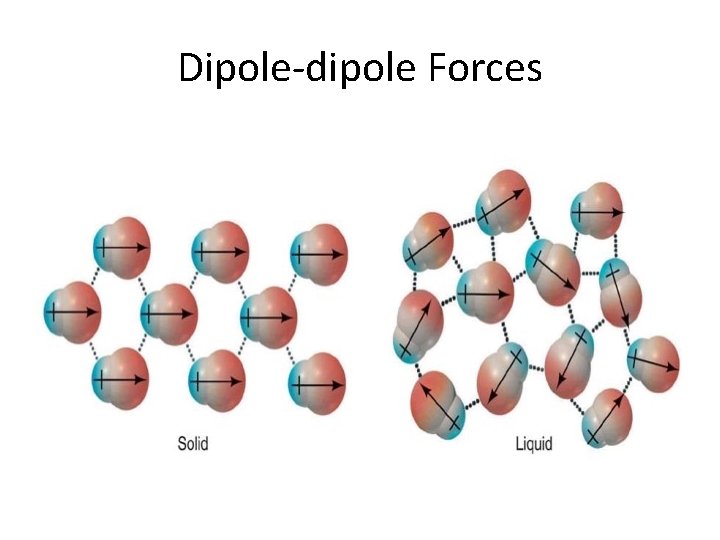
Properties of Liquids • • LEAST common state of matter in universe Fluids (as are gases) Lower kinetic energy than gases Interactive forces keep molecules connected – Dipole-dipole forces • Equal but opposite charges separated by short distance – London dispersion forces • Spontaneous creation of dipoles (polar & nonpolar) – Hydrogen bonding (electronegativity)

Hydrogen Bonding

Properties of Liquids, continued Density: 100 x > gases; 10% < solids Compressibility: @ 103 atm. , volume ~ 4% Diffusion: present, but slower than in gases Surface tension: high intermolecular attraction Capillary action: attraction between surfaces of liquid and a solid • Vaporization: evaporation & boiling gas • • •

Nature of Solids • Interparticle attractions stronger than others • Two types of solids – Crystalline (orderly arrangement) – Amorphous (random arrangement) • supercooled liquids: have liquid properties even if look solid • • Shape & Volume: Definite Melting Point: Definite Density & Incompressibility: High Diffusion: Low rate (10 -6 less than others)

Dipole-dipole Forces

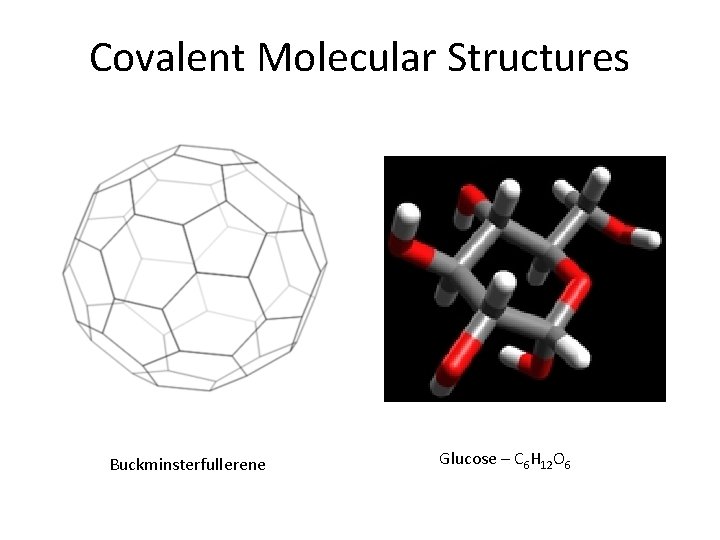
Covalent Molecular Structures Buckminsterfullerene Glucose – C 6 H 12 O 6

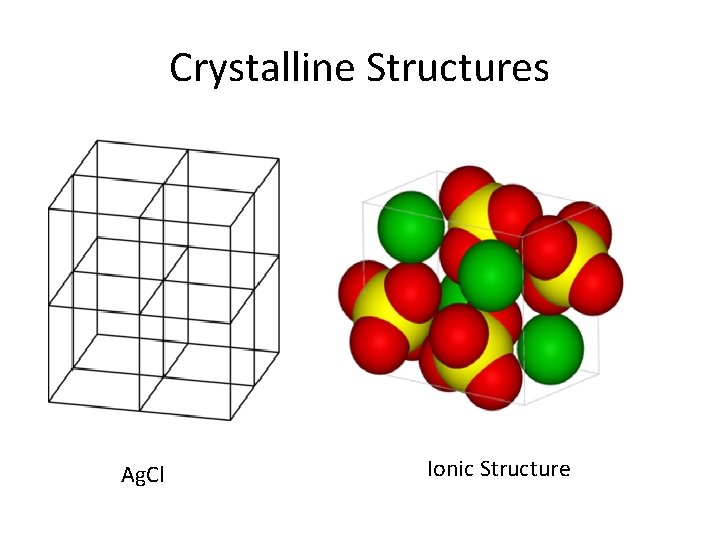
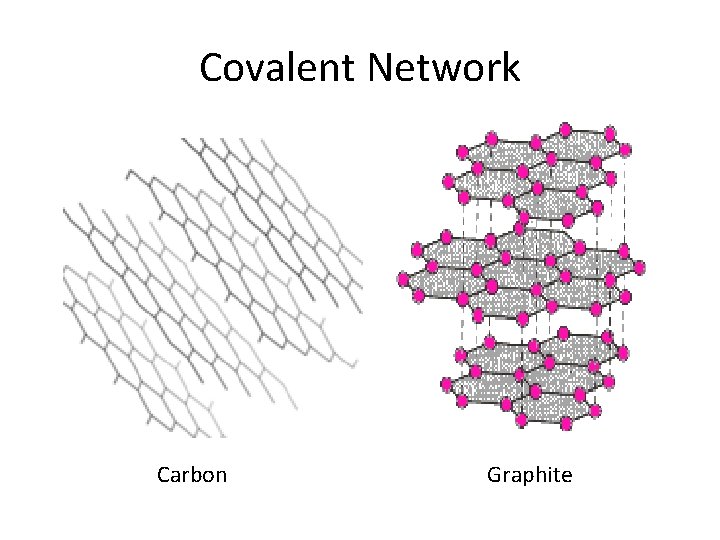
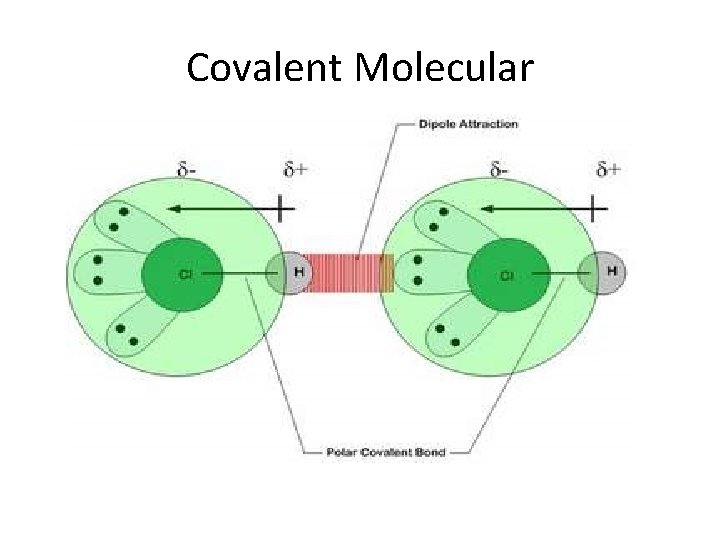
Crystalline Solids • Ionic • Alkali & alkaline earth with halogens & Group 16 • Hard, brittle, high melting points, good insulators • Covalent network • Cx (diamonds), (Si. O 2)x quartz, (Si. C)x • Very hard and brittle, high MP, semi- or nonconductors • Covalent molecular (nonpolar & polar) • H 2, CH 4, C 6 H 6: only weak London dispersion forces • H 2 O & NH 3, : stronger forces but weaker than covalent • Soft, low MP, low BP, good insulators

Crystalline Structures Ag. Cl Ionic Structure

Covalent Network Crystals Diamond Quartz

Covalent Network Carbon Graphite

Covalent Molecular