Valence Bond Theory and Molecular Orbital Theory Shapes
Valence Bond Theory and Molecular Orbital Theory

Shapes of Atomic Orbitals for Electrons �Four different kinds of orbitals for electrons denoted s, p, d, and f �s and p orbitals most important in organic and biological chemistry �s orbitals: spherical, nucleus at center �p orbitals: dumbbell-shaped, nucleus at middle �d orbitals: elongated dumbbell-shaped, nucleus at center 2

The Nature of Chemical Bonds: �Covalent bond forms when two atoms approach each other closely so that a singly occupied orbital on one atom overlaps a singly occupied orbital on the other atom �Two models to describe covalent bonding. Valence bond theory, Molecular orbital theory

Central Themes of Valence Bond Theory Basic Principle of Valence Bond Theory: a covalent bond forms when the orbitals from two atoms overlap and a pair of electrons occupies the region between the nuclei. 1) Opposing spins of the electron pair. The region of space formed by the overlapping orbitals has a maximum capacity of two electrons that must have opposite spins.

2) Maximum overlap of bonding orbitals. The bond strength depends on the attraction of nuclei for the shared electrons, so the greater the orbital overlap, the stronger the bond.

Central Themes of Valence Bond Theory 3) Hybridization of atomic orbitals. bonding in simple diatomic molecules: Example 1: HF (direct overlap of the s and p orbitals of isolated ground state atoms). Example 2: CH 4 (4 hydrogen atoms are bonded to a central carbon atom)- hybridization happens to obtain the correct bond angles. Pauling proposed that the valence atomic orbitals in the molecule are different from those in the isolated atoms. We call this Hybridization!

Hybrization of Orbitals
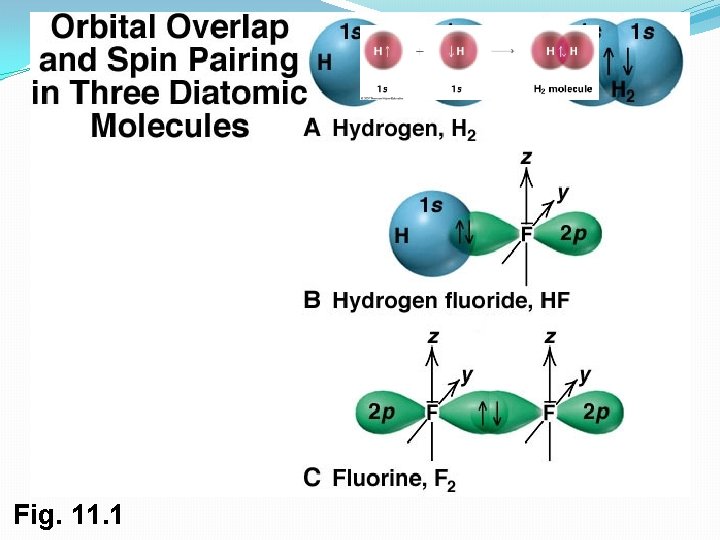
Fig. 11. 1

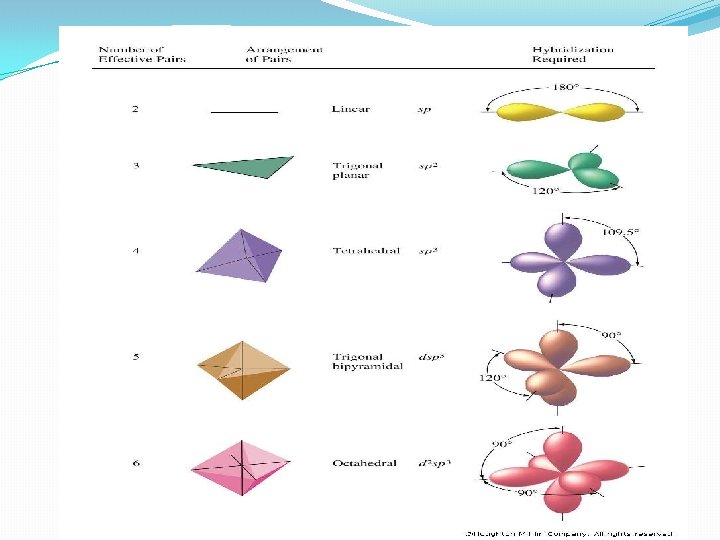
What Is A Hybrid Orbital? � are a type of atomic orbital that results when two or more atomic orbitals of an isolated atom mix (the number of hybrid orbitals on a covalently bonded atom is equal to the number of atomic orbitals used to form the hybrid orbitals) � are used to describe the orbitals in covalently bonded atoms (hybrid orbitals do not exist in isolated atoms), � have shapes and orientations that are very different from those of atomic orbitals in isolated atoms � in a set are equivalent, and form identical bonds (when the bonds are to a set of identical atoms), and � are usually involved in sigma bonds in polyatomic molecules; pi bonds usually involve the overlap of unhybridized orbitals.

In Hybridization there is ‘mixing’ or ‘blending’ of atomic orbitals to accommodate the spatial requirements in a molecule. Hybridization occurs to minimize electron pair repulsions when atoms are brought together to form molecules.
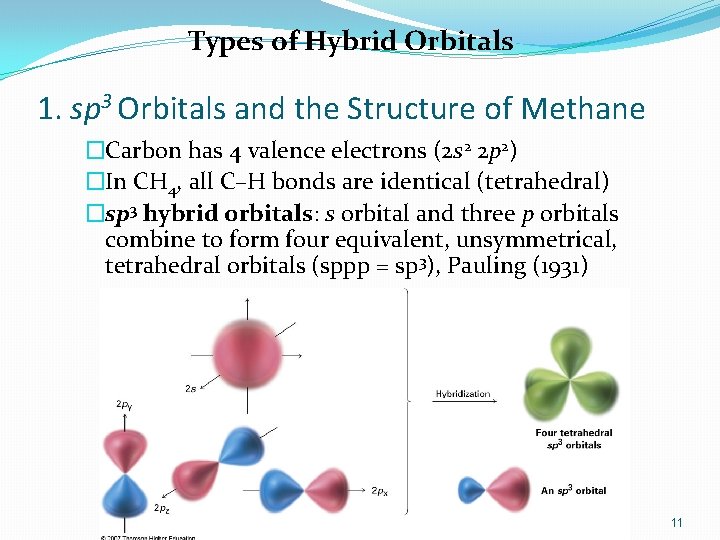
Types of Hybrid Orbitals 1. sp 3 Orbitals and the Structure of Methane �Carbon has 4 valence electrons (2 s 2 2 p 2) �In CH 4, all C–H bonds are identical (tetrahedral) �sp 3 hybrid orbitals: s orbital and three p orbitals combine to form four equivalent, unsymmetrical, tetrahedral orbitals (sppp = sp 3), Pauling (1931) 11
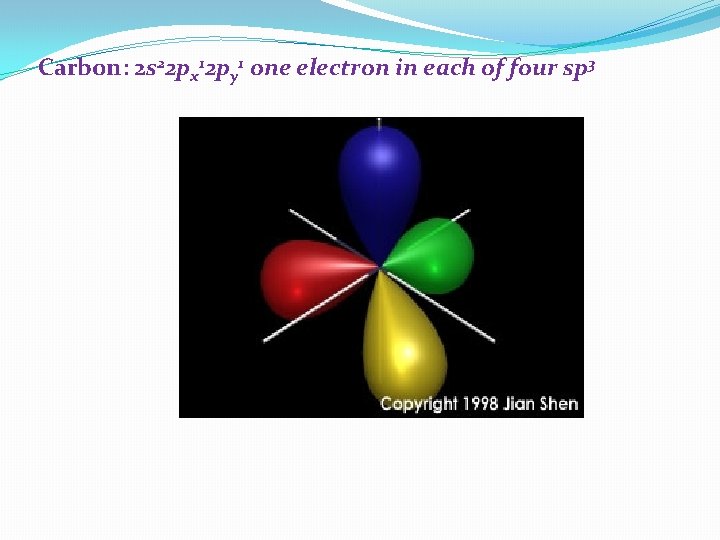
Carbon: 2 s 22 px 12 py 1 one electron in each of four sp 3
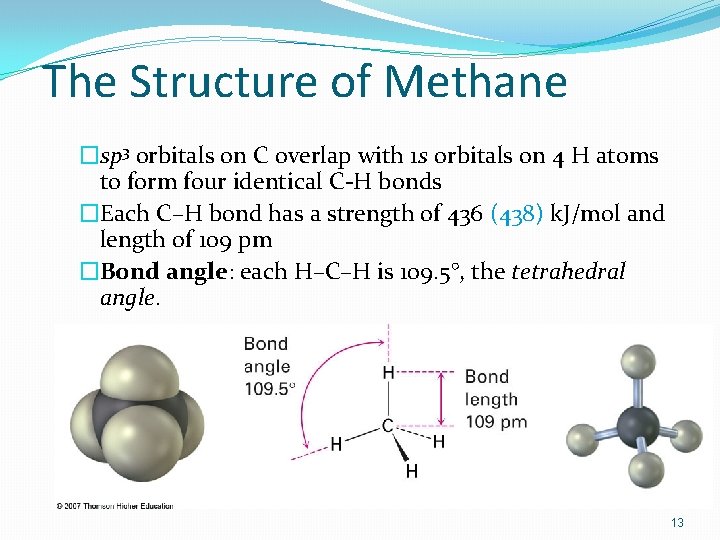
The Structure of Methane �sp 3 orbitals on C overlap with 1 s orbitals on 4 H atoms to form four identical C-H bonds �Each C–H bond has a strength of 436 (438) k. J/mol and length of 109 pm �Bond angle: each H–C–H is 109. 5°, the tetrahedral angle. 13
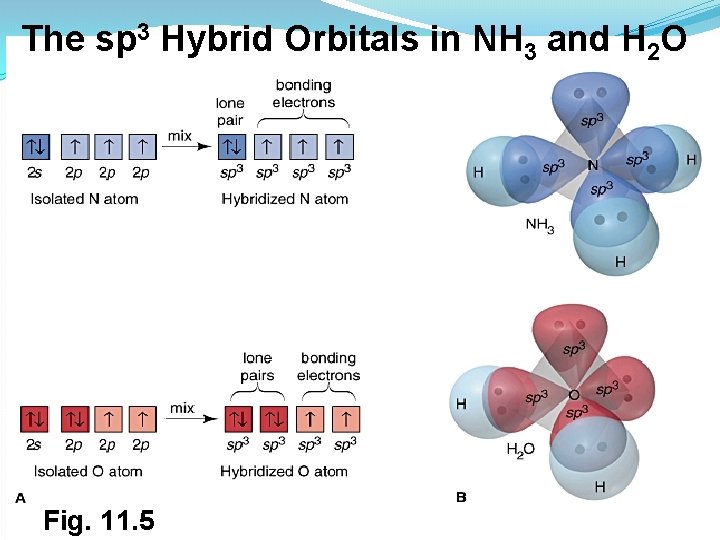
The sp 3 Hybrid Orbitals in NH 3 and H 2 O Fig. 11. 5
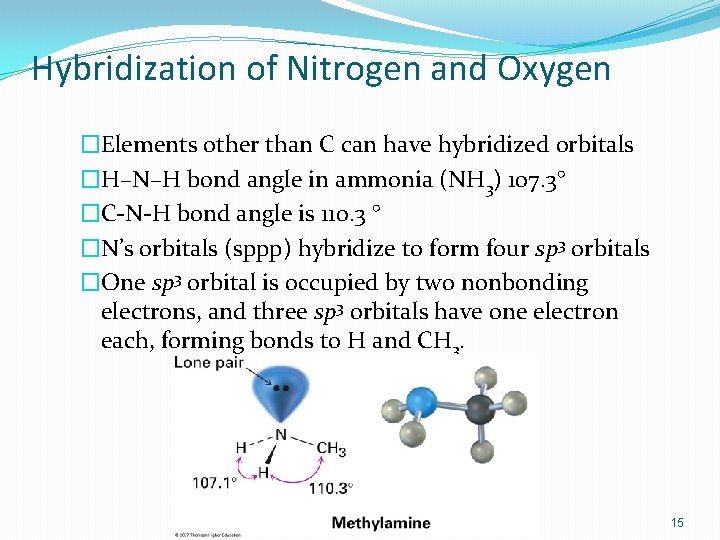
Hybridization of Nitrogen and Oxygen �Elements other than C can have hybridized orbitals �H–N–H bond angle in ammonia (NH 3) 107. 3° �C-N-H bond angle is 110. 3 ° �N’s orbitals (sppp) hybridize to form four sp 3 orbitals �One sp 3 orbital is occupied by two nonbonding electrons, and three sp 3 orbitals have one electron each, forming bonds to H and CH 3. 15
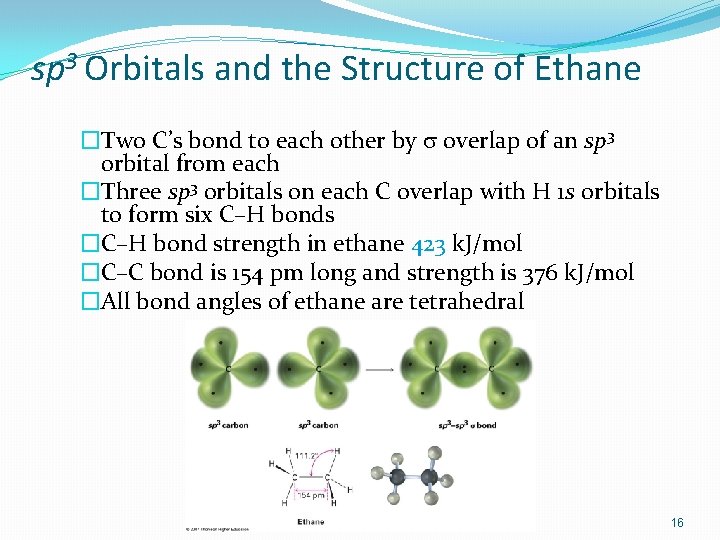
sp 3 Orbitals and the Structure of Ethane �Two C’s bond to each other by s overlap of an sp 3 orbital from each �Three sp 3 orbitals on each C overlap with H 1 s orbitals to form six C–H bonds �C–H bond strength in ethane 423 k. J/mol �C–C bond is 154 pm long and strength is 376 k. J/mol �All bond angles of ethane are tetrahedral 16

2. sp 2 Orbitals and the Structure of Ethylene �sp 2 hybrid orbitals: 2 s orbital combines with two 2 p orbitals, giving 3 orbitals (spp = sp 2). This results in a double bond. �sp 2 orbitals are in a plane with 120° angles �Remaining p orbital is perpendicular to the plane 17

Bonds From 2 sp Hybrid Orbitals �Two sp 2 -hybridized orbitals overlap to form a s bond �p orbitals overlap side-to-side to formation a pi ( ) bond �sp 2–sp 2 s bond and 2 p– 2 p bond result in sharing four electrons and formation of C-C double bond �Electrons in the s bond are centered between nuclei �Electrons in the bond occupy regions are on either side of a line between nuclei 18
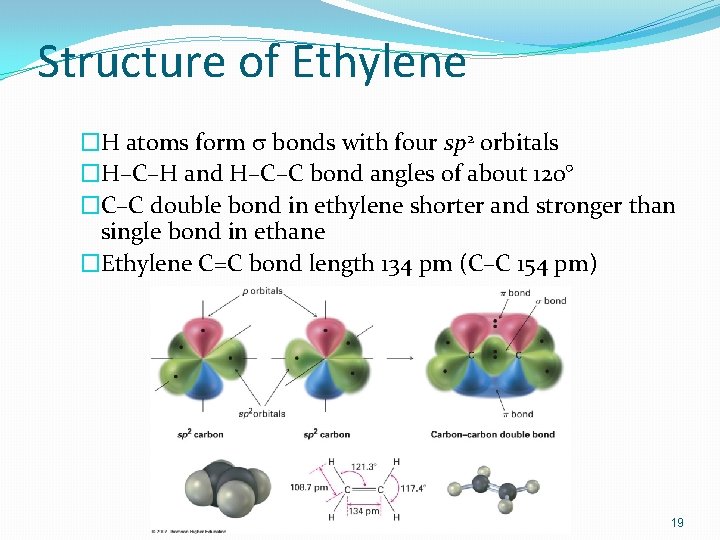
Structure of Ethylene �H atoms form s bonds with four sp 2 orbitals �H–C–H and H–C–C bond angles of about 120° �C–C double bond in ethylene shorter and stronger than single bond in ethane �Ethylene C=C bond length 134 pm (C–C 154 pm) 19
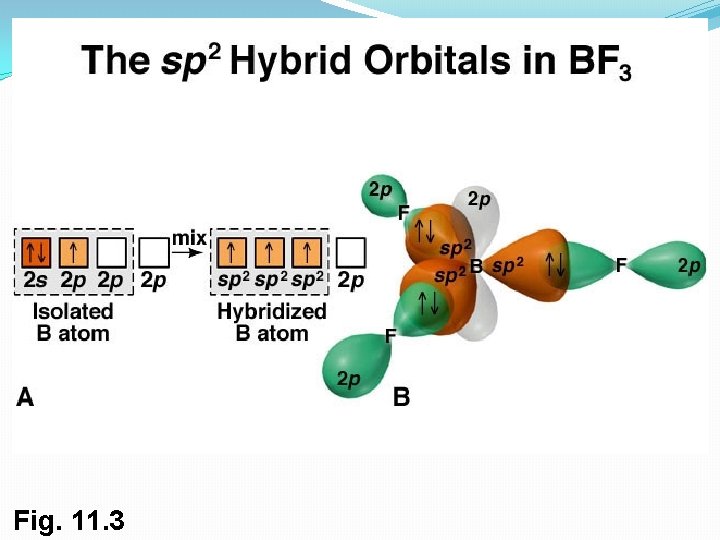
Fig. 11. 3
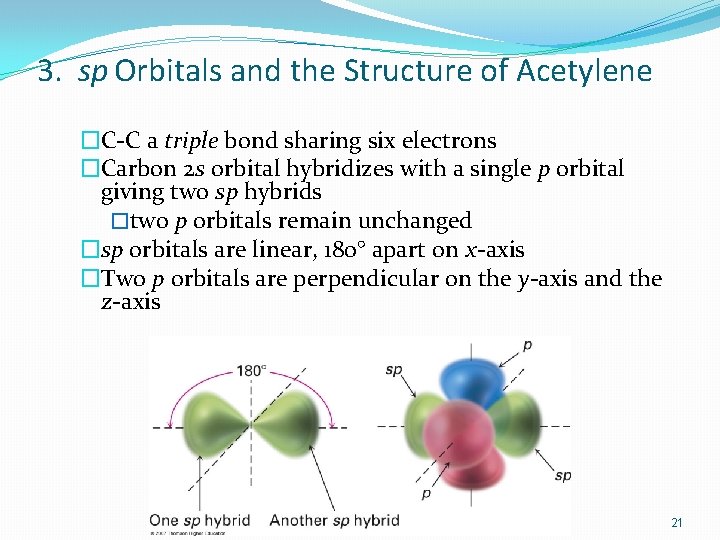
3. sp Orbitals and the Structure of Acetylene �C-C a triple bond sharing six electrons �Carbon 2 s orbital hybridizes with a single p orbital giving two sp hybrids �two p orbitals remain unchanged �sp orbitals are linear, 180° apart on x-axis �Two p orbitals are perpendicular on the y-axis and the z-axis 21
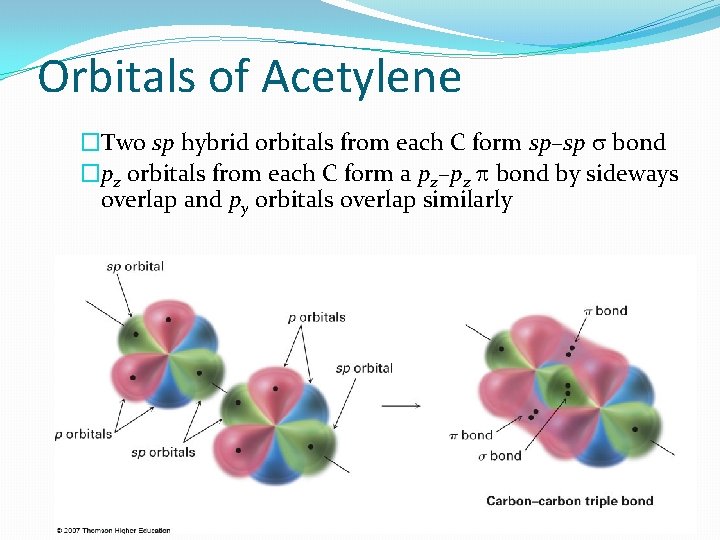
Orbitals of Acetylene �Two sp hybrid orbitals from each C form sp–sp s bond �pz orbitals from each C form a pz–pz bond by sideways overlap and py orbitals overlap similarly 22
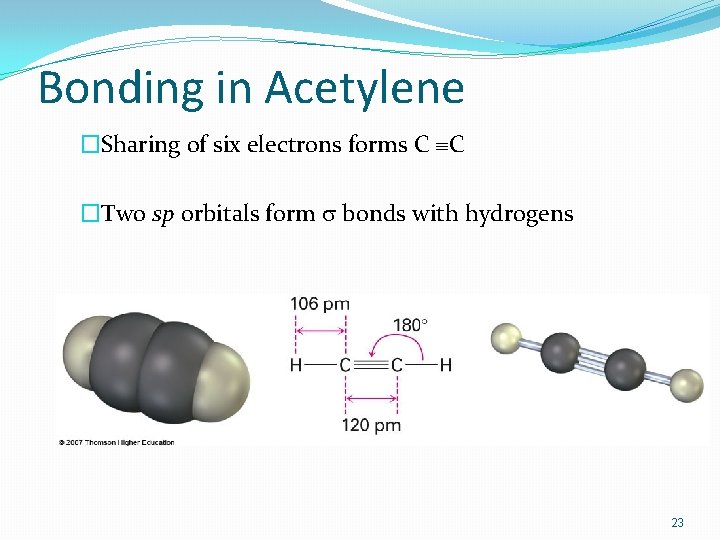
Bonding in Acetylene �Sharing of six electrons forms C ºC �Two sp orbitals form s bonds with hydrogens 23

4. The sp 3 d Hybrid Orbitals in PCl 5 Fig. 11. 6
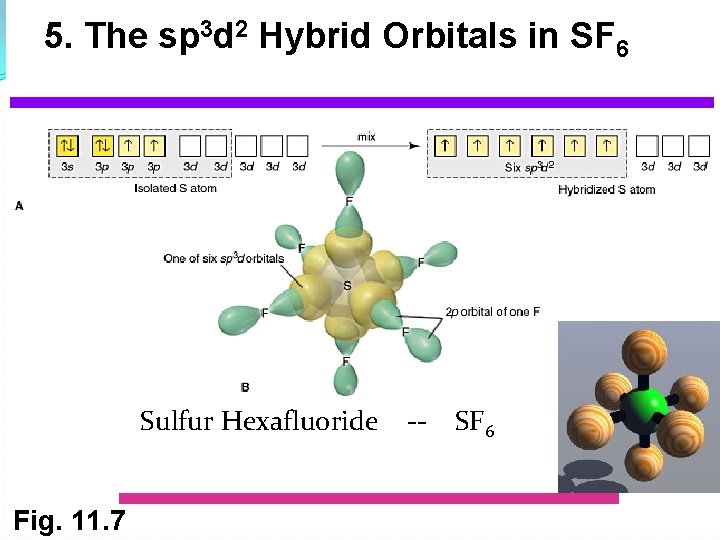
5. The sp 3 d 2 Hybrid Orbitals in SF 6 Sulfur Hexafluoride Fig. 11. 7 -- SF 6

Molecular Orbital Theory �A molecular orbital (MO): where electrons are most likely to be found (specific energy and general shape) in a molecule �Additive combination (bonding) MO is lower in energy �Subtractive combination (antibonding) MO is higher energy 27

Molecular Orbitals in Ethylene �The bonding MO is from combining p orbital lobes with the same algebraic sign �The antibonding MO is from combining lobes with opposite signs �Only bonding MO is occupied 28

Valence Bond Theory vs. MO Theory �VB Theory begins with two steps: �hybridization (where necessary to get atomic orbitals that “point at each other”) �combination of hybrid orbitals to make bonds with electron density localized between the two bonding atoms �Key differences between MO and VB theory: �MO theory has electrons distributed over molecule �VB theory localizes an electron pair between two atoms �MO theory combines AOs on DIFFERENT atoms to make MOs (LCAO) �VB theory combines AOs on the SAME atom to make hybridized atomic orbitals (hybridization) �In MO theory, the symmetry (or antisymmetry) must be retained in each orbital. �In VB theory, all orbitals must be looked at once to see retention of the molecule’s symmetry. 29

Sigma and Pi Bonds

�Difference between sigma and pi bond Sigma bond(σ) �Formed by head to head overlap of AO’s �The 2 AO’s that overlap are symmetrical about the x axis joining the 2 nuclei. �Has free rotation �Lower energy �Only one bond can exist between two atoms( a single covalent bond) Pi bond(π) �Formed by side-to-side overlap of AO’s �No free rotation �Has higher energy �One or two bonds can exist between two atoms �Have nodal plane on the molecular axis and no longer symmetrical about the molecular axis.
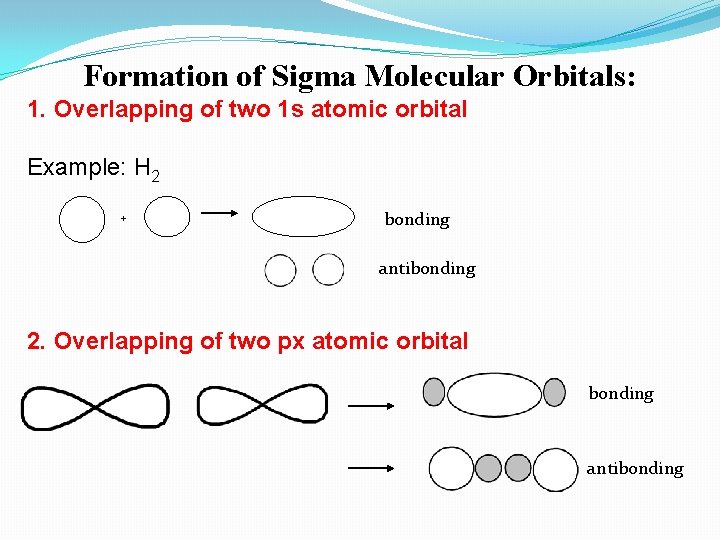
Formation of Sigma Molecular Orbitals: 1. Overlapping of two 1 s atomic orbital Example: H 2 + bonding antibonding 2. Overlapping of two px atomic orbital bonding antibonding
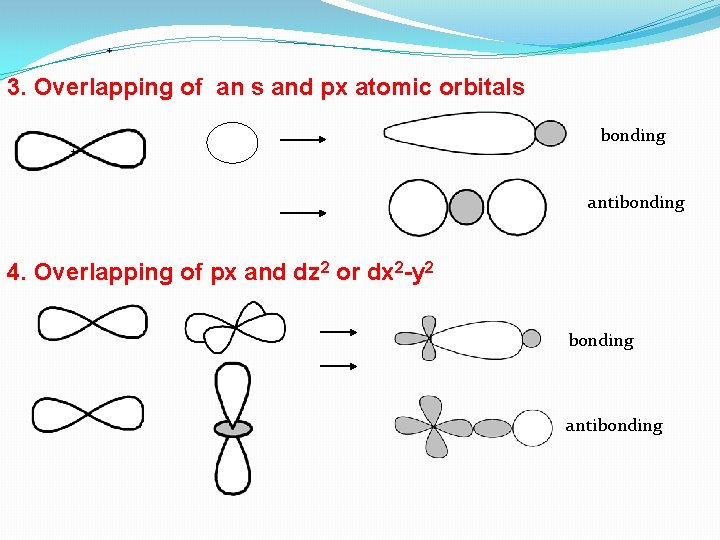
+ 3. Overlapping of an s and px atomic orbitals + bonding antibonding 4. Overlapping of px and dz 2 or dx 2 -y 2 bonding antibonding
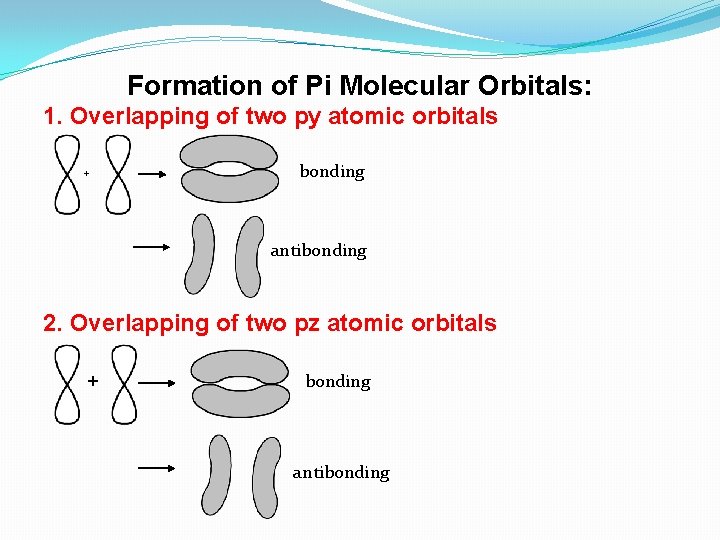
Formation of Pi Molecular Orbitals: 1. Overlapping of two py atomic orbitals + bonding antibonding 2. Overlapping of two pz atomic orbitals + bonding antibonding
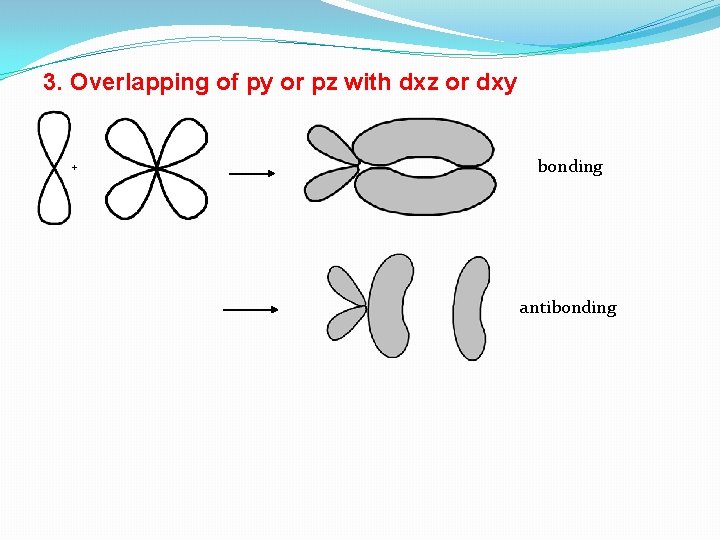
3. Overlapping of py or pz with dxz or dxy + bonding antibonding
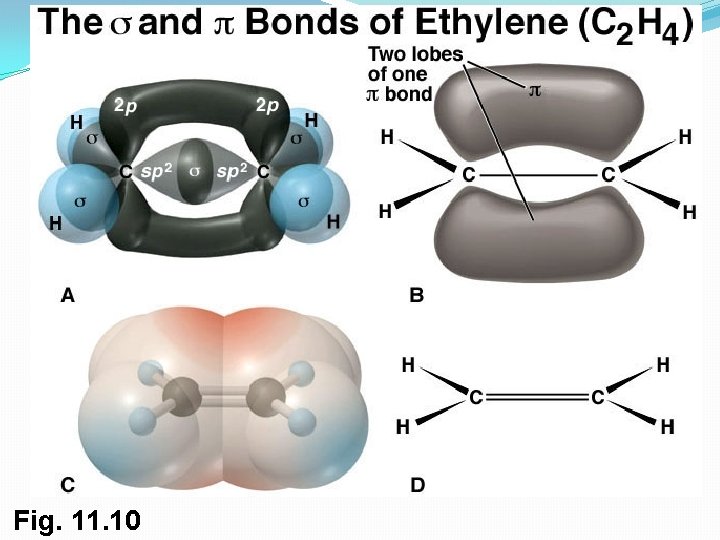
Fig. 11. 10
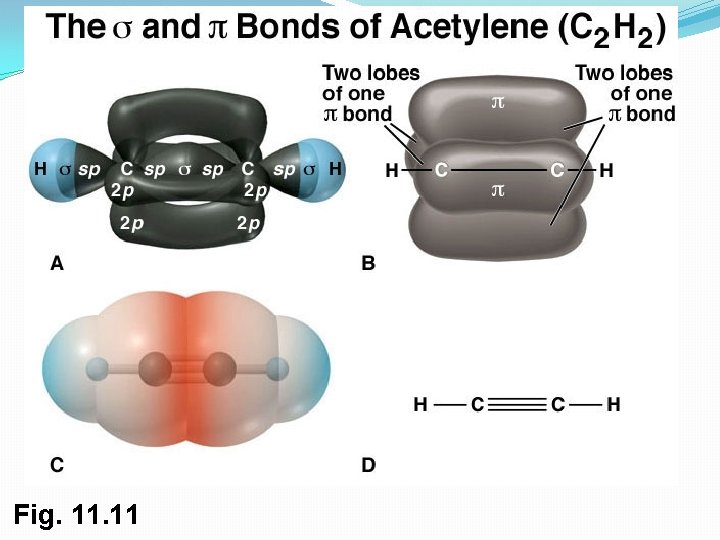
Fig. 11

Comparison between sigma and pi electrons in ethylene and acetylene 1. Pi electron are made exposed to the environment than the sigma electrons. 2. Pi electrons are more reactive than sigma electrons. 3. The looseness of the pi electrons in C 2 H 2 is less than in C 2 H 4. This is the result of the greater s character in sp hybrids as compared with the s character in sp 2 hydrid. 4. Pi electrons in C 2 H 2 are attracted more strongly towards the nucleus than the pi electrons in C 2 H 4. 5. The pi bonds in C 2 H 2 are more susceptible to attack by other chemical entities than in C 2 H 4.
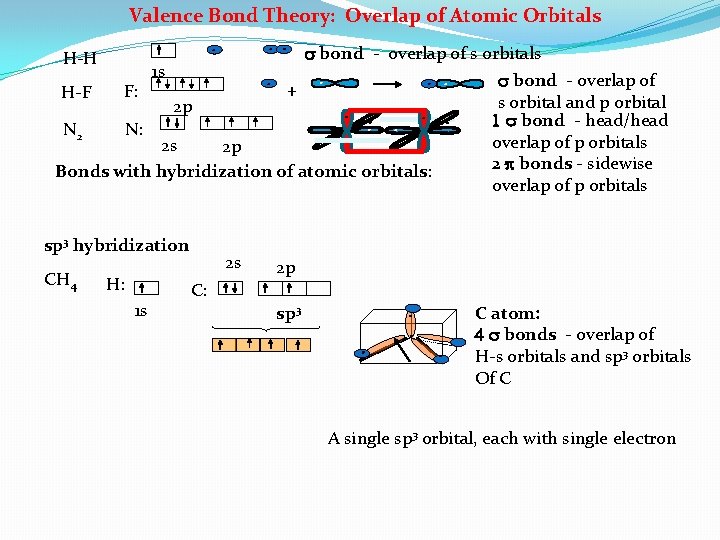
Valence Bond Theory: Overlap of Atomic Orbitals s bond - overlap of s orbitals 1 s s bond - overlap of + F: H-F s orbital and p orbital 2 p 1 s bond - head/head N 2 N: overlap of p orbitals 2 s 2 p 2 bonds - sidewise Bonds with hybridization of atomic orbitals: overlap of p orbitals H-H sp 3 hybridization CH 4 H: 1 s 2 s 2 p C: sp 3 C atom: 4 s bonds - overlap of H-s orbitals and sp 3 orbitals Of C A single sp 3 orbital, each with single electron

sp 2 hybridization H 2 CO C: 2 s H 2 p H p sp 2 C O C atom 3 s bond; 1 bond A single sp 2 orbital, each with single electron sp hybridization CO 2 C: 2 s 2 p sp O p C O C atom: 2 s bonds 2 bond A single sp orbital, each with single electron
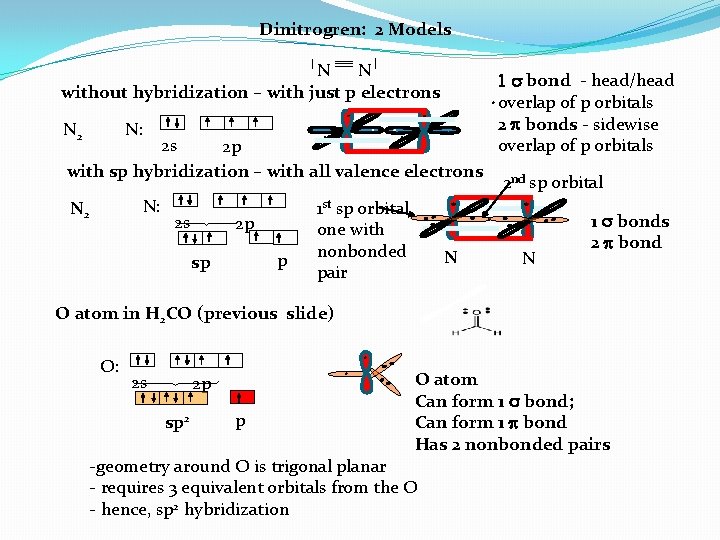
Dinitrogren: 2 Models N N without hybridization – with just p electrons N 2 N: 2 s 2 p with sp hybridization – with all valence electrons N: N 2 2 s 2 p p sp 1 st sp orbital one with nonbonded pair N 1 s bond - head/head overlap of p orbitals 2 bonds - sidewise overlap of p orbitals 2 nd sp orbital N 1 s bonds 2 bond O atom in H 2 CO (previous slide) O: 2 s 2 p sp 2 p O atom Can form 1 s bond; Can form 1 bond Has 2 nonbonded pairs -geometry around O is trigonal planar - requires 3 equivalent orbitals from the O - hence, sp 2 hybridization
- Slides: 41