Chapter 13 Molecular Shapes VSEPR Theory Molecular Shape










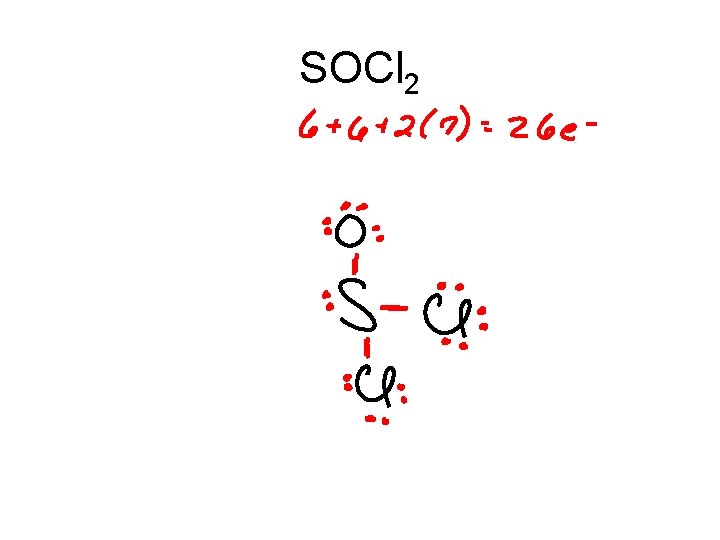
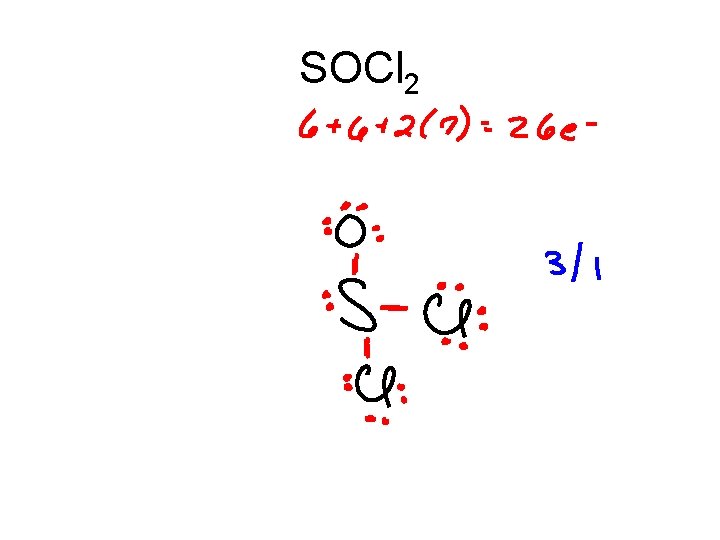

- Slides: 42

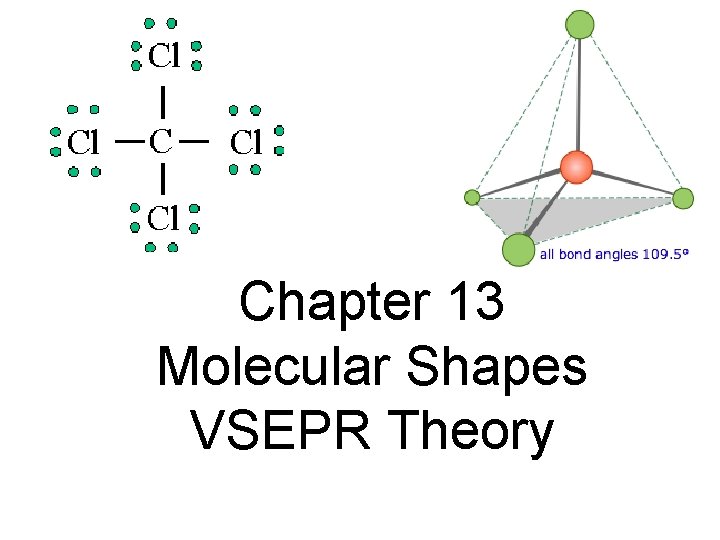
Chapter 13 Molecular Shapes VSEPR Theory

Molecular Shape • Molecular shape or molecular geometry is the three-dimensional arrangement of the atoms in a molecule.

Molecular Shape • Molecular shape determines several properties of a substance including: – reactivity – polarity – phase of matter – color – magnetism – biological activity

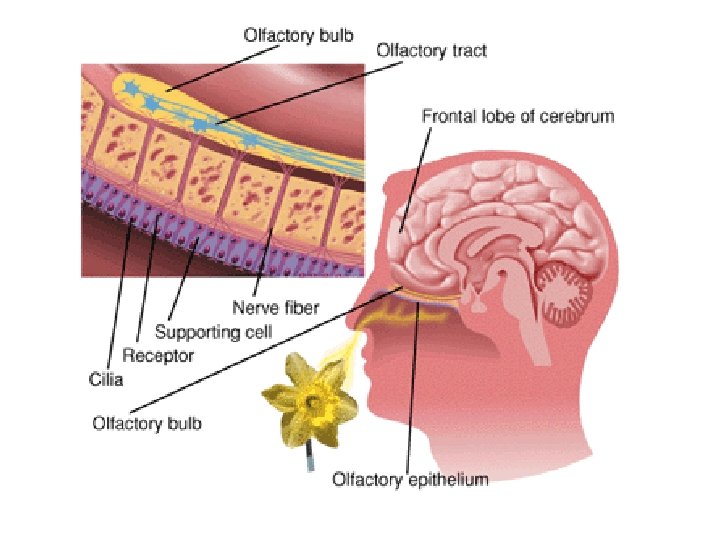
Olfaction – sense of smell • Lock-and-Key Theory: humans can smell various odors because each threedimensional odor molecule fits into only one type of receptor.


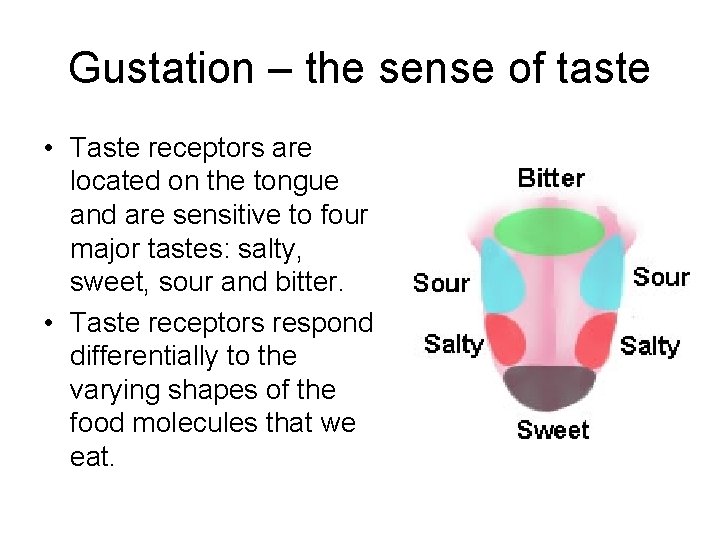
Gustation – the sense of taste • Taste receptors are located on the tongue and are sensitive to four major tastes: salty, sweet, sour and bitter. • Taste receptors respond differentially to the varying shapes of the food molecules that we eat.

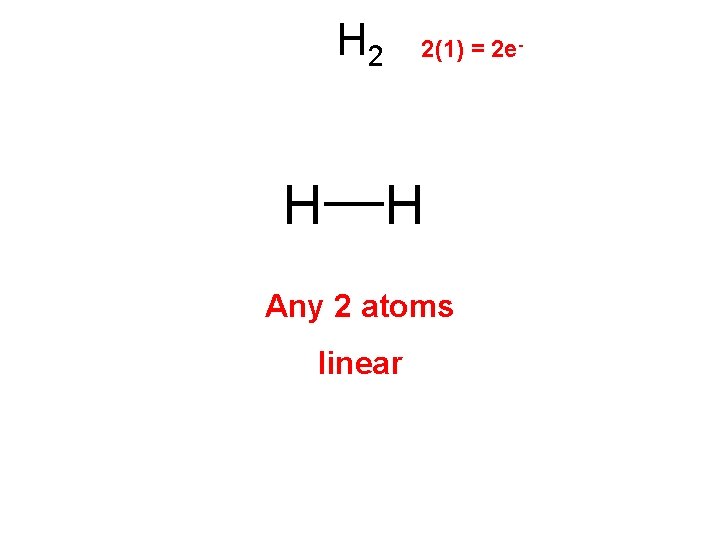
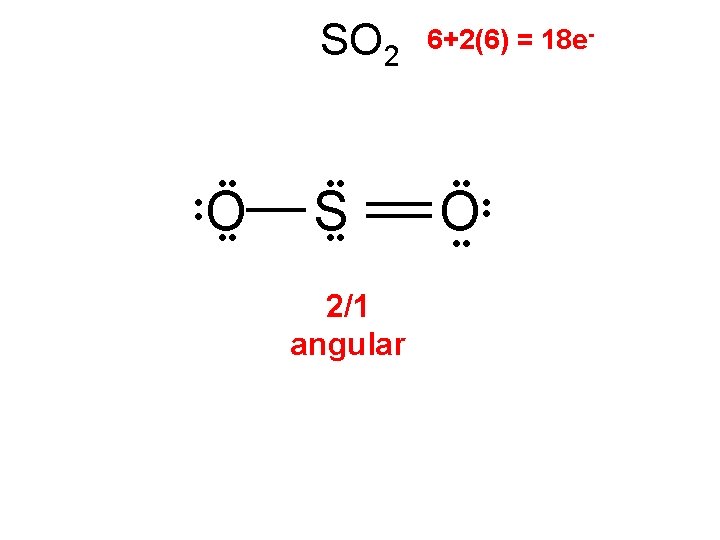
H 2 H 2(1) = 2 e- H Any 2 atoms linear

What about molecules consisting of more than two atoms For this we must use the VSEPR Theory

VSEPR Theory • Valence Shell Electron Pair Repulsion Theory • Predicts the molecular shape of a bonded molecule containing a central atom(s). • Electrons around the central atom arrange themselves as far apart from each other as possible • So only electrons (lone pairs or bonds) connected to the central atom are important.

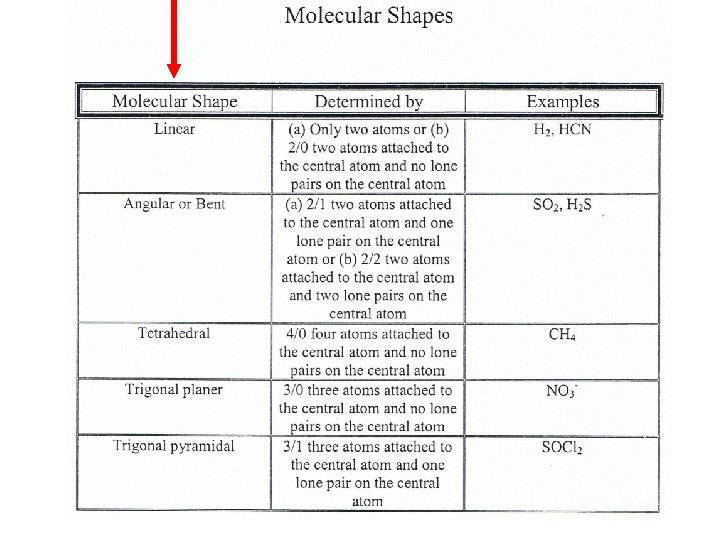
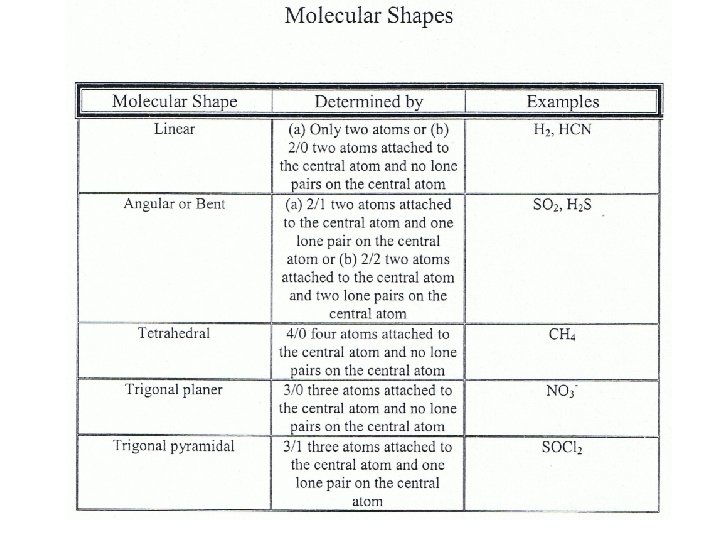
Five Molecular Shapes See the table in your notes


Steps in Determining Molecular Shape 1. Draw the Lewis Structure for the Molecule. 2. Count the number of atoms attached to the central atom. 3. Count the number of lone pairs attached to the central atom. 4. Use your counts on steps 2 and 3 to determine the shape of the molecule.


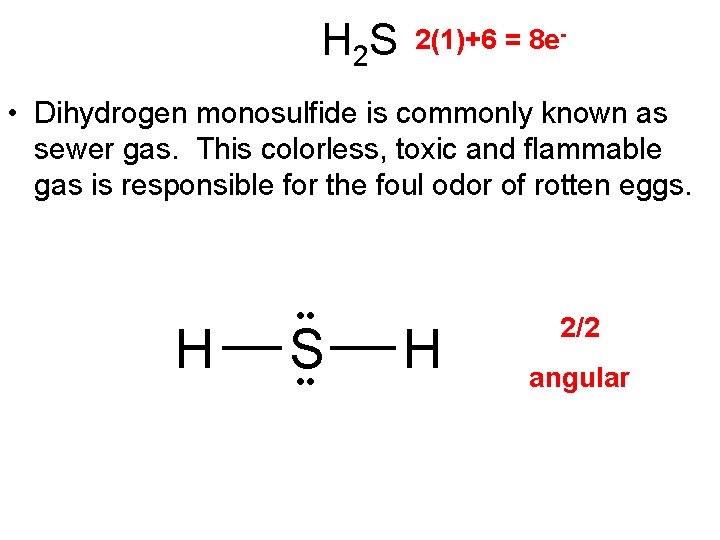
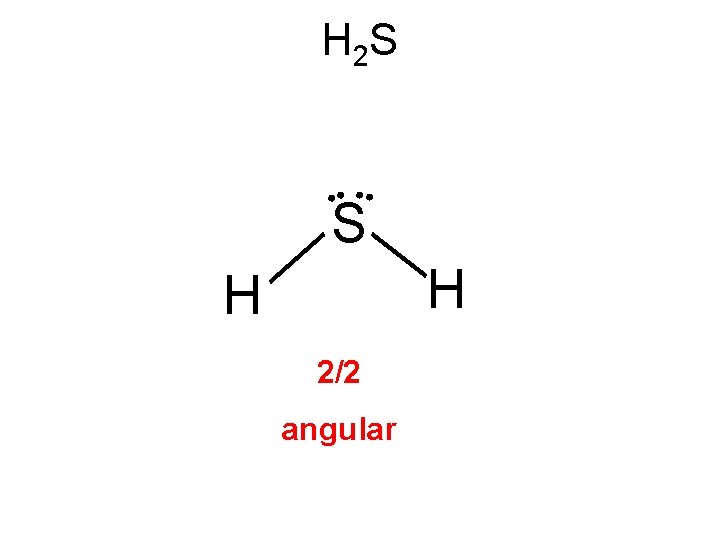
H 2 S 2(1)+6 = 8 e- • Dihydrogen monosulfide is commonly known as sewer gas. This colorless, toxic and flammable gas is responsible for the foul odor of rotten eggs. H • • S • • H 2/2 angular

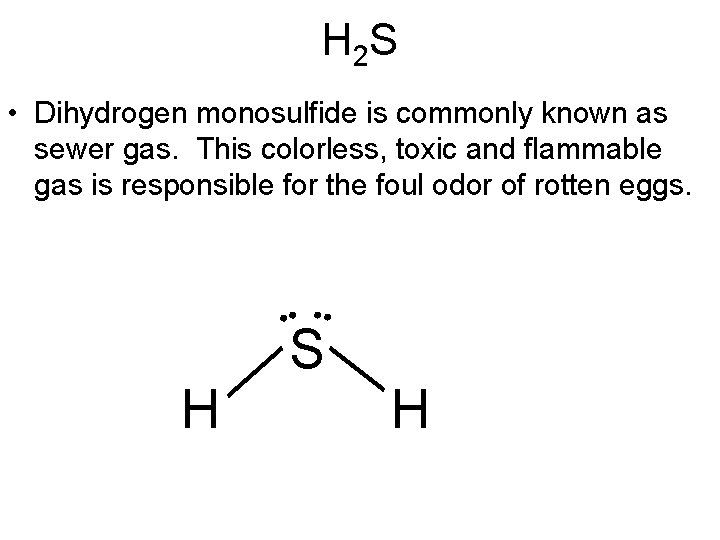
H 2 S • Dihydrogen monosulfide is commonly known as sewer gas. This colorless, toxic and flammable gas is responsible for the foul odor of rotten eggs. • • H S H

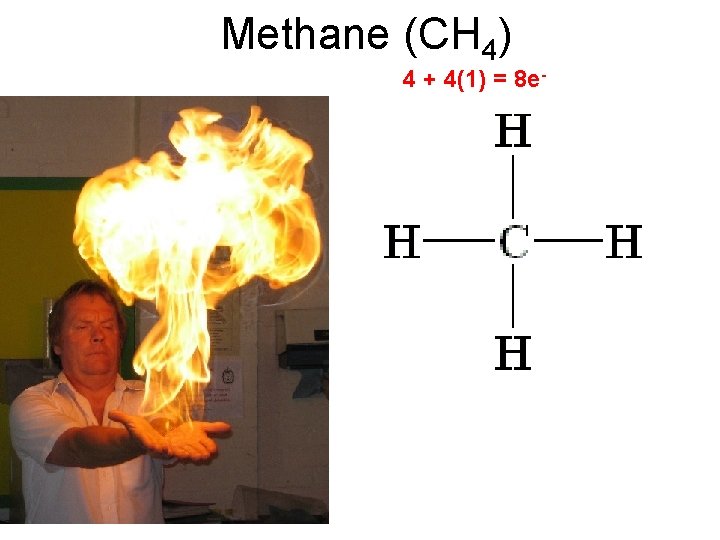
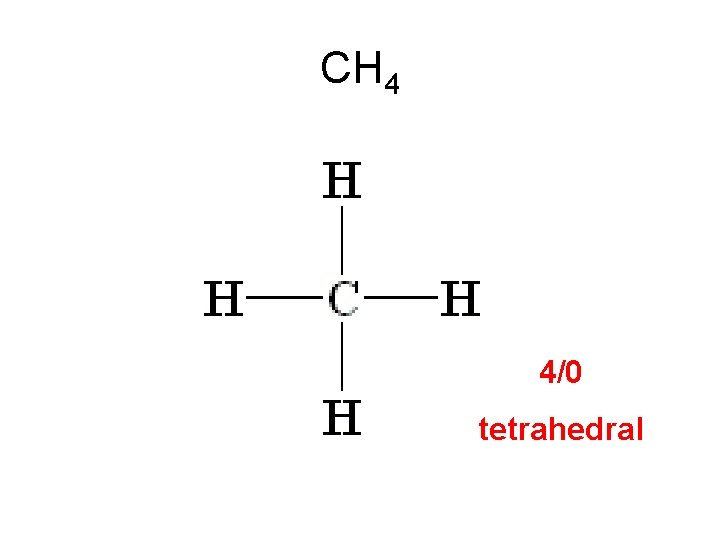
Methane (CH 4) 4 + 4(1) = 8 e-

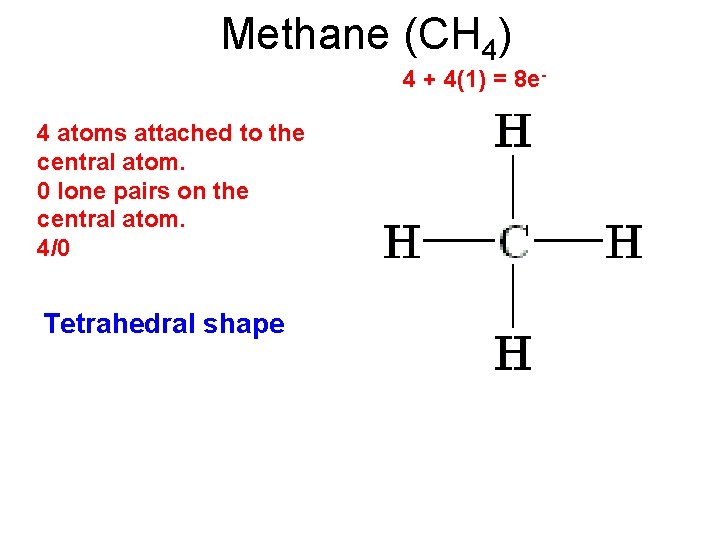
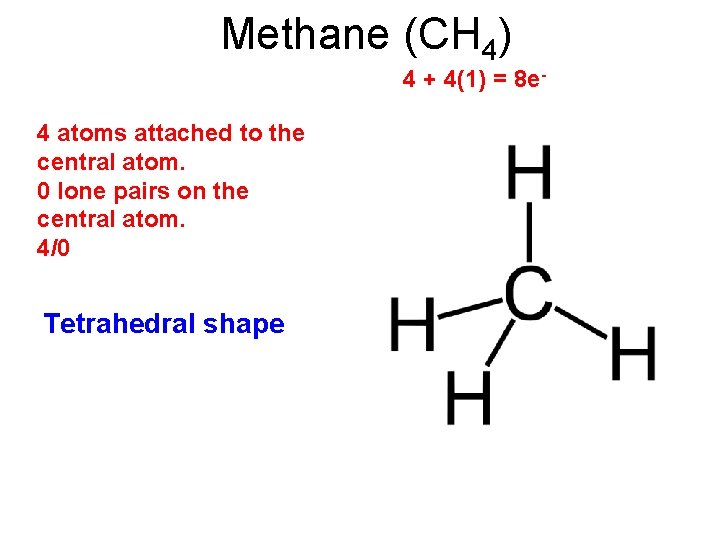
Methane (CH 4) 4 + 4(1) = 8 e- 4 atoms attached to the central atom. 0 lone pairs on the central atom. 4/0 Tetrahedral shape

Methane (CH 4) 4 + 4(1) = 8 e- 4 atoms attached to the central atom. 0 lone pairs on the central atom. 4/0 Tetrahedral shape






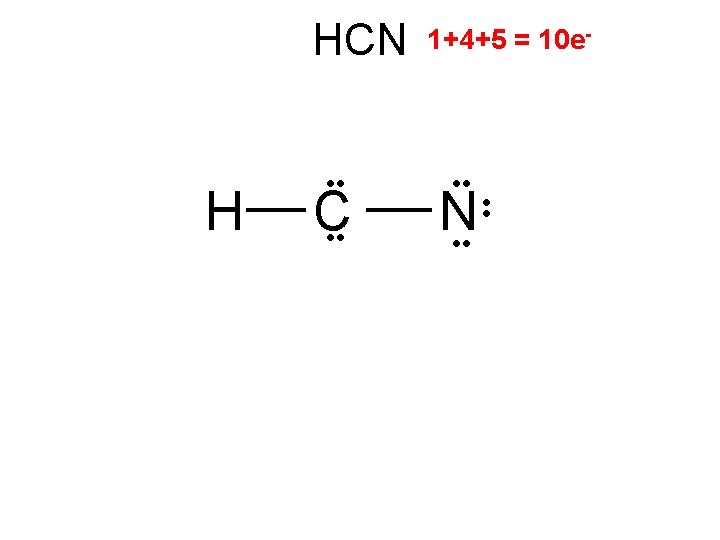
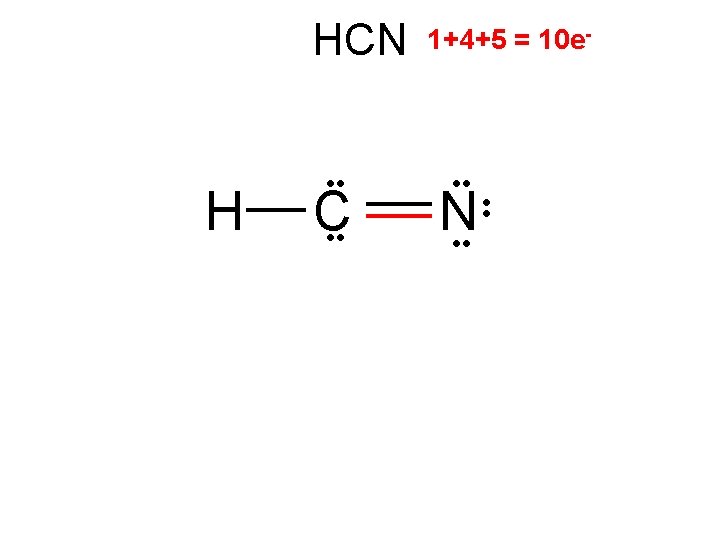
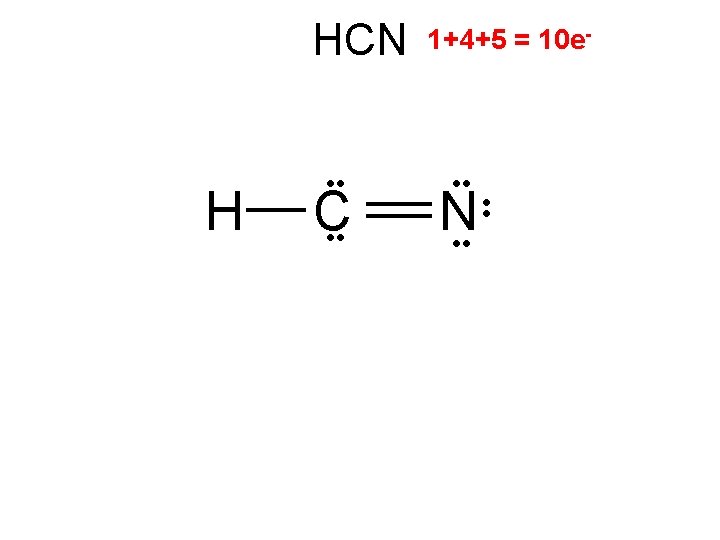
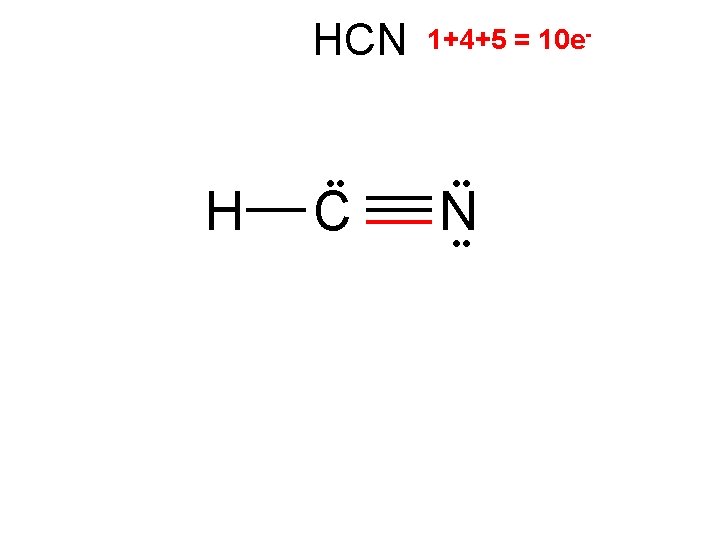
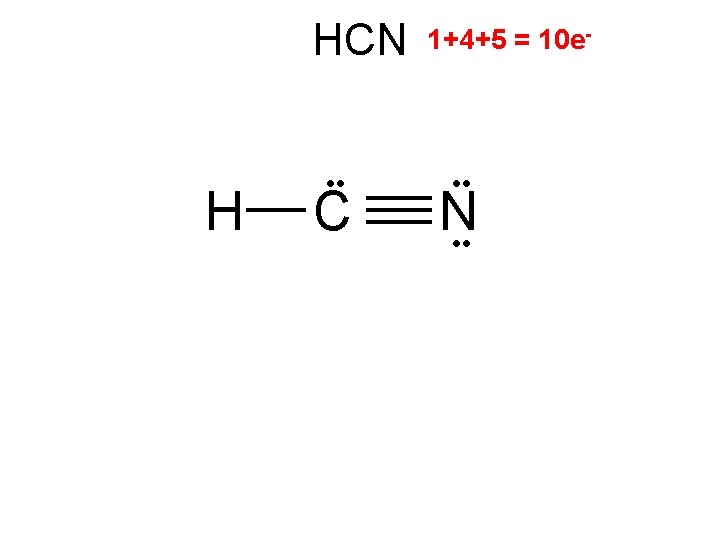
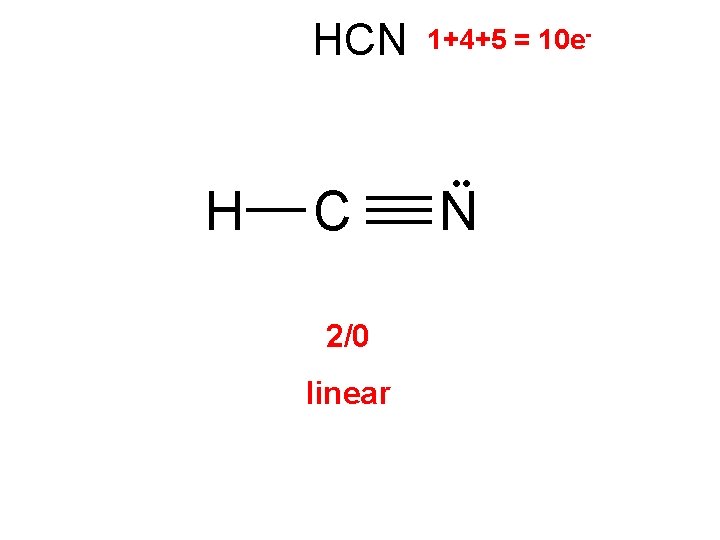
HCN H C 2/0 linear 1+4+5 = 10 e- • • N

H 2 S • • S H H 2/2 angular





CH 4 4/0 tetrahedral





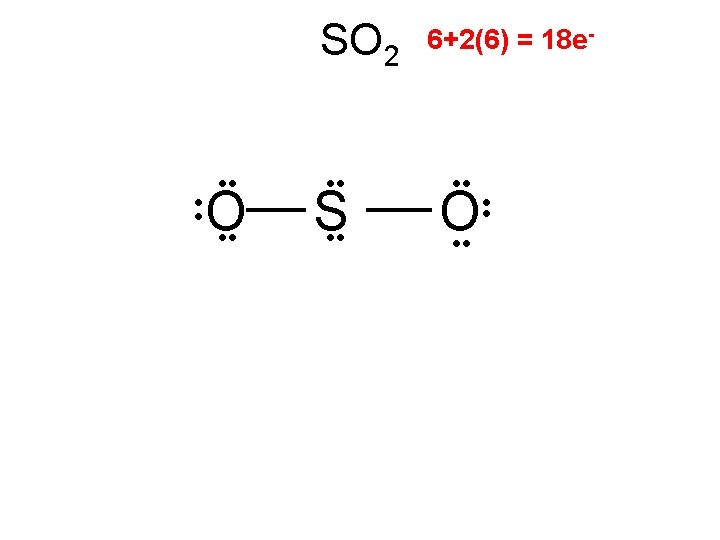
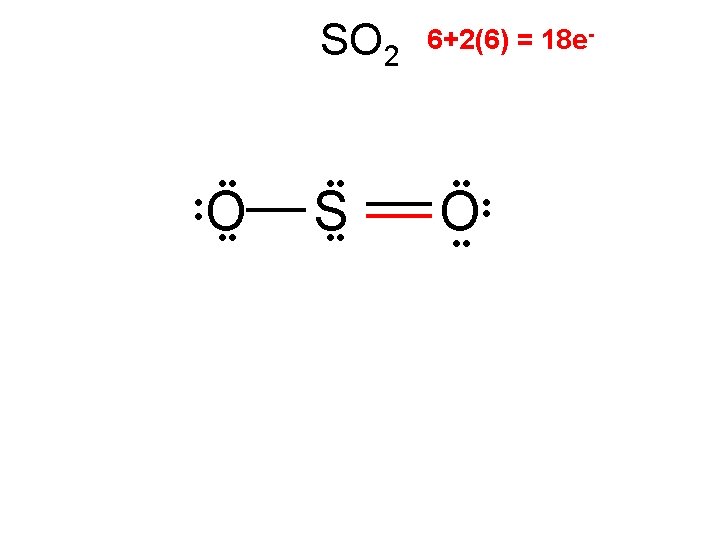
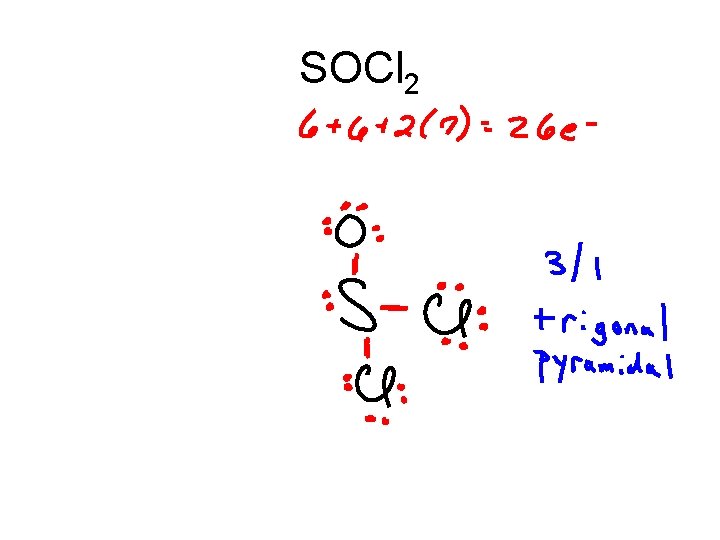
SOCl 2

SOCl 2 .
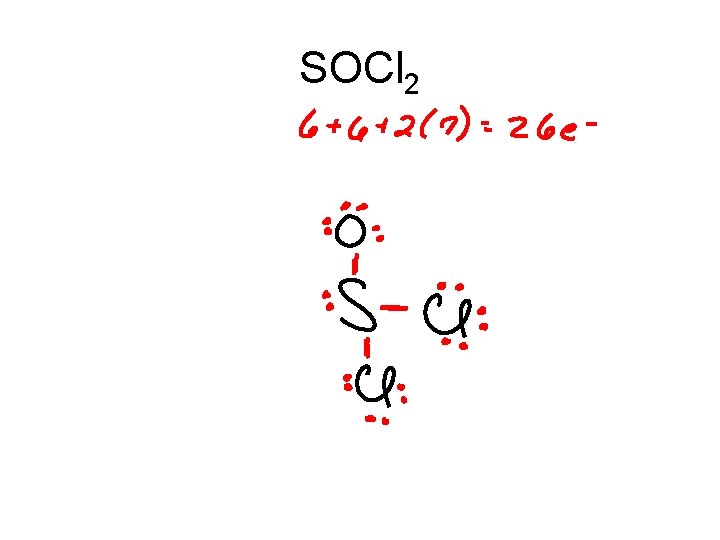
SOCl 2 .
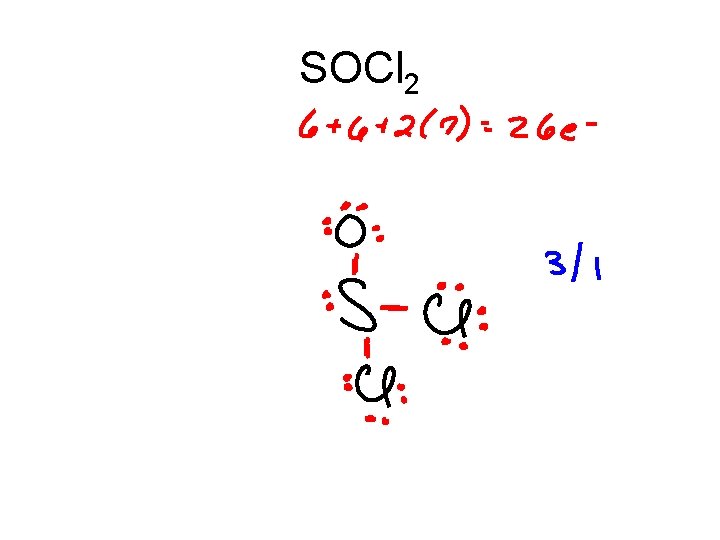
SOCl 2

SOCl 2

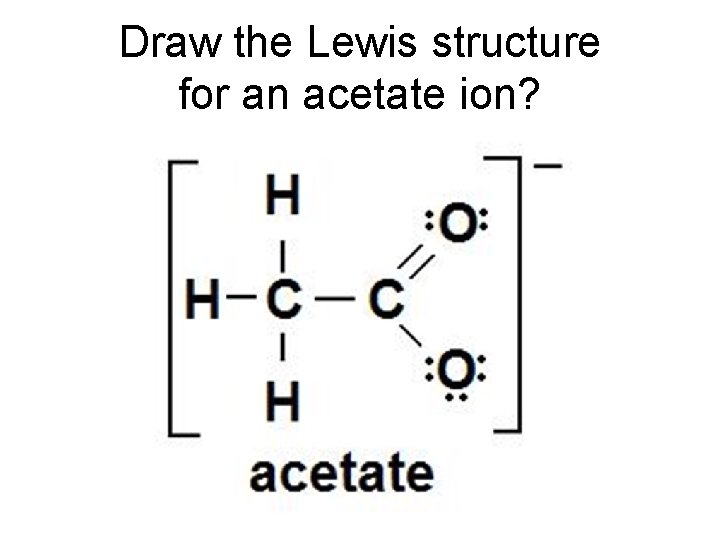
Draw the Lewis structure for an acetate ion? You learned acetate as C 2 H 3 O 2 -. However writing it as CH 3 CO 2 - will give you a better understanding of its Lewis structure.

Draw the Lewis structure for an acetate ion?

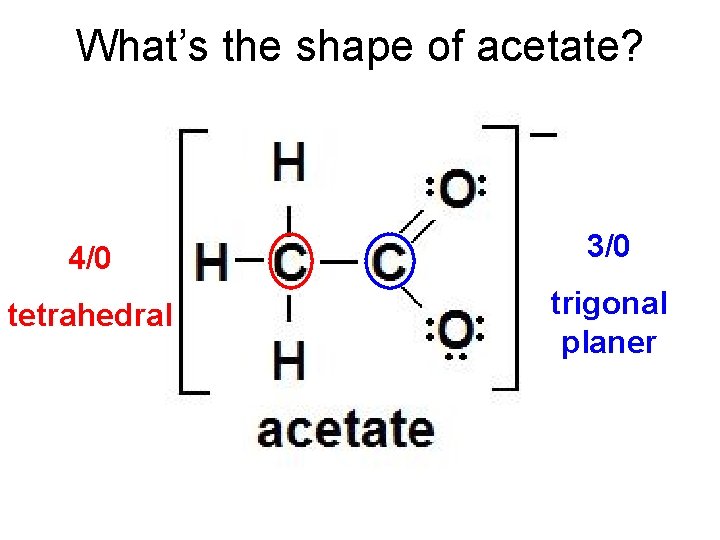
What’s the shape of acetate? 4/0 3/0 tetrahedral trigonal planer