Molecular Shapes Lewis Structure 2 D Ballandstick Model
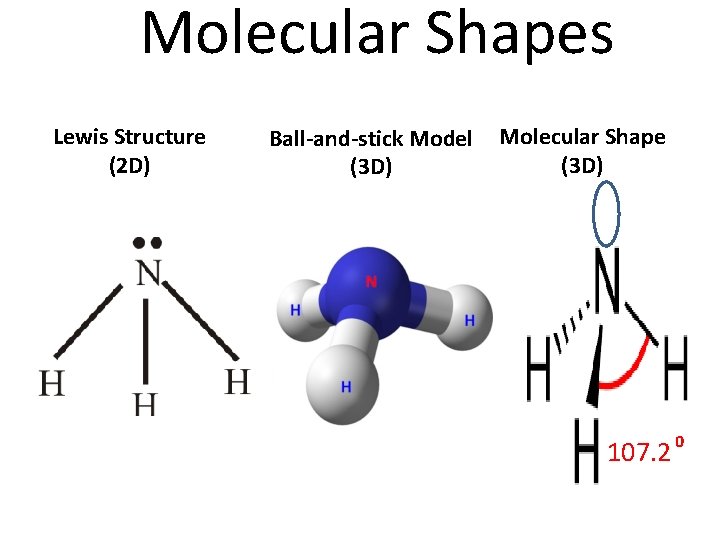
Molecular Shapes Lewis Structure (2 D) Ball-and-stick Model (3 D) Molecular Shape (3 D) 107. 2 0

VSEPR Theory Valence Shell Electron Pair Repulsion Theory Main ideas – Electron pairs have repulsions of each other, but they have to be positioned as requested Need to position these electron pairs as far as possible As to minimize repulsions between electron pairs Determines the shape of 3 D molecule

Left Over e- (Lone Pair) @ Central Atom Lone Pair: Valence e- pair that is not involved in bonding 1 Lone Pair = 2 Left Over e. If there are lone pair around the central atom? REMEMBER: Lone Pair (LP) 1) also take up space!!! 2) repel each other 3) repel bonded pairs 4) Use as lone pair@ central atom instead of electron pairs. Linear, Trigonal Planar, and Tetrahedral have NO lone pair around the central atoms.

Steps for Predicting Molecular Structures using VSEPR Model 1. Draw Lewis Structure. 2. If Lone pair @ Central Atom Bend. 3. Count the central atom, bonded atom(s), total atoms, and lone pair @ central atom. 4. Determine the name of the molecular structure. 5. Must Show 3 -D shape by using and
![Be Careful When Drawing Molecular Shapes • No Charge and No [ ] around Be Careful When Drawing Molecular Shapes • No Charge and No [ ] around](http://slidetodoc.com/presentation_image/1f492e31e964362d9d8ce0c2c6bf9201/image-5.jpg)
Be Careful When Drawing Molecular Shapes • No Charge and No [ ] around the shape • Only Draw the Lone Pair @ Central Atom Don’t draw the Left Over e- around other atoms • Bend when having Lone Pair @ central atom • When Bending, Lone Pair stay on the top, Bonds stay on the bottom. • More Lone Pair @ Central Atom Bend More

5 Molecular Shapes A) Linear (2 kinds) B) Trigonal Planar C) Tetrahedral D) Trigonal Pyramidal E) Bent (2 kinds) Total Atoms = Central Atom + Bonded Atom ? TA = ? CA + ? BA Lone Pair @ Central Atom ? LP @ CA

1) Perform NASL 2) Draw 2 D Lewis Structure 3) Give Reasons: 1) ? TA = ? CA + ? BA 2)? LP @ CA Molecular Shape Name. 4) Draw the 3 -D Molecular Shape + Label the Angle
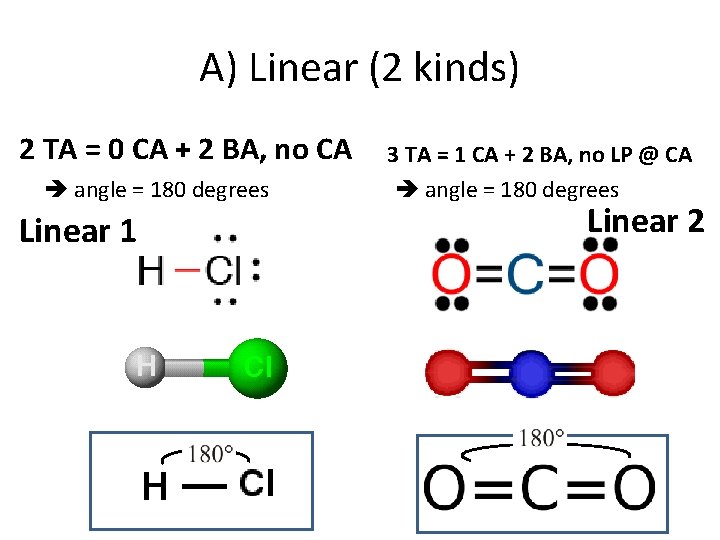
A) Linear (2 kinds) 2 TA = 0 CA + 2 BA, no CA angle = 180 degrees Linear 1 3 TA = 1 CA + 2 BA, no LP @ CA angle = 180 degrees Linear 2
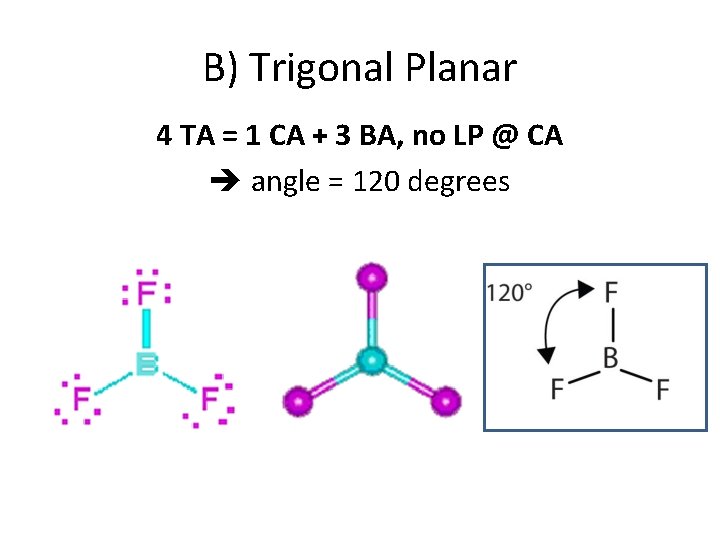
B) Trigonal Planar 4 TA = 1 CA + 3 BA, no LP @ CA angle = 120 degrees
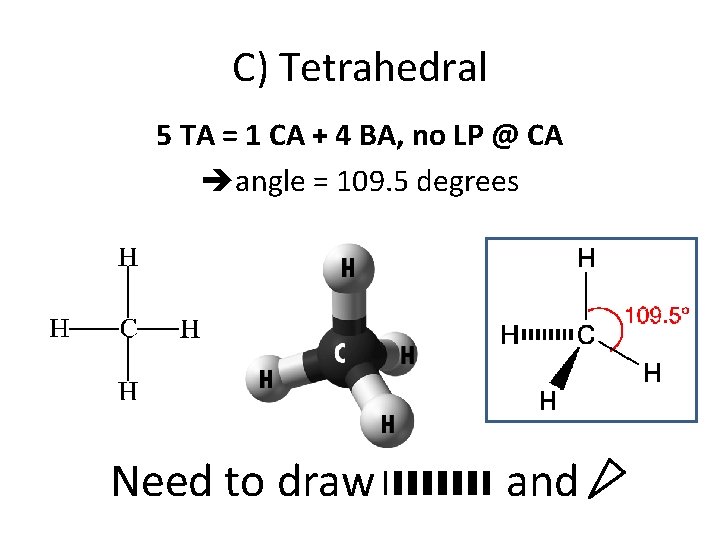
C) Tetrahedral 5 TA = 1 CA + 4 BA, no LP @ CA angle = 109. 5 degrees Need to draw and
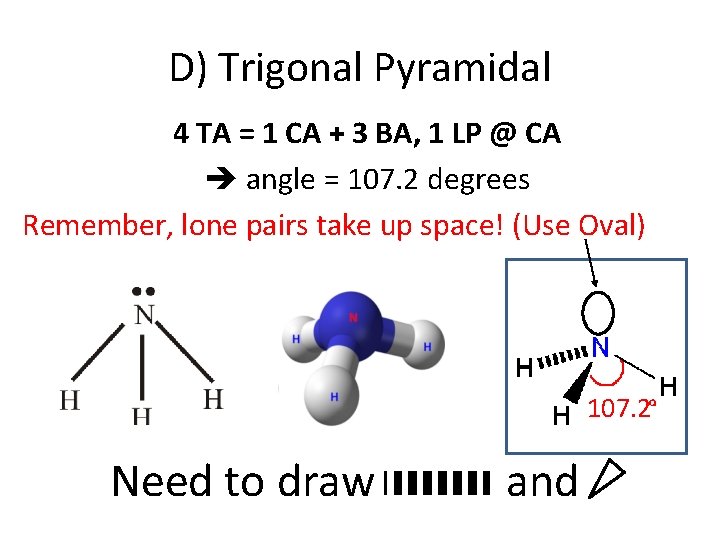
D) Trigonal Pyramidal 4 TA = 1 CA + 3 BA, 1 LP @ CA angle = 107. 2 degrees Remember, lone pairs take up space! (Use Oval) 107. 2 Need to draw and
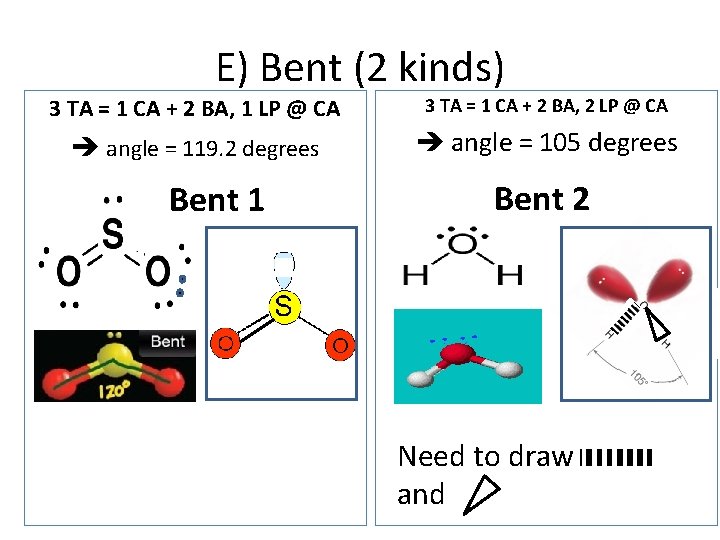
E) Bent (2 kinds) 3 TA = 1 CA + 2 BA, 1 LP @ CA 3 TA = 1 CA + 2 BA, 2 LP @ CA angle = 119. 2 degrees angle = 105 degrees Bent 1 Bent 2 Need to draw and
- Slides: 12