Molecular Shapes Lewis structures VSEPR physical properties b
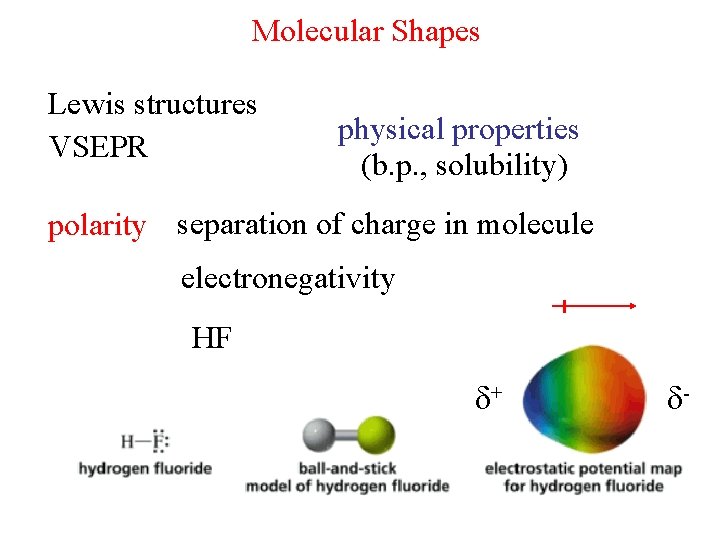
Molecular Shapes Lewis structures VSEPR physical properties (b. p. , solubility) polarity separation of charge in molecule electronegativity HF + -
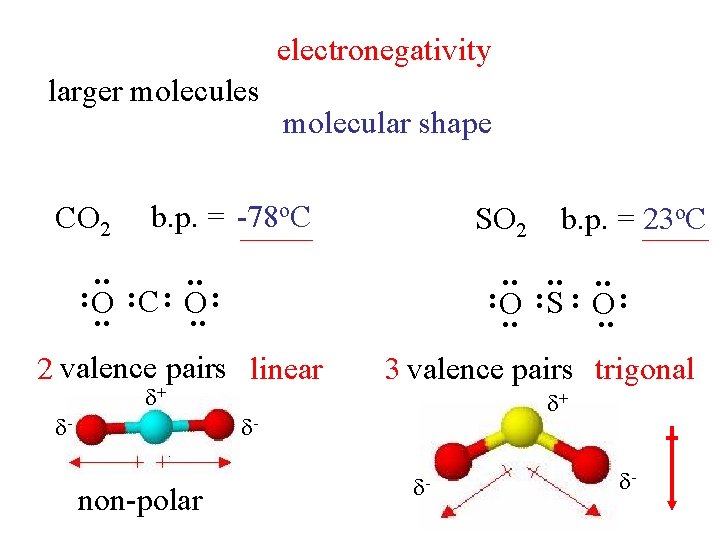
electronegativity larger molecules CO 2 molecular shape b. p. = -78 o. C SO 2 : : : : O : C : O : S : O : 2 valence pairs linear + - 3 valence pairs trigonal + - non-polar b. p. = 23 o. C - -
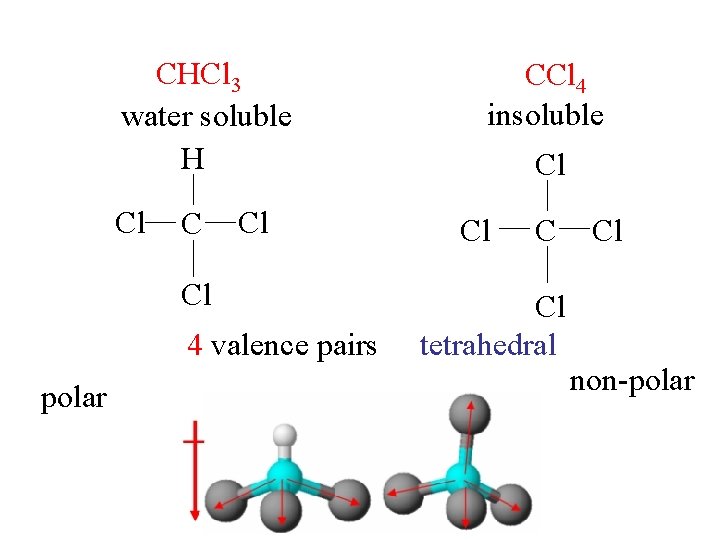
CHCl 3 water soluble H Cl Cl 4 valence pairs polar CCl 4 insoluble Cl Cl C Cl tetrahedral Cl non-polar
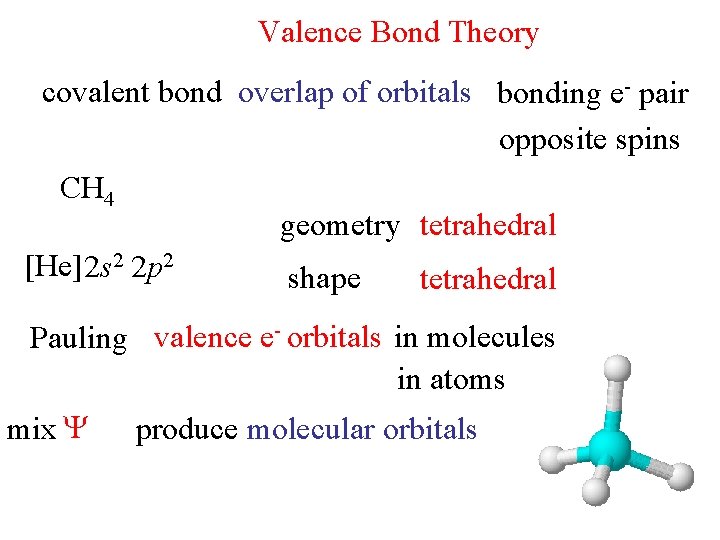
Valence Bond Theory covalent bond overlap of orbitals bonding e- pair opposite spins CH 4 geometry tetrahedral [He] 2 s 2 2 p 2 shape tetrahedral Pauling valence e- orbitals in molecules in atoms mix produce molecular orbitals
![mixing hybridization depends on CH 4 [He] 2 s 2 2 p 2 mix mixing hybridization depends on CH 4 [He] 2 s 2 2 p 2 mix](http://slidetodoc.com/presentation_image/f04af8413245f740918a249a1e167106/image-5.jpg)
mixing hybridization depends on CH 4 [He] 2 s 2 2 p 2 mix s and 3 p sp 3 number of atomic orbitals mixed type of atomic orbitals mixed need 4 equivalent orbitals mix 4 atomic orbitals sp 3
![sp 3 energy p hybridization CH 4 sp 3 s hydrogen [He] 2 s sp 3 energy p hybridization CH 4 sp 3 s hydrogen [He] 2 s](http://slidetodoc.com/presentation_image/f04af8413245f740918a249a1e167106/image-6.jpg)
sp 3 energy p hybridization CH 4 sp 3 s hydrogen [He] 2 s 2 2 p 2 sigma bond s sp 3 bond
![sp 2 formaldehyde : : H: C : H : O: [He] 2 s sp 2 formaldehyde : : H: C : H : O: [He] 2 s](http://slidetodoc.com/presentation_image/f04af8413245f740918a249a1e167106/image-7.jpg)
sp 2 formaldehyde : : H: C : H : O: [He] 2 s 2 2 p 2 CH 2 O 3 valence pairs on C 3 hybrid orbitals sp 2
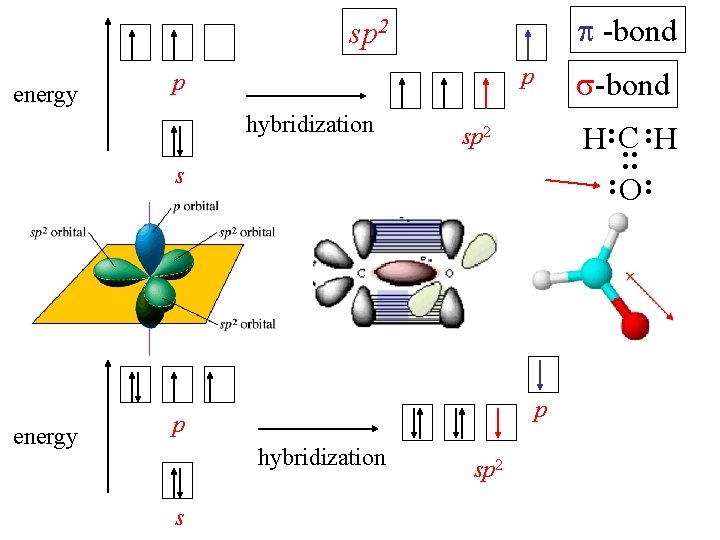
-bond sp 2 hybridization -bond H: C : H sp 2 : : energy p p s energy : O: p p hybridization s sp 2
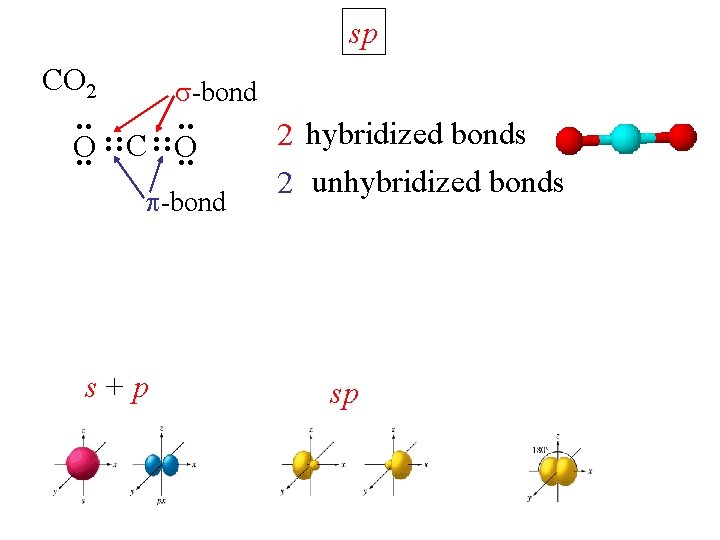
sp CO 2 : : -bond O : : C : : O -bond s+ p 2 hybridized bonds 2 unhybridized bonds sp
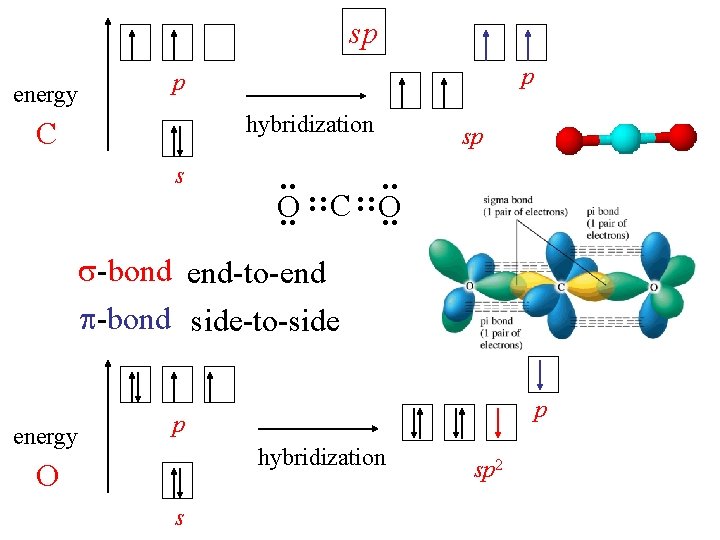
sp energy p p sp : : s : : hybridization C O : : C : : O -bond end-to-end -bond side-to-side energy p p hybridization O s sp 2
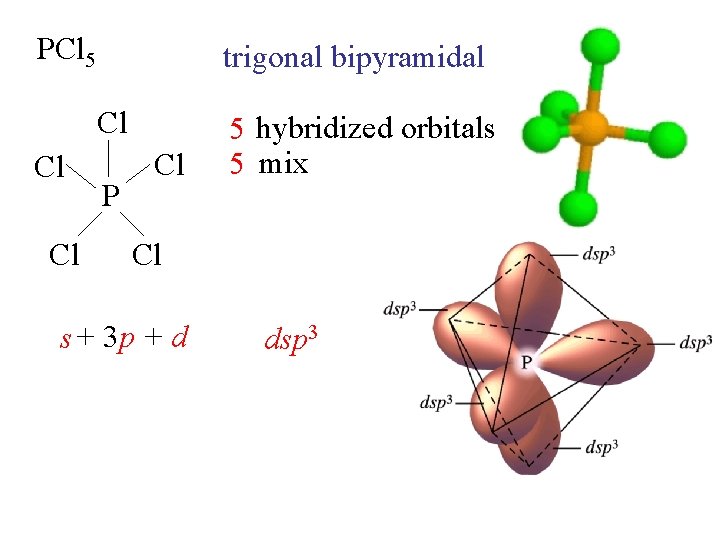
PCl 5 trigonal bipyramidal Cl Cl Cl P Cl 5 hybridized orbitals 5 mix Cl s + 3 p + d dsp 3
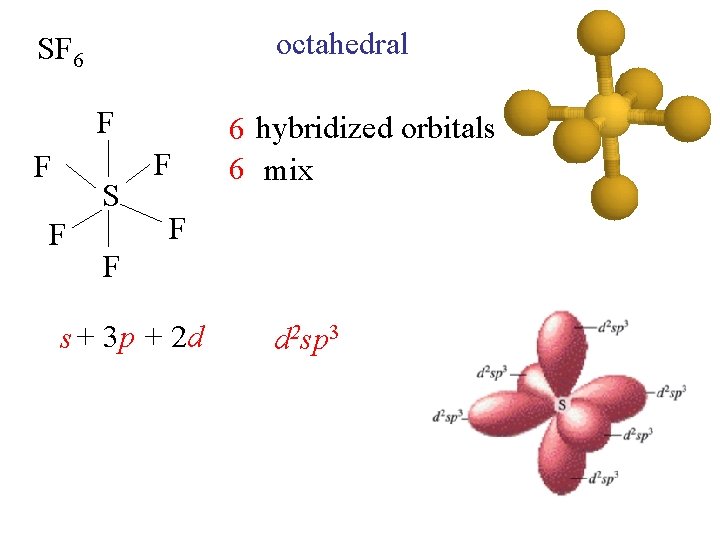
octahedral SF 6 F F S F F 6 hybridized orbitals 6 mix F F s + 3 p + 2 d d 2 sp 3
- Slides: 12