Molecular Shape VSEPR theory assumes that the shape









- Slides: 16

Molecular Shape VSEPR theory assumes that the shape of a molecule is determined by the repulsion of electron pairs.

VSEPR THEORY • Based on Electron Dot (Lewis structures) • • Theory predicts shapes of compounds abbreviated VSEPR • VSEPR (pronounced “vesper”) stands for Valence Shell Electron Pair Repulsion • VSEPR predicts shapes based on electron pairs repelling (in bonds or by themselves) eachother. • Electrons around central nucleus repel each other. Structures have atoms maximally spread out.

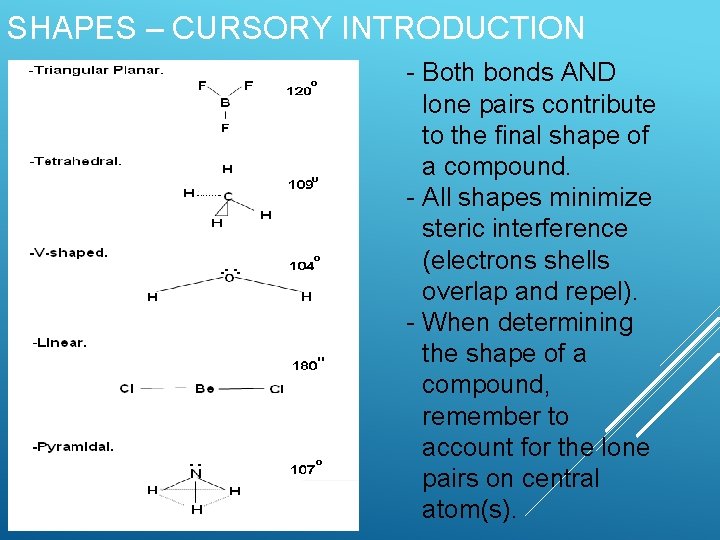
SHAPES – CURSORY INTRODUCTION - Both bonds AND lone pairs contribute to the final shape of a compound. - All shapes minimize steric interference (electrons shells overlap and repel). - When determining the shape of a compound, remember to account for the lone pairs on central atom(s).

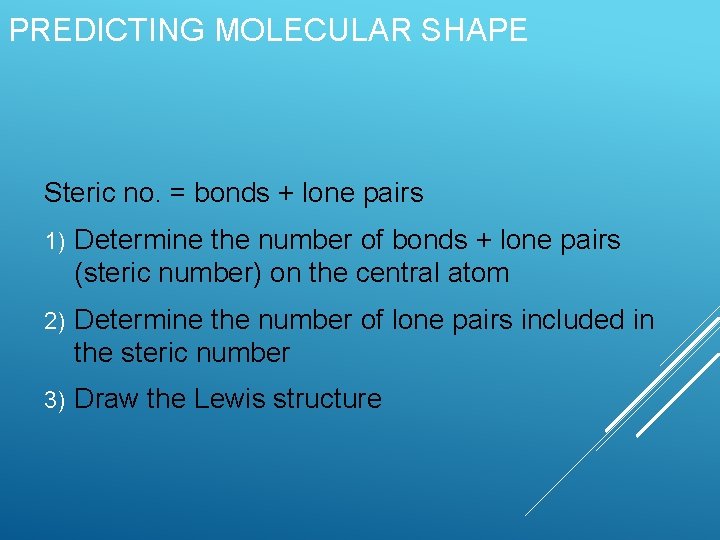
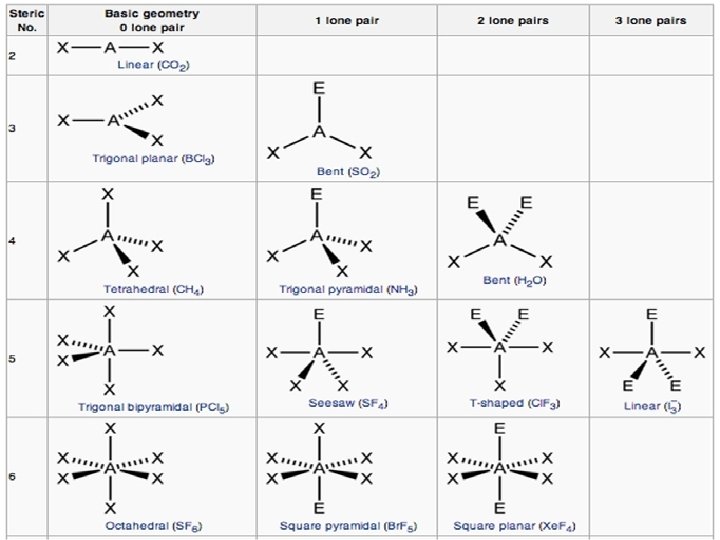
PREDICTING MOLECULAR SHAPE Steric no. = bonds + lone pairs 1) Determine the number of bonds + lone pairs (steric number) on the central atom 2) Determine the number of lone pairs included in the steric number 3) Draw the Lewis structure



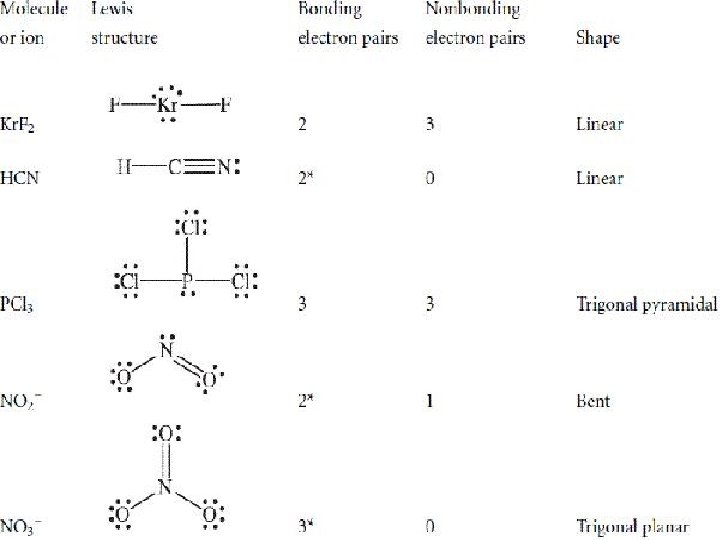
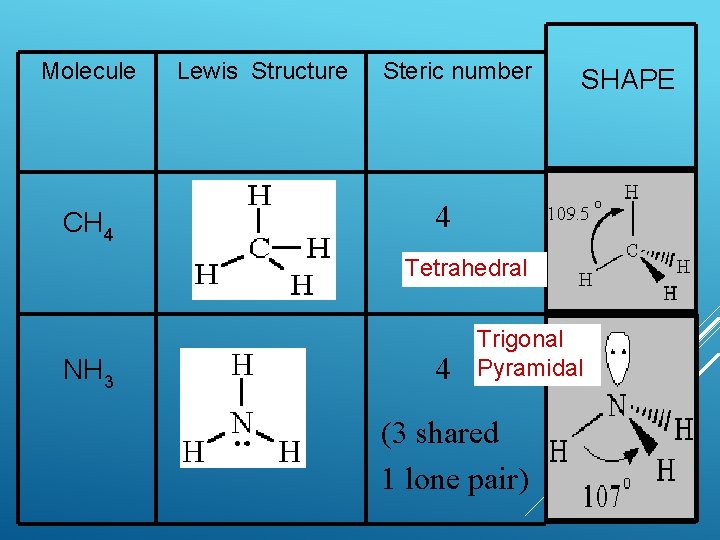
Molecule CH 4 Lewis Structure Steric number SHAPE 4 Tetrahedral NH 3 4 Trigonal Pyramidal (3 shared 1 lone pair)

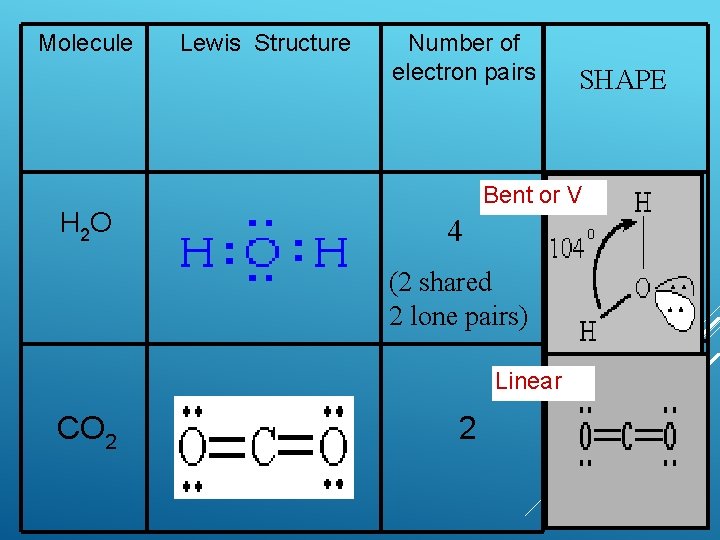
Molecule H 2 O Lewis Structure Number of electron pairs Bent or V 4 (2 shared 2 lone pairs) Linear CO 2 SHAPE 2

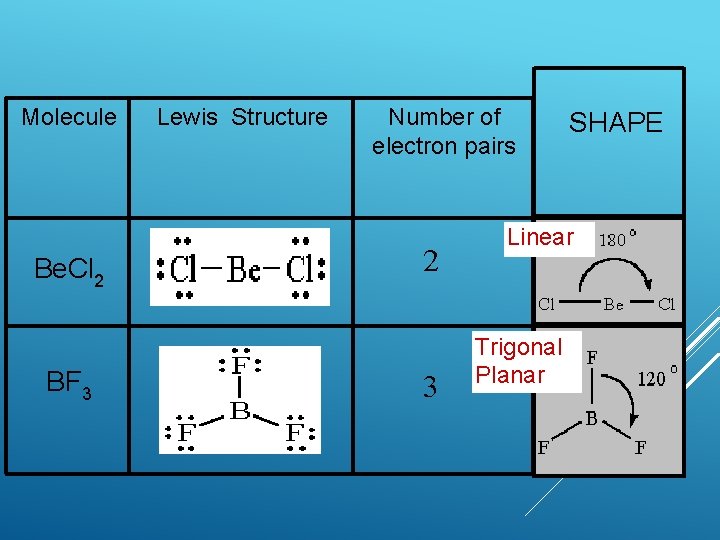
Molecule Be. Cl 2 BF 3 Lewis Structure Number of electron pairs 2 3 SHAPE Linear Trigonal Planar

POLARITY Electrons are held more closely by one atom than another Depends on electronegativity of the elements and the shape of the molecule

IONIC BONDS Metal and a non-metal Always polar, since the non-metal “holds” all electrons and the metal gives them away Electronegativity difference greater than 1. 8 Examples: Cl-K(3. 0 – 0. 8 = 2. 2) N-Na (3. 0 – 0. 9 = 2. 1)

NON-POLAR COVALENT Two or more non-metals Equal or almost equal sharing of electrons Electronegativity difference of < 0. 4 Examples: N-N (3. 0 – 3. 0 = 0. 0) Cl-Br (3. 0 – 2. 8 = 0. 2)

POLAR COVALENT Two or more non-metals Unequal sharing of electrons Electronegativity difference of 0. 5 to 1. 7 Examples: Cl – C (3. 0 – 2. 5 = 0. 5) O – S (3. 5 – 2. 5 = 1. 0)

SHOWING POLARITY One end has a partial positive charge (the less electronegative atom) and the other has a partial negative charge (the atom that is holding on to the electrons) Represented by: δ+ and δ- (in the Lewis diagram) – these are called dipoles – they cause attraction between molecules

POLARITY AND MOLECULAR GEOMETRY If the shape of a molecule is symmetrical, the polarity will cancel Generally, if the central atom has lone pairs or if the outer atoms are not identical, the molecule will be polar. Polar Shapes Non-Polar Shapes Bent (one lone pair) Linear Bent (two lone pairs) Trigonal Planar Trigonal Pyramid Tetrahedral

EXAMPLES HCl CO 2 HCN CCl 4