VSEPR Electronmolecular geometry Googleyoutube VSEPR theory and VSEPR

VSEPR Electron/molecular geometry Google-youtube: VSEPR theory and VSEPR theory introduction, part s 2 and 3. See Also Tro Page 408.
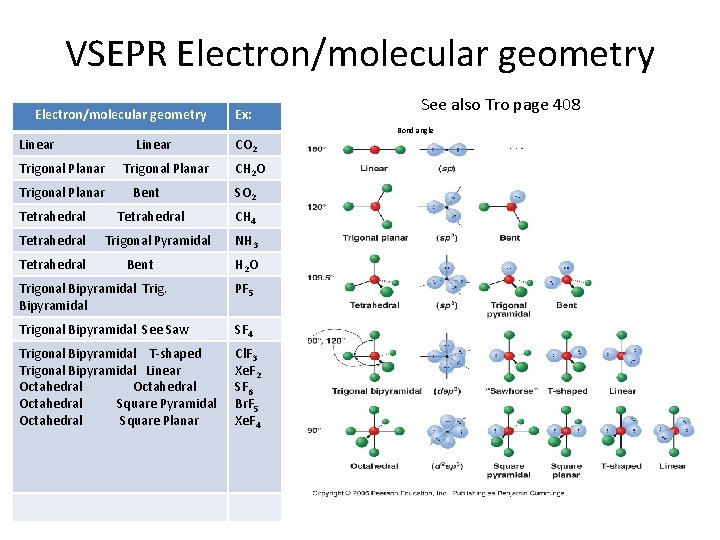
VSEPR Electron/molecular geometry Ex: See also Tro page 408 Bond angle Linear Trigonal Planar Tetrahedral Linear Trigonal Planar Bent Tetrahedral Trigonal Pyramidal Bent CO 2 CH 2 O SO 2 CH 4 NH 3 H 2 O Trigonal Bipyramidal Trig. Bipyramidal PF 5 Trigonal Bipyramidal See Saw SF 4 Trigonal Bipyramidal T-shaped Trigonal Bipyramidal Linear Octahedral Square Pyramidal Octahedral Square Planar Cl. F 3 Xe. F 2 SF 6 Br. F 5 Xe. F 4
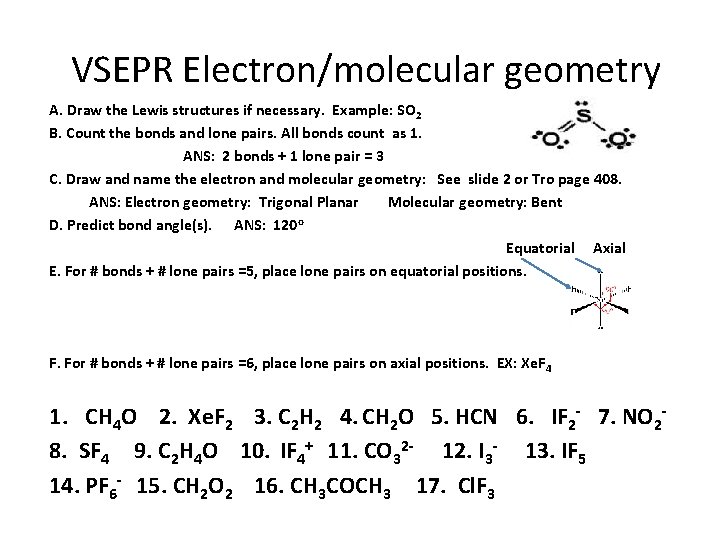
VSEPR Electron/molecular geometry A. Draw the Lewis structures if necessary. Example: SO 2 B. Count the bonds and lone pairs. All bonds count as 1. ANS: 2 bonds + 1 lone pair = 3 C. Draw and name the electron and molecular geometry: See slide 2 or Tro page 408. ANS: Electron geometry: Trigonal Planar Molecular geometry: Bent D. Predict bond angle(s). ANS: 120 o Equatorial Axial E. For # bonds + # lone pairs =5, place lone pairs on equatorial positions. F. For # bonds + # lone pairs =6, place lone pairs on axial positions. EX: Xe. F 4 1. CH 4 O 2. Xe. F 2 3. C 2 H 2 4. CH 2 O 5. HCN 6. IF 2 - 7. NO 28. SF 4 9. C 2 H 4 O 10. IF 4+ 11. CO 32 - 12. I 3 - 13. IF 5 14. PF 6 - 15. CH 2 O 2 16. CH 3 COCH 3 17. Cl. F 3
- Slides: 3