Lecture Presentation Chapter 9 Molecular Geometry and Bonding

Lecture Presentation Chapter 9 Molecular Geometry and Bonding Theories 考題詳解 James F. Kirby Quinnipiac University Hamden, CT















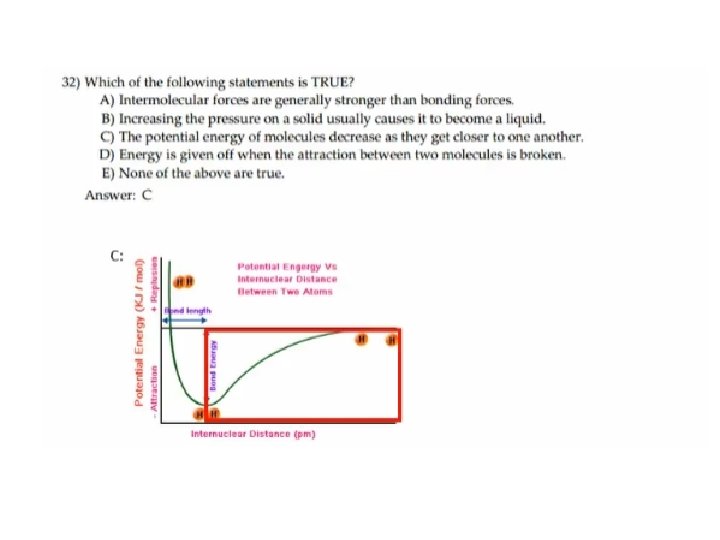


31) The basis of the VSEPR model of molecular bonding is _____. A) hybrid orbitals will form as necessary to, as closely as possible, achieve spherical symmetry B) regions of electron density on an atom will organize themselves so as to maximize s-character C) electron domains in the valence shell of an atom will arrange themselves so as to minimize repulsions D) atomic orbitals of the bonding atoms must overlap for a bond to form E) regions of electron density in the valence shell of an atom will arrange themselves so as to maximize overlap

31) The basis of the VSEPR model of molecular bonding is _____. A) hybrid orbitals will form as necessary to, as closely as possible, achieve spherical symmetry B) regions of electron density on an atom will organize themselves so as to maximize s-character C) electron domains in the valence shell of an atom will arrange themselves so as to minimize repulsions D) atomic orbitals of the bonding atoms must overlap for a bond to form E) regions of electron density in the valence shell of an atom will arrange themselves so as to maximize overlap Answer: C 分子結合的VSEPR模型的基礎是_______。 A)混成軌域要形成,是盡可能接近地實現球形對稱性(無此要求) B)原子上的電子密度區域將自身最大化s特性(無此要求) C)原子的價電子殼中的電子域將自身排列,以便最小化排斥(正確 ) D)原子軌域鍵結原子必須重疊以形成鍵(不用重疊 例如反鍵結軌 域) E)電子密度在一個原子的價電子殼中的區域將自己排列以便最大 化重疊(沒有特別說明 ) 課本 9. 1 p 384

32) The electron-domain geometry of _____ is tetrahedral. A) PH 3 B) CBr 4 C) Xe. F 4 D) CCl 2 Br 2 E) all of the above except Xe. F 4
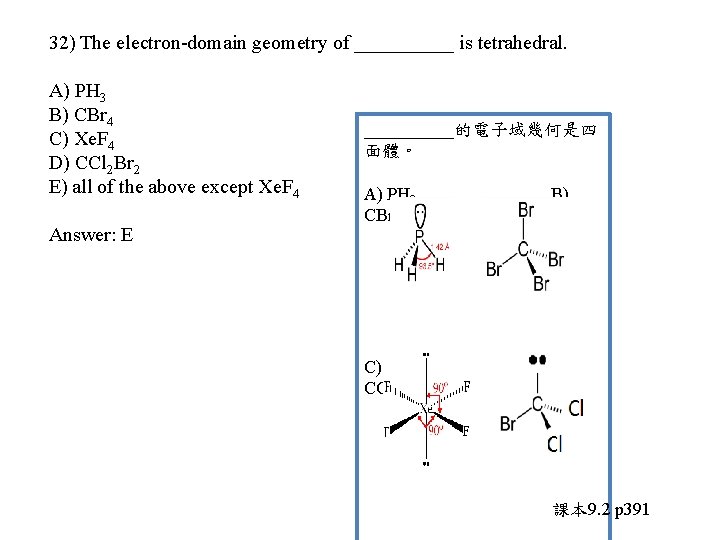
32) The electron-domain geometry of _____ is tetrahedral. A) PH 3 B) CBr 4 C) Xe. F 4 D) CCl 2 Br 2 E) all of the above except Xe. F 4 Answer: E _____的電子域幾何是四 面體。 A) PH 3 CBr 4 B) C) Xe. F 4 CCl 2 Br 2 D) 課本 9. 2 p 391

33) The central Xe atom in the Xe. F 4 molecule has _____ unbonded electron pairs and _____ bonded electron pairs in its valence shell. A) 4, 0 B) 2, 4 C) 4, 1 D) 1, 4 E) 4, 2

33) The central Xe atom in the Xe. F 4 molecule has _____ unbonded electron pairs and _____ bonded electron pairs in its valence shell. A) 4, 0 B) 2, 4 C) 4, 1 D) 1, 4 E) 4, 2 Answer: B Xe. F 4分子中的中心Xe原子具有未鍵合的電子對和鍵在其 價鍵殼中的電子對。 課本 9. 2 p 388

34) The molecular geometry of the PF 3 molecule is _____, and this molecule is _____. A) trigonal planar, polar B) trigonal pyramidal, polar C) trigonal planar, nonpolar D) tetrahedral, unipolar E) trigonal pyramidal, nonpolar
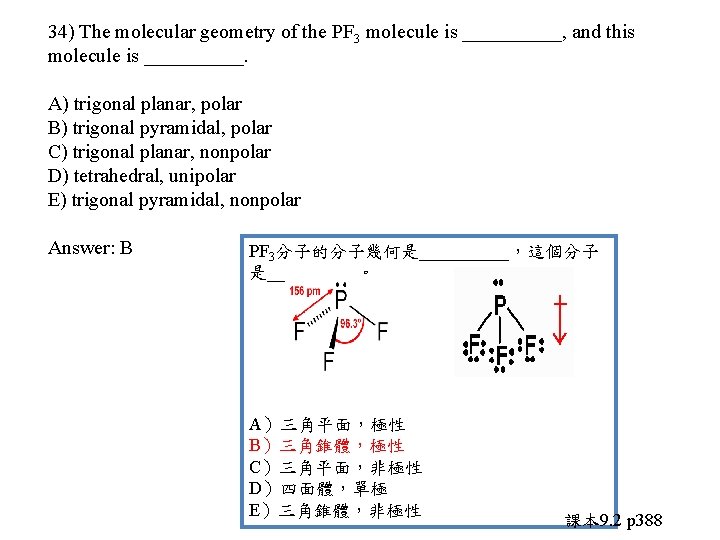
34) The molecular geometry of the PF 3 molecule is _____, and this molecule is _____. A) trigonal planar, polar B) trigonal pyramidal, polar C) trigonal planar, nonpolar D) tetrahedral, unipolar E) trigonal pyramidal, nonpolar Answer: B PF 3分子的分子幾何是_____,這個分子 是_____。 A)三角平面,極性 B)三角錐體,極性 C)三角平面,非極性 D)四面體,單極 E)三角錐體,非極性 課本 9. 2 p 388
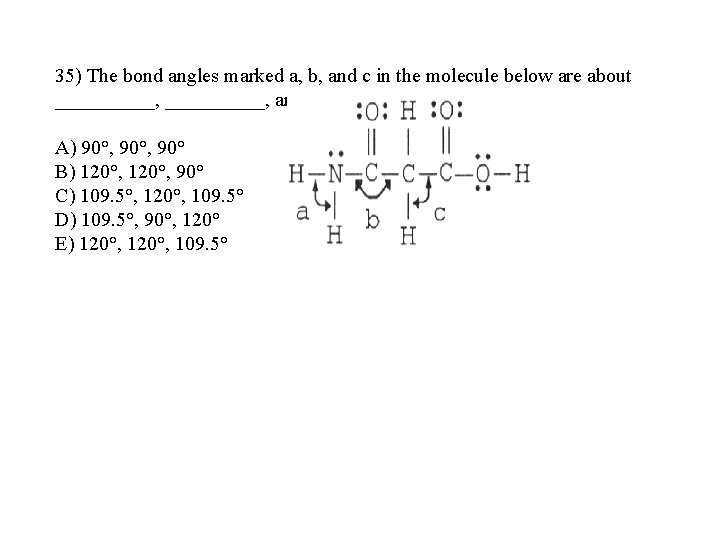
35) The bond angles marked a, b, and c in the molecule below are about __________, and _____, respectively A) 90°, 90° B) 120°, 90° C) 109. 5°, 120°, 109. 5° D) 109. 5°, 90°, 120° E) 120°, 109. 5°
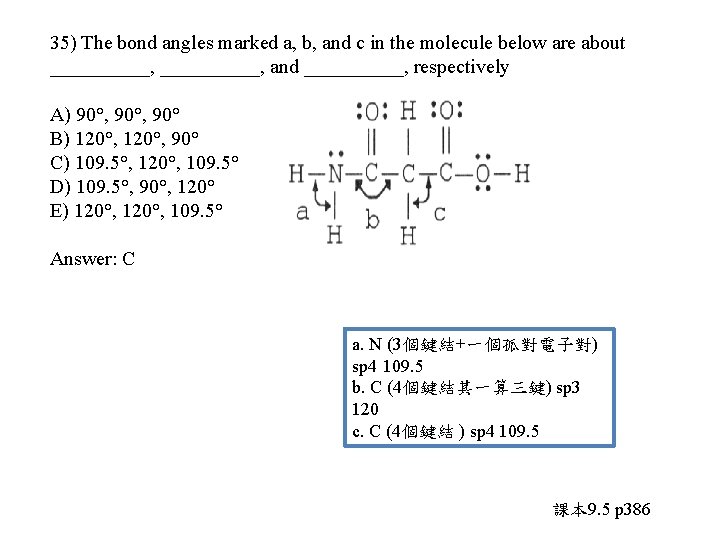
35) The bond angles marked a, b, and c in the molecule below are about __________, and _____, respectively A) 90°, 90° B) 120°, 90° C) 109. 5°, 120°, 109. 5° D) 109. 5°, 90°, 120° E) 120°, 109. 5° Answer: C a. N (3個鍵結+一個孤對電子對) sp 4 109. 5 b. C (4個鍵結其一算三鍵) sp 3 120 c. C (4個鍵結 ) sp 4 109. 5 課本 9. 5 p 386

36) The hybridizations of iodine in IF 3 and IF 5 are _____ and _____, respectively. A) sp 3 d 2, sp 3 d 2 B) sp 3, sp 3 d C) sp 3 d, sp 3 D) sp 3 d 2, sp 3 d E) sp 3 d, sp 3 d 2 Answer: E
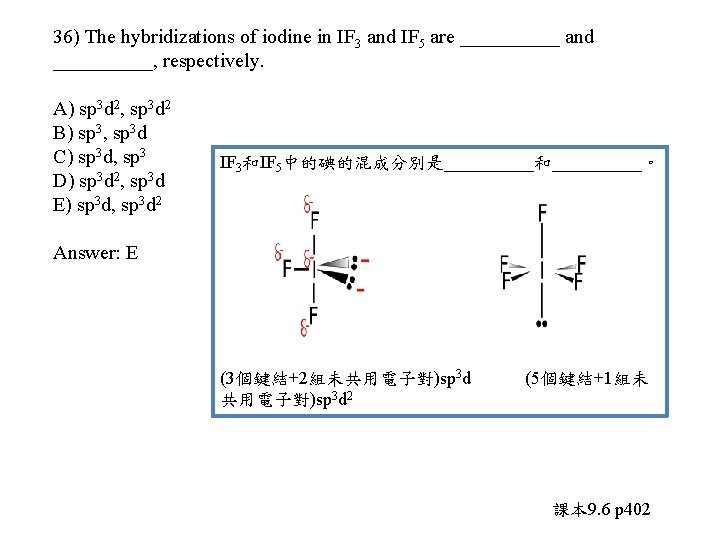
36) The hybridizations of iodine in IF 3 and IF 5 are _____ and _____, respectively. A) sp 3 d 2, sp 3 d 2 B) sp 3, sp 3 d C) sp 3 d, sp 3 D) sp 3 d 2, sp 3 d E) sp 3 d, sp 3 d 2 IF 3和IF 5中的碘的混成分別是_____和_____。 Answer: E (3個鍵結+2組未共用電子對)sp 3 d 2 (5個鍵結+1組未 課本 9. 6 p 402

37) Based on molecular orbital theory, the bond orders of the H�� H bonds in H 2, H 2+, and H 2 - are _____, respectively A) 1, 1/2, and 0 B) 1, 1/2, and 1/2 C) 1, 0, and 1/2 D) 1, 0, and 0 E) 1, 2, and 0
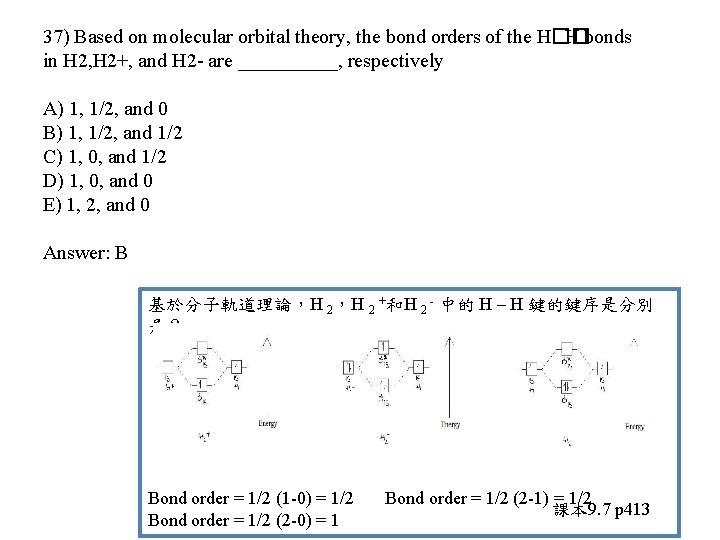
37) Based on molecular orbital theory, the bond orders of the H�� H bonds in H 2, H 2+, and H 2 - are _____, respectively A) 1, 1/2, and 0 B) 1, 1/2, and 1/2 C) 1, 0, and 1/2 D) 1, 0, and 0 E) 1, 2, and 0 Answer: B 基於分子軌道理論,H 2 +和H 2 - 中的 H – H 鍵的鍵序是分別 是? Bond order = 1/2 (1 -0) = 1/2 Bond order = 1/2 (2 -0) = 1 Bond order = 1/2 (2 -1) = 1/2 課本 9. 7 p 413

38) A molecule has the formula AB 3 and the central atom is in a different plane from the surrounding three atoms. Its molecular shape is _____. A) bent B) tetrahedral C) trigonal pyramidal D) T-shaped E) linear
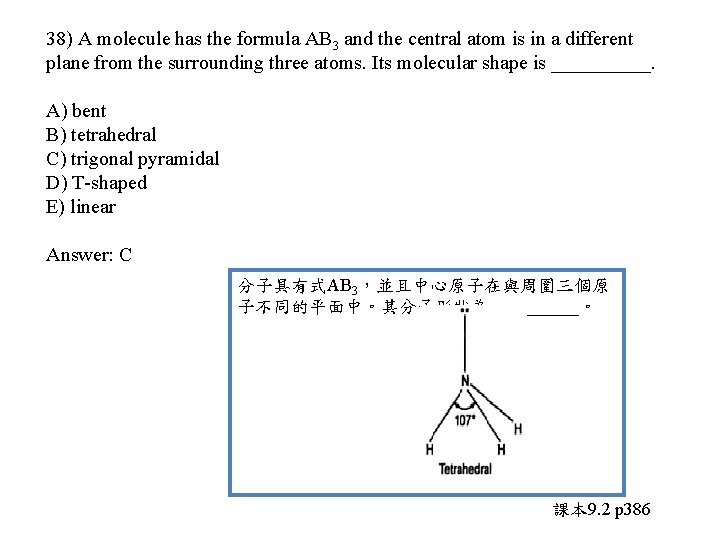
38) A molecule has the formula AB 3 and the central atom is in a different plane from the surrounding three atoms. Its molecular shape is _____. A) bent B) tetrahedral C) trigonal pyramidal D) T-shaped E) linear Answer: C 分子具有式AB 3,並且中心原子在與周圍三個原 子不同的平面中。其分子形狀為_____。 課本 9. 2 p 386
39) PCl 5 has _____ electron domains and a _____ molecular arrangement. A) 5, square pyramidal B) 6, trigonal bipyramidal C) 6, tetrahedral D) 5, trigonal bipyramidal E) 6, seesaw
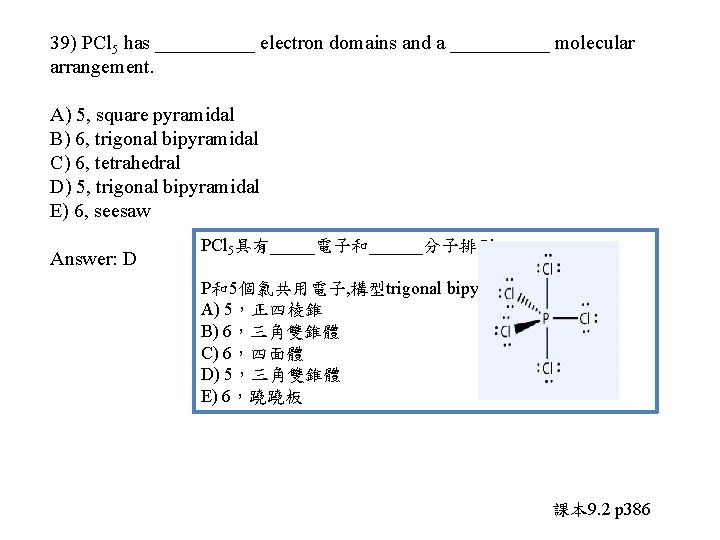
39) PCl 5 has _____ electron domains and a _____ molecular arrangement. A) 5, square pyramidal B) 6, trigonal bipyramidal C) 6, tetrahedral D) 5, trigonal bipyramidal E) 6, seesaw Answer: D PCl 5具有_____電子和______分子排列。 P和5個氯共用電子, 構型trigonal bipyramidal A) 5,正四棱錐 B) 6,三角雙錐體 C) 6,四面體 D) 5,三角雙錐體 E) 6,蹺蹺板 課本 9. 2 p 386

40) Three monosulfur fluorides are observed: SF 2, SF 4, and SF 6. Of these, _____ is/are polar. A) SF 2 only B) SF 4 only C) SF 2, SF 4, and SF 6 D) SF 6 only E) SF 2 and SF 4 only
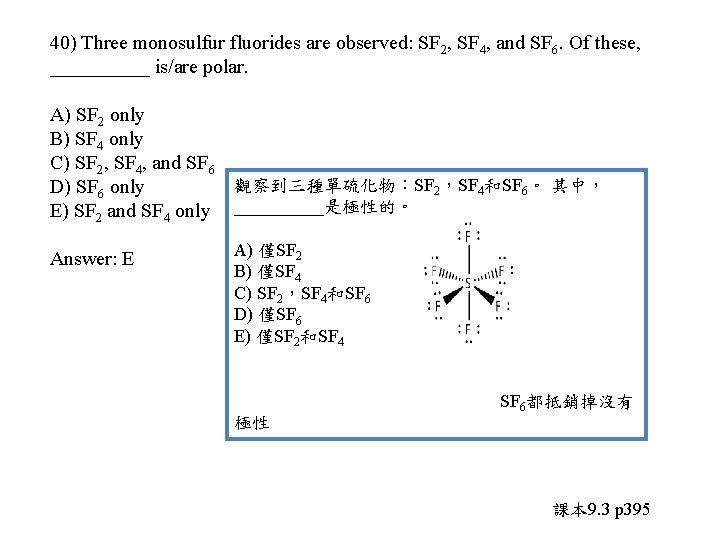
40) Three monosulfur fluorides are observed: SF 2, SF 4, and SF 6. Of these, _____ is/are polar. A) SF 2 only B) SF 4 only C) SF 2, SF 4, and SF 6 觀察到三種單硫化物:SF 2,SF 4和SF 6。 其中, D) SF 6 only E) SF 2 and SF 4 only _____是極性的。 Answer: E A) 僅SF 2 B) 僅SF 4 C) SF 2,SF 4和SF 6 D) 僅SF 6 E) 僅SF 2和SF 4 SF 6都抵銷掉沒有 極性 課本 9. 3 p 395
- Slides: 46