VSEPR Theory and Molecular Shapes Lab Objective Today

VSEPR Theory and Molecular Shapes Lab

Objective Today I will be able to: Draw Lewis Structures to represent the valance electrons of atoms Represent the formation of ionic and covalent compounds using Lewis Structures Predict the molecular shape of a molecule using the VSEPR theory Evaluation/ Assessment Informal assessment – Listening to group interactions as they complete the illustrating ionic and covalent compounds practice and the molecular shapes lab. Formal Assessment – Analyzing student responses to the exit ticket and the practice worksheets. Common Core Connection Make sense of problem and persevere in solving them Reason abstractly and quantitatively Use appropriate tools strategically Look for and make use of structure

Lesson Sequence Warm – Up Explain: VSEPR Theory (Day 1) Explore and elaborate: Molecular Shapes Lab (Day 1 & 2) Informal Assessment Evaluate: Exit Ticket • Formal assessment

Warm-Up Draw the Lewis Structures for the following elements: -Sulfur -Potassium -Beryllium cation Which molecular geometry is made up of all 90° bond angles? Draw the structure that represents the geometry for bent (tetrahedral) Don't forget about lone pairs!

Objective Today I will be able to: Draw Lewis Structures to represent the valence electrons of atoms Represent the formation of ionic and covalent compounds using Lewis Structures Predict the molecular shape of a molecule using the VSEPR theory
Homework STEM Fair In Class Presentations Wednesday January 22

Agenda Warm-Up VSEPR Notes (Day 1) Molecular Shapes Lab (Day 1 & 2) Exit Ticket
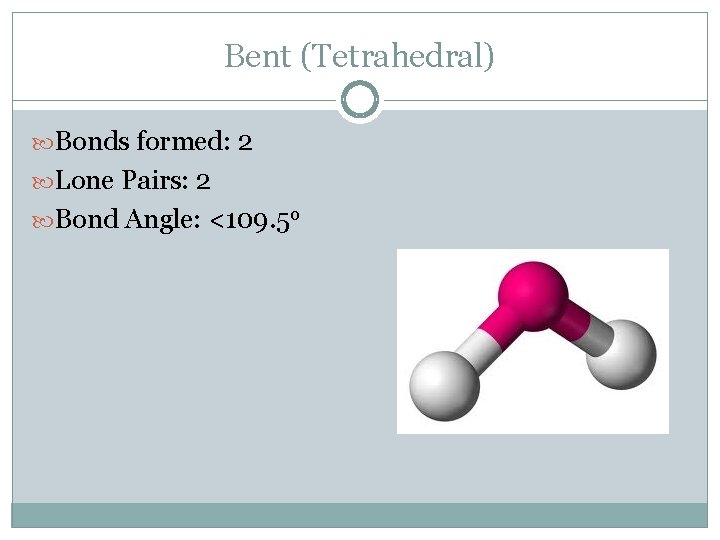
Bent (Tetrahedral) Bonds formed: 2 Lone Pairs: 2 Bond Angle: <109. 5 o
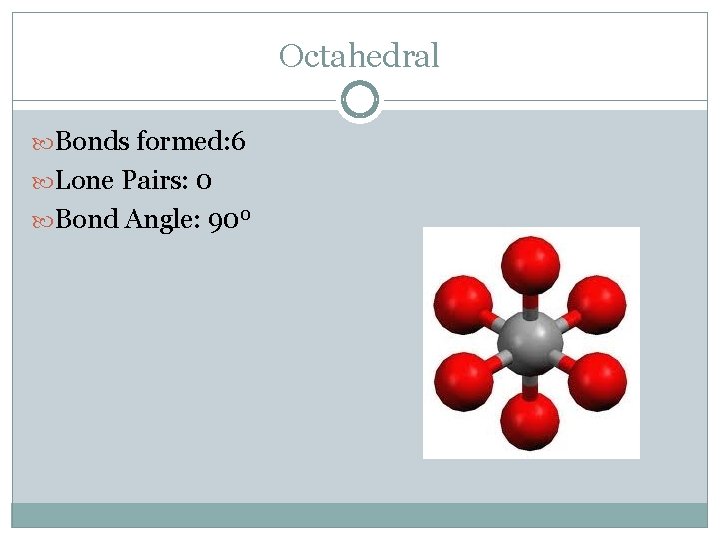
Octahedral Bonds formed: 6 Lone Pairs: 0 Bond Angle: 900
Molecular Shapes Lab

Molecular Shapes Lab Directions Draw a Lewis Dot Structure for the molecule Make a model using the kit and draw the shape on your paper Write the shape name Write the bond angle Skip POLARITY for now, and we will come back to this part Be careful with the kits, do not drop or lose pieces!

Exit Ticket Draw the Lewis Structure for CO 2 Determine the shape and bond angles of CO 2 according to the VSEPR theory.
- Slides: 12