Tetramer analysis by Sadegh feizollahzadeh 1 Monitoring vaccine

Tetramer analysis by Sadegh feizollahzadeh

1. Monitoring vaccine studies 2. Monitoring effect of Immunotherapy 3. Monitoring T-cell responses in infectious disease, cancer, autoimmunity and other malignancies 4. Detection of antigen-specific T-cell responses in immunological research
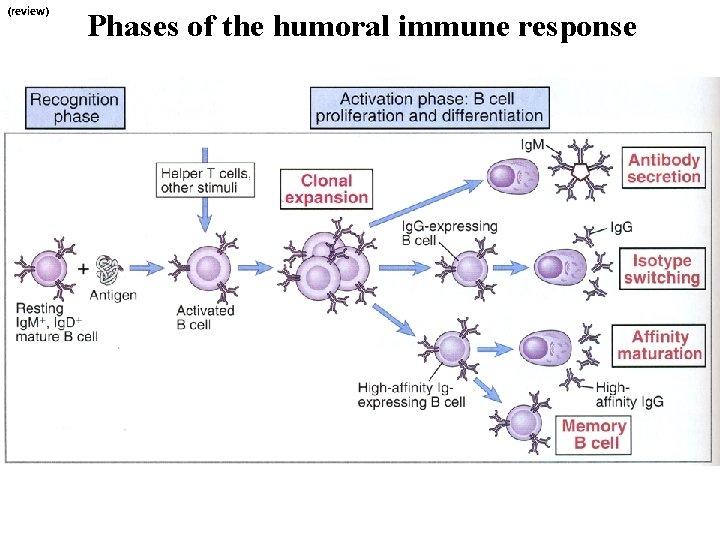
(review) Phases of the humoral immune response
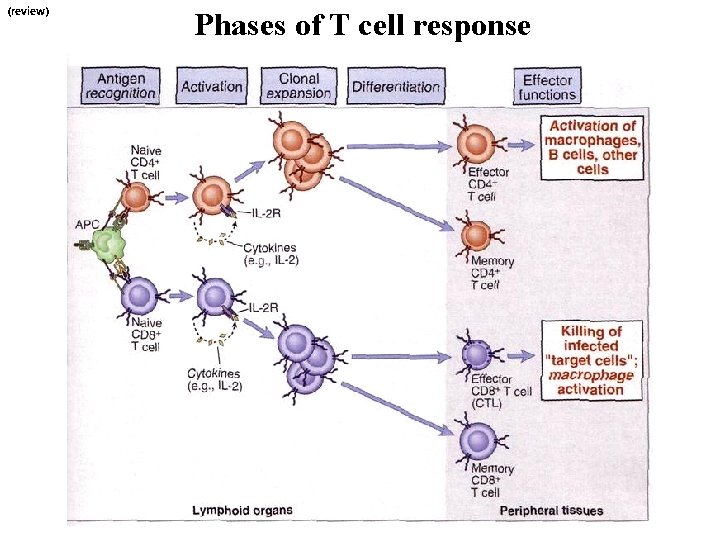
(review) Phases of T cell response
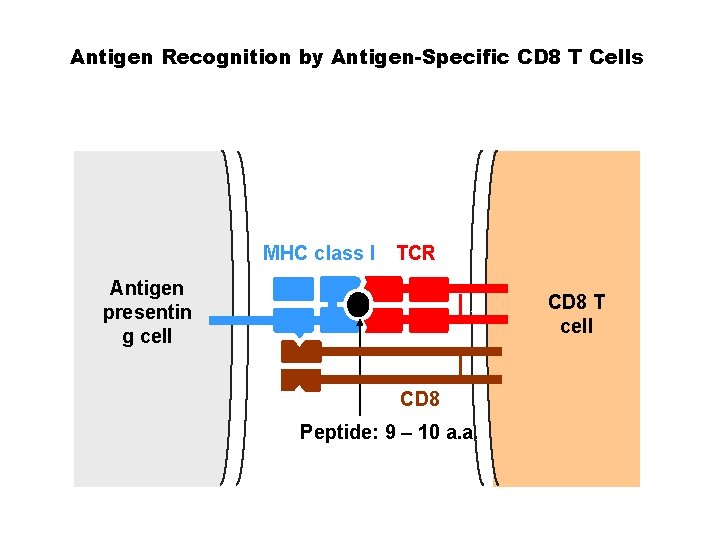
Antigen Recognition by Antigen-Specific CD 8 T Cells MHC class I TCR Antigen presentin g cell CD 8 T cell CD 8 Peptide: 9 – 10 a. a.
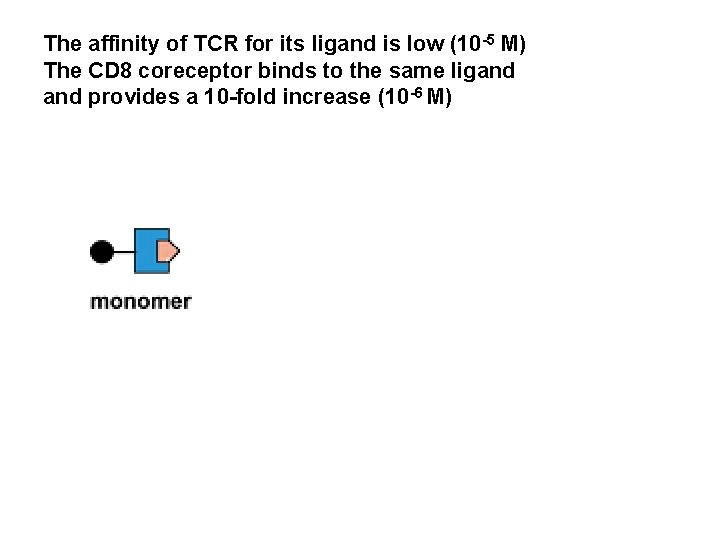
The affinity of TCR for its ligand is low (10 -5 M) The CD 8 coreceptor binds to the same ligand provides a 10 -fold increase (10 -6 M)
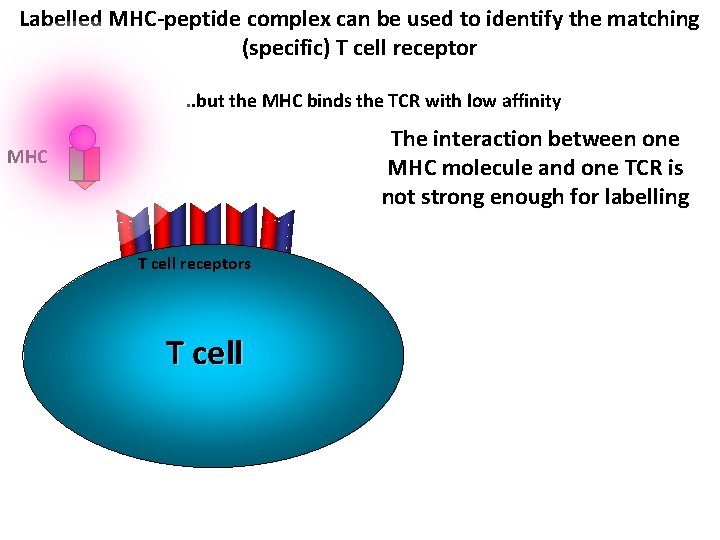
Labelled MHC-peptide complex can be used to identify the matching (specific) T cell receptor. . but the MHC binds the TCR with low affinity The interaction between one MHC molecule and one TCR is not strong enough for labelling MHC T cell receptors T cell
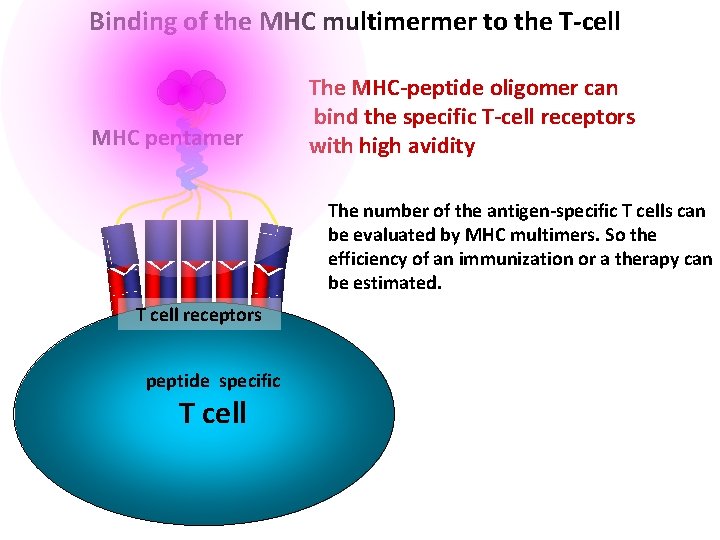
Binding of the MHC multimermer to the T-cell MHC pentamer The MHC-peptide oligomer can bind the specific T-cell receptors with high avidity The number of the antigen-specific T cells can be evaluated by MHC multimers. So the efficiency of an immunization or a therapy can be estimated. T cell receptors peptide specific T cell

• Peptid/MHC (p. MHC) tetramers were invented and first reported by Altman and Davis in 1996. • This novel reagents have revolutionized the study of T cell responses by allowing fore accurate and rapid: 1. enumeration of antigen-specific T cells, 2. The phenotypic characterization of such cells and 3. Their viable isolation for further analysis.



BIOLOGY OF TCR-p. MHC INTERACTIONS AND IMPLICATIONS IN TETRAMERS • The affinity of the TCR-p. MHC interaction in solution is much lower than that of an antibody–antigen interaction. • The typical dissociation constant (Kd) of an antibody–antigen interaction is around 1 n. M, a typical TCR-p. MHC interaction may be in the range of 10 M. • Previous efforts to use soluble p. MHC complexes as a staining reagent to identify antigen-specific T cells were unsuccessful. • Potentially three p. MHC complexes can interact with three TCRs simultaneously on a T cell surface, such that the avidity of this interaction becomes very strong, making staining possible.

COMPARISONS TO OTHER ASSAYS • limiting dilution assays (LDA), cytokine flow cytometry (CFC) and ELISPOT analyses. • Typically, tetramer analyses have provided frequency estimates that exceed those detected using these alternate assay systems. • when combined with a functional readout, this yields important additional information • Such a combination was used successfully to demonstrate for the first time that a tumor-specific T cell population was anergic in vivo
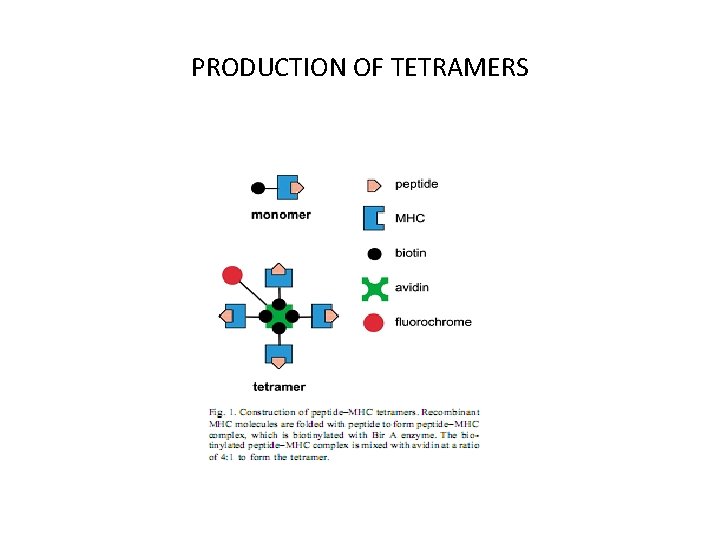
PRODUCTION OF TETRAMERS

• The MHC class I molecule is engineered with a C-terminal substrate peptide for Bir. A-mediated biotinylation, biotin signal peptide (bsp). • A Bir. A substrate peptide (usually the 15 -mer, LHHILDAQKMVWNHR) is fused to the extracellular portion of the MHC molecule. • This MHC I-bsp recombinant protein (as well as β 2 m) is generally produced using E. coli in inclusion bodies (BL 21 DE 3 systems). • Briefly, recombinant MHC I-bsp, 2 m and the peptide of interest are mixed together in a folding reaction which generally goes over 48– 72 h.

• Resulting in a 1– 5 per cent final yield. • Properly folded p. MHC complexes are extensively purified from unfolded materials via size exclusion columns (FPLC) and further cleaned up with anion exchange. • During this process, the biotinylation reaction is carried out via the addition of biotin-ligase (Bir. A enzyme), biotin and ATP. • Streptavidin directly conjugated to a fluorophore (generally phycoerythrin or APC) is used to facilitate their use in flow cytometry or sorting.

• Under optimal conditions, the lower level of detection for tetramer-based assays is approximately 1/8000– 1/10 000 (i. e. 0. 01– 0. 0125 per cent). • periodic retesting of tetramers by retitration analyses on a regular (every 6– 8 weeks) basis depending on the peptide used.

BASIC METHODOLOGY AND IMPORTANT FACTORS • • • Quality and numbers of cells Staining volume, temperature and time Tetramer concentration Anti-CD 8 and other antibodies Data interpretation

• The same numbers of cells should be added per stain in all tetramer assays. • one stains 1– 2 million PBMCs per condition and collects as many events as possible (105– 106) for each analysis (0. 01 per cent of CD 8+ T cells). • As an example, if one collects 5× 105 events and CD 8+T cells represent 10 per cent of PBMC, then a 0. 01 per cent antigenspecific population would represent five events. • frozen/thawed PBMC samples may be used without compromising the quality of tetramer staining data.

• To maximize cell viability and quality, thawed cells may be ‘rested’ in complete media (containing human serum) for several hours to overnight and then enriched by discontinuous gradient centrifugation (Ficoll/Percoll) prior to staining. • As an example, CD 62 L (an important marker for the distinction between naive versus memory T cells) contains glycosylations which may be cleaved during the freeze/thaw process.

Staining volume, temperature and time • To conserve reagents, it is advisable to stain in a small volume, generally 30– 50μ l. This is adequate for 1– 2 million PBMCs. • The temperature at which the staining occurs may significantly impact the degree of specific tetramer staining. • The main reason to perform tetramer staining on ice rather than room temperature is to favor detection of low avidity tetramer–T cell interactions. • At room temperature (23˚C) or 37˚C, tetramer staining generally saturates at 15– 30 min, while on ice (or at 4˚C) saturation does not occur until at least 2 h.

Tetramer concentration • Staining with suboptimal tetramer concentrations may lead to an underestimate of antigen-specific T cell frequencies, while staining with excessive tetramer concentrations may lead to high backgrounds and overestimate of antigen-specific T cell frequencies. • For most tetramers, they are generally used at a 1– 20 g/ml final concentration.

Anti-CD 8 and other antibodies • The MHC I molecule naturally has a low affinity for CD 8. • Depending on the CD 8 determinant recognized by the antibody, anti-CD 8 counterstaining may block, have little impact on, or even augment the intensity of p. MHC tetramer staining of T cells. • Tetramers from Beckman Coulter Immunomics are made with non-CD 8 binding mutant MHC I molecules. • Antibodies to markers not present on the cells of interest – for CD 8+T cells, useful ‘dump’ antibodies may include anti. CD 4, 14, or 19.


Data interpretation • Specific tetramer staining of T cells may be confirmed via competition with unlabeled p. MHC monomers or anti-CD 3 antibodies. • Tetramer-positive events are generally ‘normalized’ as a percentage of total CD 8+ T cells. • Data may be reported as the absolute number of tetramer+, CD 8+cells per amount (i. eμ/ml) of blood.

ADVANCED USES OF TETRAMERS • • • Isolation by sorting and cloning Phenotypic analysis by multi-color flow cytometry Intracellular staining Analysis of ‘structural avidity’ Functional analysis In situ hybridization

Isolation by sorting and cloning • Care should be taken to keep the cells cold and in serumrich media to maximize their function before and after sorting.

Phenotypic analysis by multi-color flow cytometry • Even with three- or four- color FACS, the expression of several markers by an antigen-specific T cell population of interest may be determined by performing a few different stains. • With the increasing availability of six- and even 10 -color FACS (Table 22. 2), large numbers of markers could be determined from only a few stains. • Not all antibodies work well in combination, so considerable effort must be made to optimize phenotypic panels which work well together.


Intracellular staining • Tetramers can be combined successfully with intracellular staining to analyze expression of cytokines (e. g. IFN-γ, IL- 2, IL 4, TNF-α) or cytolytic granules (perforin, granzyme A, granzyme B) by antigen-specific T cells. • Start with large numbers of cells. • Tetramer staining could be performed prior to cell activation.

Analysis of ‘structural avidity’ • ‘Functional avidity’ or ‘recognition efficiency’ is emerging as a key factor in the effectiveness of an antigen-specific T cell response. • The rate of dissociation of bound p. MHC tetramers from antigen-specific T cells upon addition of a competing antibody (such as anti-TCR) may be used as a relative measure of the difference in TCR affinities between T cells.

Functional analysis • Staining for cytokine production, cytolytic granules and direct ex vivo testing for cytolytic activity after sorting. • CD 107 mobilization could be combined with peptide/MHC tetramer staining to directly correlate antigen-specificity and cytolytic ability on a single-cell level.

In situ hybridization • This is a technically challenging procedure which requires extensive optimization of staining conditions. • Most successful in situ tetramer staining has been achieved using fresh or frozen tissue sections, although some success has also been achieved with ‘lightly fixed’ tissue samples.

USE IN CLINICAL TRIALS AND FUTURE PROSPECTS • Certain hurdles prevent the widespread use of tetramers in the clinical setting. • In cancer vaccination, peptide-specific T cell responses as detected by tetramers have largely not correlated with clinical outcome. • Peptide-specificity does not necessarily guarantee tumorreactivity of a T cell response – recognition efficiency of the T cells is a key factor. • One cannot simply rely on enumeration of antigen-specific T cells to predict clinical outcome in clinical trials.

• It is critical to be able to study the biology of such cells in terms of recognition efficiency, in vivo functional status and homing patterns.

1. Monitoring vaccine studies 2. Monitoring effect of Immunotherapy 3. Determination of immune status (e. g. in connection with transplantation and other immunodeficiency) 4. Monitoring T-cell responses in infectious disease, cancer, autoimmunity and other malignancies 5. Detection of antigen-specific T-cell responses in immunological research
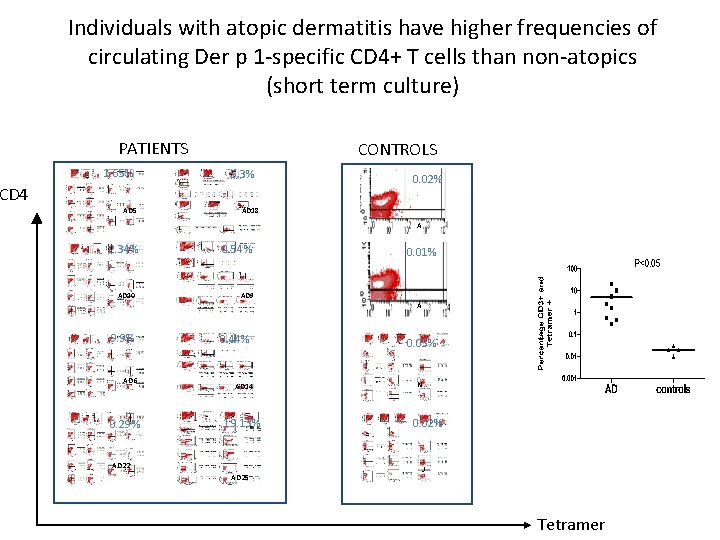
Individuals with atopic dermatitis have higher frequencies of circulating Der p 1 -specific CD 4+ T cells than non-atopics (short term culture) PATIENTS 1. 65% CONTROLS 5. 3% CD 4 AD 5 0. 02% AD 18 A 2. 34% AD 10 9. 9% AD 6 0. 29% 0. 54% AD 9 0. 44% AD 14 19. 13% AD 22 AD 25 0. 01% A 0. 03% N 0. 02% J Tetramer
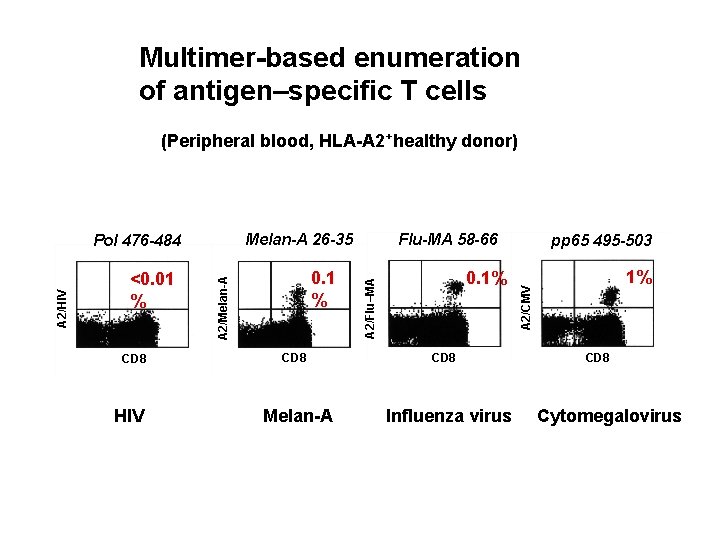
Multimer-based enumeration of antigen–specific T cells (Peripheral blood, HLA-A 2+healthy donor) CD 8 HIV % CD 8 Melan-A 0. 1% CD 8 Influenza virus pp 65 495 -503 1% 1% A 2/CMV 0. 1% 0. 1 A 2/Melan-A A 2/HIV <0. 01% <0. 01 % Flu-MA 58 -66 A 2/Flu–MA Melan-A 26 -35 Pol 476 -484 CD 8 Cytomegalovirus
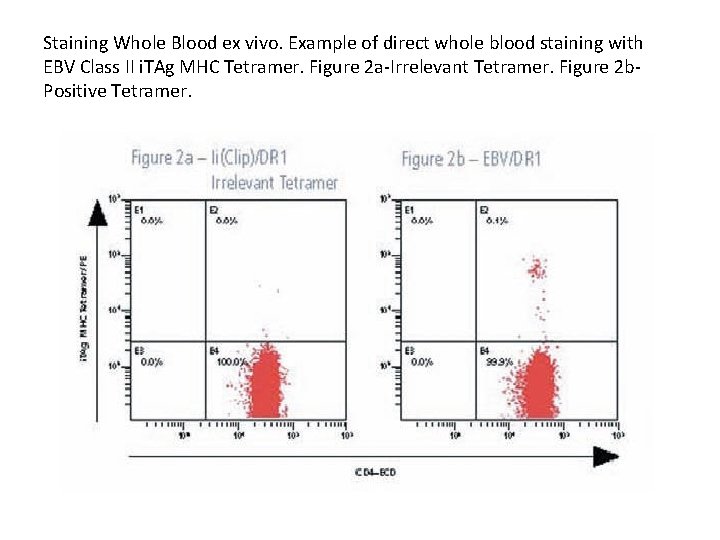
Staining Whole Blood ex vivo. Example of direct whole blood staining with EBV Class II i. TAg MHC Tetramer. Figure 2 a-Irrelevant Tetramer. Figure 2 b. Positive Tetramer.
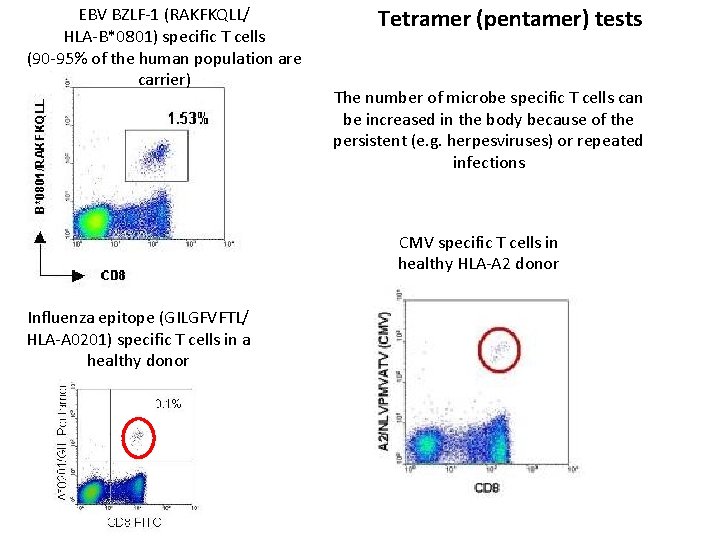
EBV BZLF-1 (RAKFKQLL/ HLA-B*0801) specific T cells (90 -95% of the human population are carrier) Tetramer (pentamer) tests The number of microbe specific T cells can be increased in the body because of the persistent (e. g. herpesviruses) or repeated infections CMV specific T cells in healthy HLA-A 2 donor Influenza epitope (GILGFVFTL/ HLA-A 0201) specific T cells in a healthy donor


Pro 5™ MHC Pentamers contain 5 MHC-peptide complexes that are multimerised by a self-assembling coiled-coil-domain. All 5 MHC-peptide complexes are held facing in the same direction, similar to a bouquet of flowers. Therefore, with Pro 5 MHC Pentamer technology, all 5 MHC-peptide complexes are available for binding to T cell receptors (TCRs), resulting in an interaction with very high avidity. Each Pro 5 MHC Pentamer also contains up to 5 fluorescent molecules yielding an improved fluorescence intensity of the complex. For optimum flexibility Pro 5® Pentamers may be ordered labeled with R-PE, APC or biotin Any combination of R-PE-labeled, APC-labeled and biotin-labeled Pentamers may be used
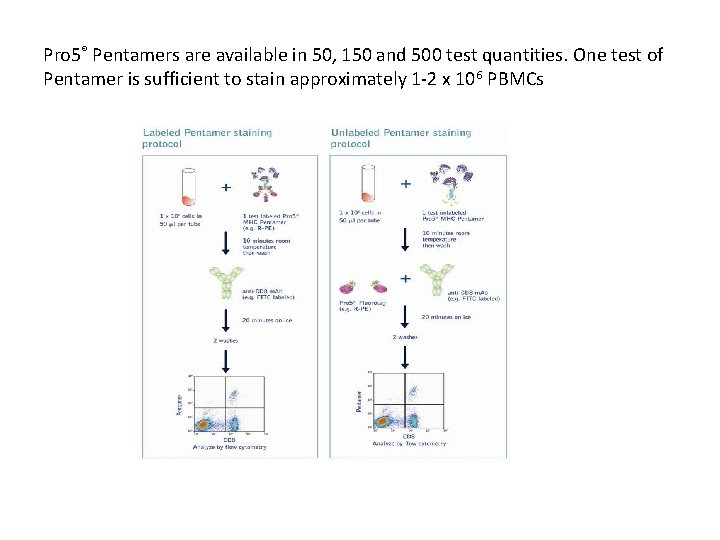
Pro 5® Pentamers are available in 50, 150 and 500 test quantities. One test of Pentamer is sufficient to stain approximately 1 -2 x 106 PBMCs

• Pro 5 MHC Pentamers feature: - Higher avidity for detecting low affinity T cells - Reduced background staining / improved specificity - Brighter overall fluorescence - Increased flexibility for long-term storage and labeling Pro 5® Pentamers can be used to detect and separate antigen-specific CD 8+ T cell populations as rare as 0. 02% of lymphocytes.
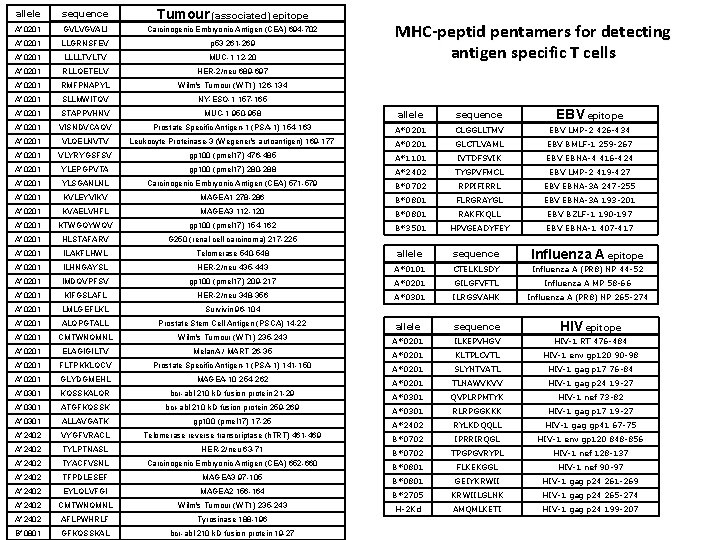
allele sequence Tumour (associated) epitope A*0201 GVLVGVALI Carcinogenic Embryonic Antigen (CEA) 694 -702 A*0201 LLGRNSFEV p 53 261 -269 A*0201 LLLLTVLTV MUC-1 12 -20 MHC-peptid pentamers for detecting antigen specific T cells A*0201 RLLQETELV HER-2/neu 689 -697 A*0201 RMFPNAPYL Wilm's Tumour (WT 1) 126 -134 A*0201 SLLMWITQV NY-ESO-1 157 -165 A*0201 STAPPVHNV MUC-1 950 -958 allele sequence A*0201 VISNDVCAQV Prostate Specific Antigen-1 (PSA-1) 154 -163 A*0201 CLGGLLTMV EBV LMP-2 426 -434 A*0201 VLQELNVTV Leukocyte Proteinase-3 (Wegener's autoantigen) 169 -177 A*0201 GLCTLVAML EBV BMLF-1 259 -267 A*0201 VLYRYGSFSV gp 100 (pmel 17) 476 -485 A*1101 IVTDFSVIK EBV EBNA-4 416 -424 A*0201 YLEPGPVTA gp 100 (pmel 17) 280 -288 A*2402 TYGPVFMCL EBV LMP-2 419 -427 A*0201 YLSGANLNL Carcinogenic Embryonic Antigen (CEA) 571 -579 B*0702 RPPIFIRRL EBV EBNA-3 A 247 -255 A*0201 KVLEYVIKV MAGEA 1 278 -286 B*0801 FLRGRAYGL EBV EBNA-3 A 193 -201 A*0201 KVAELVHFL MAGEA 3 112 -120 B*0801 RAKFKQLL EBV BZLF-1 190 -197 A*0201 KTWGQYWQV gp 100 (pmel 17) 154 -162 B*3501 HPVGEADYFEY EBV EBNA-1 407 -417 A*0201 HLSTAFARV G 250 (renal cell carcinoma) 217 -225 A*0201 ILAKFLHWL Telomerase 540 -548 allele sequence Influenza A epitope A*0201 ILHNGAYSL HER-2/neu 435 -443 A*0101 CTELKLSDY Influenza A (PR 8) NP 44 -52 A*0201 IMDQVPFSV gp 100 (pmel 17) 209 -217 A*0201 GILGFVFTL Influenza A MP 58 -66 A*0201 KIFGSLAFL HER-2/neu 348 -356 A*0301 ILRGSVAHK Influenza A (PR 8) NP 265 -274 A*0201 LMLGEFLKL Survivin 96 -104 A*0201 ALQPGTALL Prostate Stem Cell Antigen (PSCA) 14 -22 allele sequence A*0201 CMTWNQMNL Wilm's Tumour (WT 1) 235 -243 A*0201 ILKEPVHGV HIV-1 RT 476 -484 A*0201 ELAGIGILTV Melan. A / MART 26 -35 A*0201 KLTPLCVTL HIV-1 env gp 120 90 -98 A*0201 FLTPKKLQCV Prostate Specific Antigen-1 (PSA-1) 141 -150 A*0201 SLYNTVATL HIV-1 gag p 17 76 -84 A*0201 GLYDGMEHL MAGEA-10 254 -262 A*0201 TLNAWVKVV HIV-1 gag p 24 19 -27 A*0301 KQSSKALQR bcr-abl 210 k. D fusion protein 21 -29 A*0301 QVPLRPMTYK HIV-1 nef 73 -82 A*0301 ATGFKQSSK bcr-abl 210 k. D fusion protein 259 -269 A*0301 RLRPGGKKK HIV-1 gag p 17 19 -27 A*0301 ALLAVGATK gp 100 (pmel 17) 17 -25 A*2402 RYLKDQQLL HIV-1 gag gp 41 67 -75 A*2402 VYGFVRACL Telomerase reverse transcriptase (h. TRT) 461 -469 B*0702 IPRRIRQGL HIV-1 env gp 120 848 -856 A*2402 TYLPTNASL HER-2/neu 63 -71 B*0702 TPGPGVRYPL HIV-1 nef 128 -137 A*2402 TYACFVSNL Carcinogenic Embryonic Antigen (CEA) 652 -660 B*0801 FLKEKGGL HIV-1 nef 90 -97 A*2402 TFPDLESEF MAGEA 3 97 -105 B*0801 GEIYKRWII HIV-1 gag p 24 261 -269 A*2402 EYLQLVFGI MAGEA 2 156 -164 B*2705 KRWIILGLNK HIV-1 gag p 24 265 -274 A*2402 CMTWNQMNL Wilm's Tumour (WT 1) 235 -243 H-2 Kd AMQMLKETI HIV-1 gag p 24 199 -207 A*2402 AFLPWHRLF Tyrosinase 188 -196 B*0801 GFKQSSKAL bcr-abl 210 k. D fusion protein 19 -27 EBV epitope HIV epitope


Immudex Dextramer™ Technology • The MHC Dextramer™ reagents are a new generation of fluorescent MHC multimers that are particularly efficient in the detection of T-cells carrying T-cell receptors with very low affinity for MHC. • The Dextramer™ reagents carry more MHC-molecules and more fluorochromes than conventional MHC multimers • MHC Dextramer™ reagents thus provide the following benefits: Brighter staining Higher resolution Lower background Higher stability Minimal lot-to-lot variation

• • The MHC Dextramer™ consists of a dextran polymer backbone carrying an optimized number of MHC and fluorochrome molecules. MHC Dextramer™ reagents carry more MHC molecules and more fluorochromes than conventional MHC multimers. This increases their avidity for the specific Tcell and enhances their staining intensity, thereby increasing resolution and the signal-to-noise ratio. An optimized formulation of the MHC Dextramer™ products ensures minimal background staining. The dextran polymer backbone stabilizes the conformation of the attached proteins, the MHC-peptide complexes and fluorochromes. Thus, MHC Dextramer™ reagents are highly stable reagents. MHC Dextramers are suitable for basic and clinical research in a number of applications including drug and vaccine development.

• The MHC Dextramer™ reagents can be used to detect antigen-specific Tcells in fluid cell samples (e. g. blood, cultured cell lines, CSF, lymph, synovial fluid) using flow cytometry, or antigen-specific T-cells may be detected in situ in tissue sections.
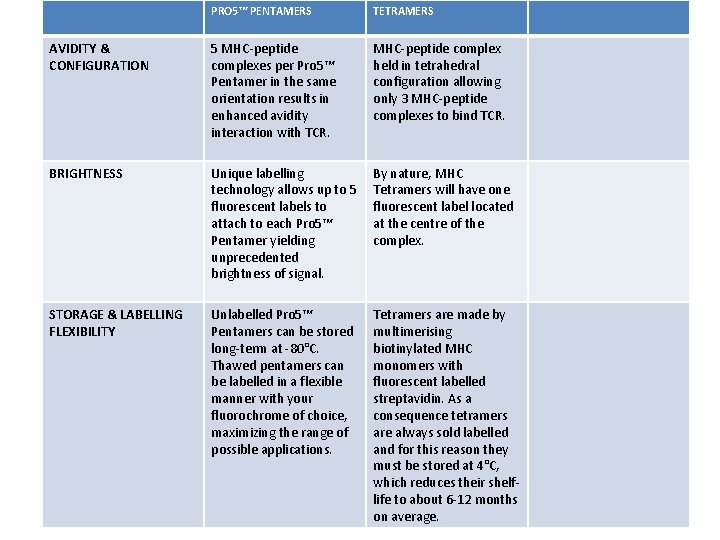
PRO 5™ PENTAMERS TETRAMERS AVIDITY & CONFIGURATION 5 MHC-peptide complexes per Pro 5™ Pentamer in the same orientation results in enhanced avidity interaction with TCR. MHC-peptide complex held in tetrahedral configuration allowing only 3 MHC-peptide complexes to bind TCR. BRIGHTNESS Unique labelling technology allows up to 5 fluorescent labels to attach to each Pro 5™ Pentamer yielding unprecedented brightness of signal. By nature, MHC Tetramers will have one fluorescent label located at the centre of the complex. STORAGE & LABELLING FLEXIBILITY Unlabelled Pro 5™ Pentamers can be stored long-term at -80°C. Thawed pentamers can be labelled in a flexible manner with your fluorochrome of choice, maximizing the range of possible applications. Tetramers are made by multimerising biotinylated MHC monomers with fluorescent labelled streptavidin. As a consequence tetramers are always sold labelled and for this reason they must be stored at 4°C, which reduces their shelflife to about 6 -12 months on average.



- Slides: 53