Essential Training Package for HPV Vaccine Introduction Module











- Slides: 12

Essential Training Package for HPV Vaccine Introduction Module 3 HPV vaccine eligibility and contraindications Cervarix. TM

Learning objectives l At the end of the module, you will have learned to: – Describe the recommended immunization schedule for Cervarix. TM – Describe when a girl is eligible for HPV vaccine and when she is not eligible – Describe the absolute contraindications for vaccination l Duration – 60’ 2 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

Key issues What is the schedule and dosage 1 for Cervarix. TM? 2 How do you determine a girl’s eligibility for HPV vaccine? 3 What to do when…? 4 What are the precautions and contraindications? 3 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

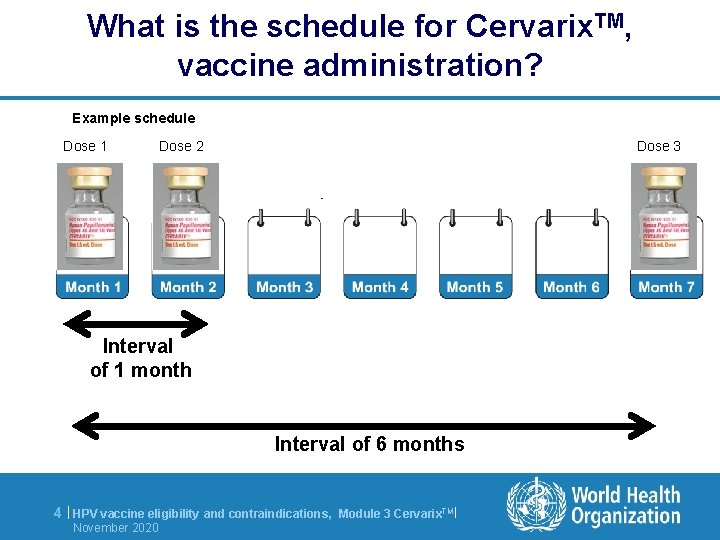
What is the schedule for Cervarix. TM, vaccine administration? Example schedule Dose 1 Dose 2 Dose 3 Interval of 1 month Interval of 6 months 4 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

Who should get HPV vaccine? 5 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

Are there contraindications to HPV vaccination? l HPV vaccine is not recommended for: SEVERE FEBRILE ILLNESS A minor infection such as a cold is not a contraindication 6 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

What should you do in this scenario? The girl is pregnant when she returns for third dose of HPV vaccine. 7 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

What should you do in this scenario? The girl is immunocompromised due to HIV/AIDS. 8 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

What should you do in this scenario? The girl came in to an immunization session. She is 4 months late for the second dose. 9 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

Key messages l Cervarix. TM is a 3 dose schedule, administered within 6 months - between 1 st and 2 nd dose: interval of 1 month - between 1 st and 3 rd dose: interval of 6 months l Girls between 9 and 13 years old are eligible for HPV vaccination l Contrindications for HPV vaccination are: - Allergy after a previous vaccine dose, or to a component of the vaccine - Pregnancy - Severe febrile illness 10 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

End of module Thank you for your attention! 11 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|

References l Comprehensive Cervical Cancer Control. A guide to essential practice (C 4 GEP). WHO 2013 l HPV vaccine introduction into national immunization programmes: Information for programme managers and health workers. WHO 2013 l Cervarix. TM package inserts. Glaxo. Smith. Kline 2009 12 | HPV vaccine eligibility and contraindications, November 2020 Module 3 Cervarix. TM|