Getting to the Point Strategies to Prevent Zoster




















- Slides: 29

Getting to the Point: Strategies to Prevent Zoster Kenneth Mc. Call, Pharm. D, BCGP, RPh, FAPh. A Associate Professor

Zoster Prevention - Objectives • Discuss the presentation and complications of herpes zoster • Recognize the ACIP recommendations for zoster vaccines • Describe the differences between the GSK zoster vaccine and the Merck zoster vaccine in terms of FDA indication, storage, preparation, administration, dosing, and contraindications

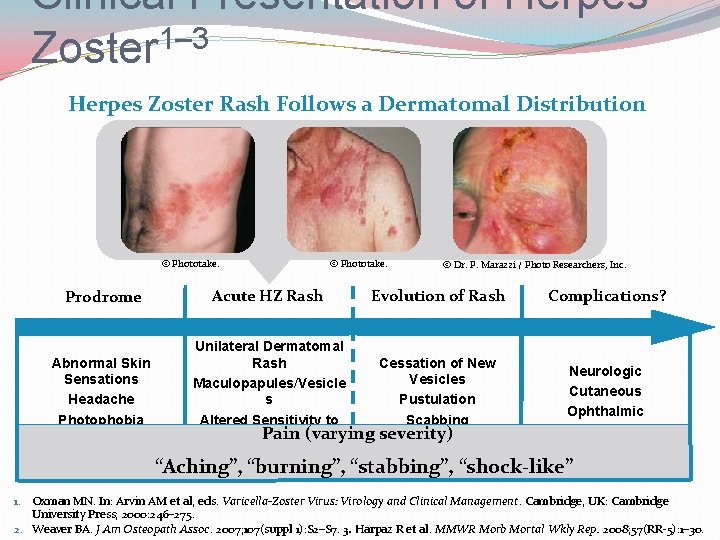
Clinical Presentation of Herpes Zoster 1– 3 Herpes Zoster Rash Follows a Dermatomal Distribution © Phototake. Prodrome Abnormal Skin Sensations Headache Photophobia Malaise Acute HZ Rash © Phototake. © Dr. P. Marazzi / Photo Researchers, Inc. Evolution of Rash Unilateral Dermatomal Rash Cessation of New Vesicles Maculopapules/Vesicle s Pustulation Altered Sensitivity to Scabbing Pain (varying severity) Touch Cutaneous Healing Unbearable Itching Complications? Neurologic Cutaneous Ophthalmic Visceral (rare) “Aching”, “burning”, “stabbing”, “shock-like” 1. Oxman MN. In: Arvin AM et al, eds. Varicella-Zoster Virus: Virology and Clinical Management. Cambridge, UK: Cambridge University Press; 2000: 246– 275. 2. Weaver BA. J Am Osteopath Assoc. 2007; 107(suppl 1): S 2–S 7. 3. Harpaz R et al. MMWR Morb Mortal Wkly Rep. 2008; 57(RR-5): 1– 30.


Herpes Zoster is the reactivation of which virus? 1. 2. 3. 4. Measles Mumps Rubella Varicella

What is the most common long-term complication of herpes zoster? 1. 2. 3. 4. Bell’s palsy Postherpetic neuralgia Vision loss Myocarditis

Prevention of Zoster and Postherpetic Neuralgia ZOSTAVAX® (Zoster Vaccine Live) 7

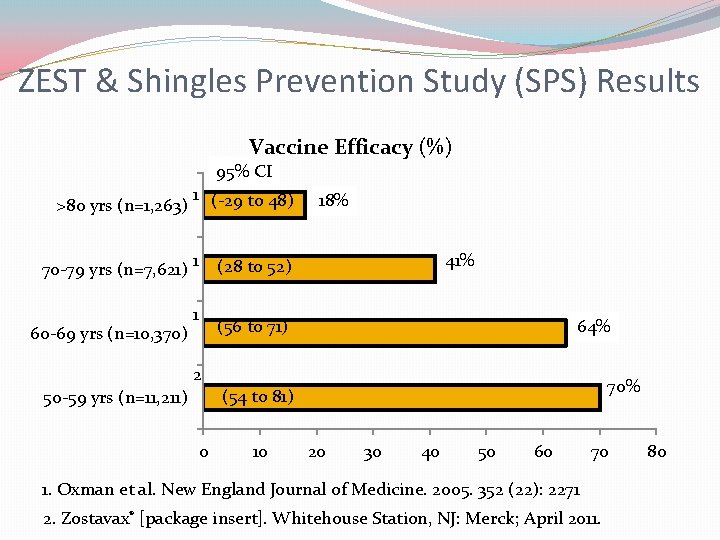
ZEST & Shingles Prevention Study (SPS) Results Vaccine Efficacy (%) 95% CI >80 yrs (n=1, 263) 1 (-29 to 48) 70 -79 yrs (n=7, 621) 1 60 -69 yrs (n=10, 370) 1 2 50 -59 yrs (n=11, 211) 0 18% 41% (28 to 52) (56 to 71) 64% 70% (54 to 81) 10 20 30 40 50 60 70 1. Oxman et al. New England Journal of Medicine. 2005. 352 (22): 2271 2. Zostavax® [package insert]. Whitehouse Station, NJ: Merck; April 2011. 80

ZOSTAVAX® Indication �ACIP recommends routine vaccination of all persons aged >60 years with 1 dose of zoster vaccine. � NEW FDA LABELING: “ZOSTAVAX is a live attenuated virus vaccine indicated for prevention of herpes zoster (shingles) in individuals 50 years of age and older. ” �Persons who report a previous episode of zoster and persons with chronic medical conditions can be vaccinated unless those conditions are contraindications or precautions. �Zoster vaccination is not indicated to treat acute zoster. Zostavax® [package insert]. Whitehouse Station, NJ: Merck; April 2011. Recommendations of the Advisory Committee on Immunization Practices (ACIP) http: //www. cdc. gov/mmwr/preview/mmwrhtml/rr 5705 a 1. htm? s_cid=rr 5705 a 1_e 9

ZOSTAVAX® Contraindications �Allergy to neomycin or any vaccine component �Pregnancy �Immunocompromised status �AIDS or other clinical manifestations of HIV, including persons with CD 4+ T-lymphocyte values <200 per mm 3 �malignant neoplasms affecting the bone marrow �chemotherapy or radiation within the last 3 months �Persons on immunosuppressive therapy, including high -dose corticosteroids (>20 mg/day of prednisone or equivalent) lasting two or more weeks 10

ZOSTAVAX® Storage and Handling �Zoster vaccine must be stored frozen �The vaccine must be discarded if not used within 30 minutes after reconstitution. �New labeling: Zostavax may be stored and/or transported at fridge temp for up to 72 hours prior to reconstitution. Any unused vaccine at fridge temp should be discarded. Zostavax® [package insert]. Whitehouse Station, NJ: Merck; April 2011. 11

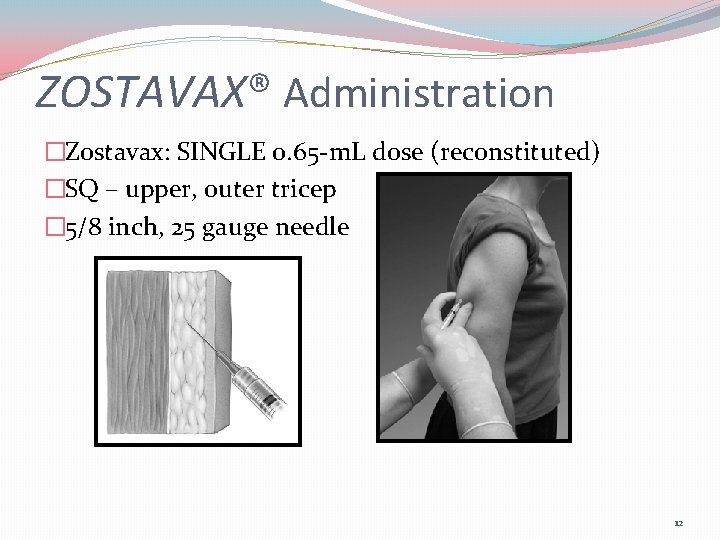
ZOSTAVAX® Administration �Zostavax: SINGLE 0. 65 -m. L dose (reconstituted) �SQ – upper, outer tricep � 5/8 inch, 25 gauge needle 12

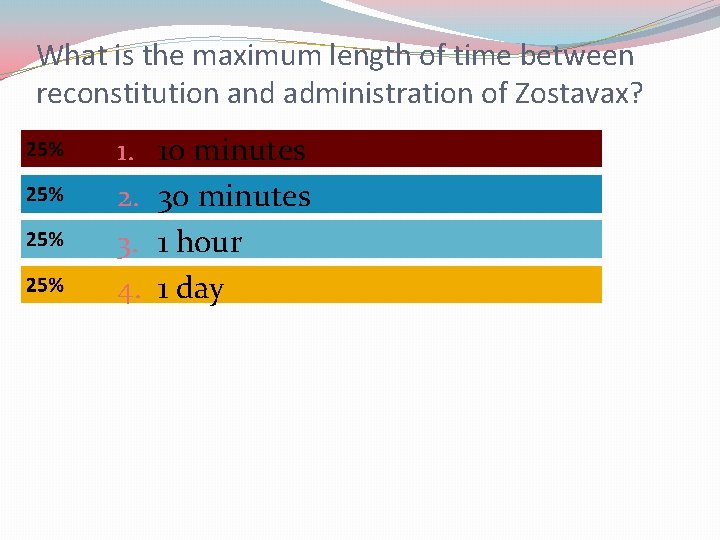
What is the maximum length of time between reconstitution and administration of Zostavax? 1. 2. 3. 4. 10 minutes 30 minutes 1 hour 1 day

Prevention of Zoster and Postherpetic Neuralgia Shingrix® (Herpes Zoster Adjuvanted Subunit Vaccine) 14

Now Approved: Shingrix® �Inactivated, recombinant zoster subunit vaccine �Manufactured by Glaxo. Smith. Kline


Shingrix® Storage and Preparation �Stored in the refrigerator at 4 o C �Suspension that must be reconstituted (o. 5 ml)* *For use within 6 hours

Shingrix® (Herpes Zoster Adjuvanted Subunit Vaccine)

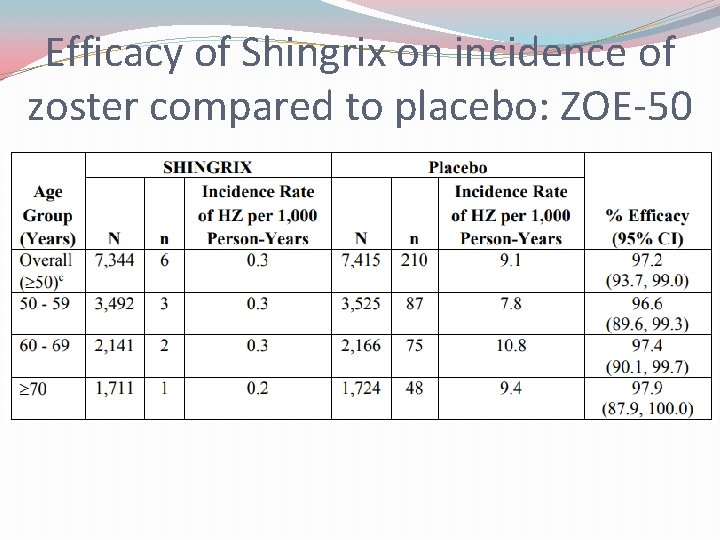
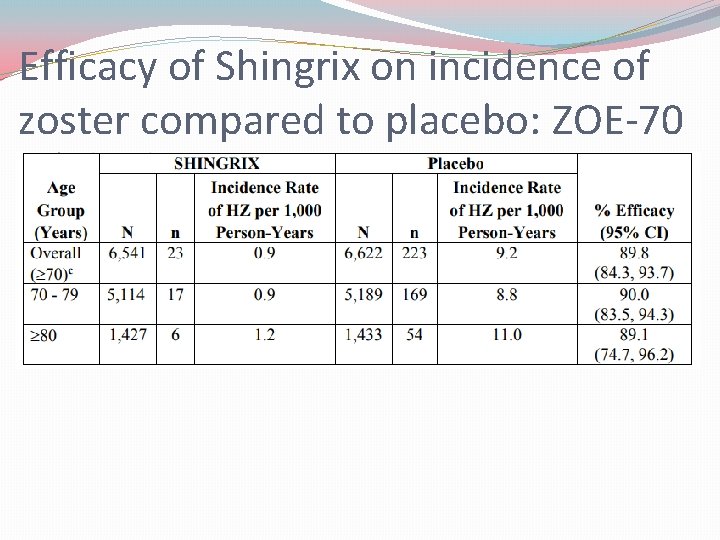
Shingrix® Trials �Trials: �ZOE-50: Shingrix reduced risk of herpes zoster by 97. 2% in patients 50 -59 year olds � (Zostavax had only 70% efficacy for the same population group) �ZOE-70: Shingrix reduced risk of herpes zoster by 90% in patients ≥ 70 years old � (Zostavax had only 38% efficacy for the same population group) �Zoster-048: Shingrix demonstrates appropriate immunogeneicity and safety data in all approved populations regardless of if the patient has received a previous Zostavax vaccine greater than 5 years earlier

Efficacy of Shingrix on incidence of zoster compared to placebo: ZOE-50

Efficacy of Shingrix on incidence of zoster compared to placebo: ZOE-70

Recommendations of the ACIP for Use of Herpes Zoster Vaccines �Recombinant zoster vaccine (RZV) is recommended for the prevention of herpes zoster and related complications for immunocompetent adults aged ≥ 50 years. �Whereas RZV is licensed for all persons aged ≥ 50 years, immunocompromised persons and those on moderate to high doses of immunosuppressive therapy were excluded from the efficacy studies (ZOE 50 and ZOE-70), and thus, ACIP has not made recommendations regarding the use of RZV in these patients. https: //www. cdc. gov/mmwr/volumes/67/wr/mm 6703 a 5. htm? s_cid=mm 6703 a 5_w

Recommendations of the ACIP for Use of Herpes Zoster Vaccines �RZV is recommended for the prevention of herpes zoster and related complications for immunocompetent adults who previously received zoster vaccine live (ZVL). �Based on expert opinion, RZV should not be given <2 months after receipt of ZVL. MMWR / January 26, 2018 / 67(3); 103– 108 https: //www. cdc. gov/mmwr/volumes/67/wr/mm 6703 a 5. htm? s_cid=mm 6703 a 5_w

Recommendations of the ACIP for Use of Herpes Zoster Vaccines �RZV may be used in adults aged ≥ 50 years, irrespective of prior receipt of varicella vaccine or ZVL. �RZV is preferred over ZVL for the prevention of herpes zoster and related complications. MMWR / January 26, 2018 / 67(3); 103– 108 https: //www. cdc. gov/mmwr/volumes/67/wr/mm 6703 a 5. htm? s_cid=mm 6703 a 5_w

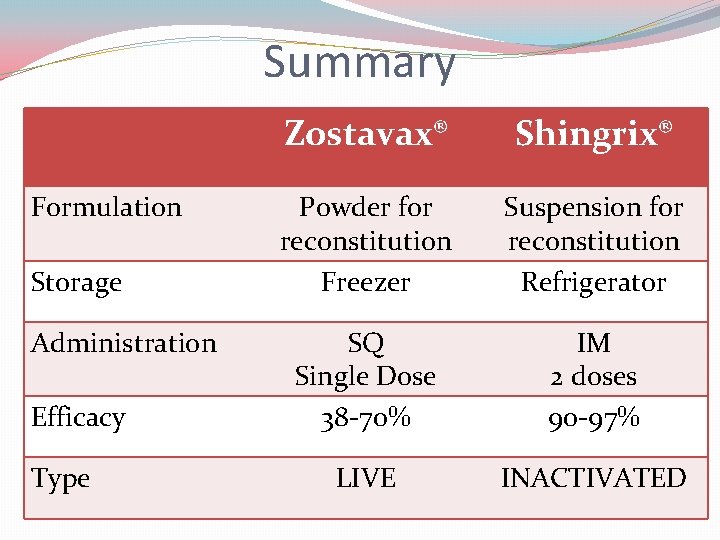
Summary Formulation Storage Administration Efficacy Type Zostavax® Shingrix® Powder for reconstitution Freezer Suspension for reconstitution Refrigerator SQ Single Dose 38 -70% IM 2 doses 90 -97% LIVE INACTIVATED

What is the maximum length of time between reconstitution and administration of Shingrix? 1. 2. 3. 4. 10 minutes 30 minutes 1 hour 6 hours

A 55 yo woman requests a zoster vaccine. Select the best option to prevent shingles as recommended by ACIP. A. B. C. D. Shingrix SQ x 1 dose Shingrix IM x 2 doses Zostavax SQ x 1 dose Zostavax IM x 2 doses

A 72 yo man previously received Zostavax 7 years ago. Despite being vaccinated, he experienced a shingles attack in January 2018. Select the best option to prevent shingles. A. B. C. D. Shingrix SQ x 1 dose Shingrix IM x 2 doses Zostavax booster SQ x 1 dose Both Shingrix and Zostavax are contraindicated

Questions and Discussion kmccall@une. edu