Zoster Vaccine A New Vaccine for Preventing Herpes




























- Slides: 39

Zoster Vaccine: A New Vaccine for Preventing Herpes Zoster and Post-herpetic Neuralgia Jane Seward, MBBS, MPH Division Viral Diseases (proposed) National Center Immunization & Respiratory Diseases (proposed) Centers for Disease Control and Prevention 41 st National Immunization Conference Kansas City, March 5 th, 2007

Acknowledgements • Centers for Disease Control and Prevention • Food and Drug Administration • Advisory Committee on Immunization Practices (ACIP) – Herpes Zoster ACIP working group • Drs Michael Oxman and Ken Schmader

Outline • Herpes zoster – Clinical manifestations and complications – Epidemiology • Zoster vaccine – Efficacy and safety – ACIP vaccine policy recommendations • Vaccine program implementation and surveillance

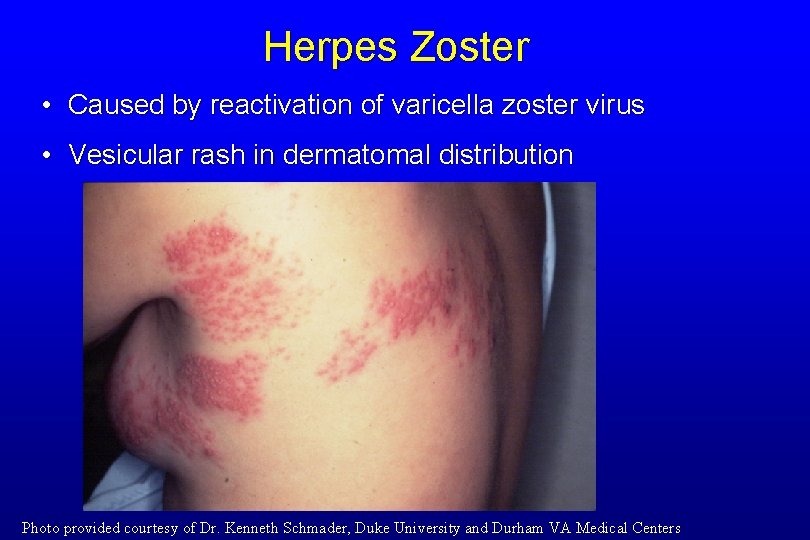
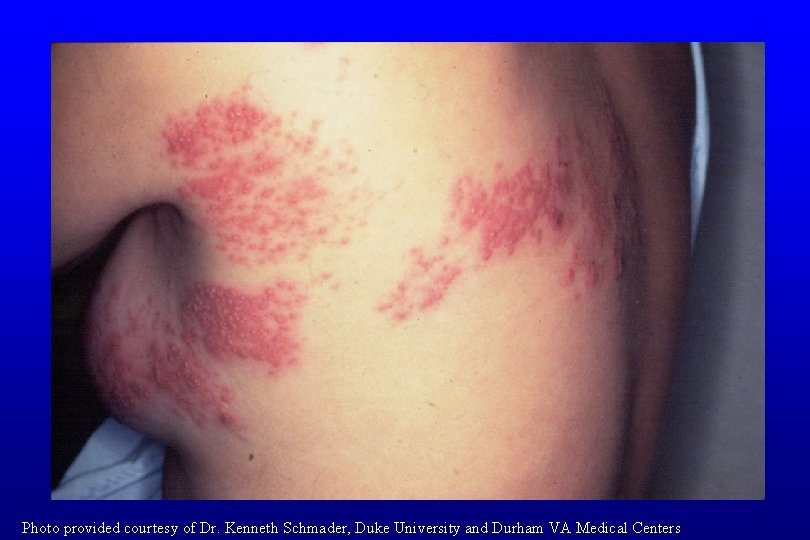
Herpes Zoster • Caused by reactivation of varicella zoster virus • Vesicular rash in dermatomal distribution Photo provided courtesy of Dr. Kenneth Schmader, Duke University and Durham VA Medical Centers


Herpes Zoster Epidemiology • Incidence – – ~ 3 -4 per 1, 000 person years May have been increasing before availability of varicella and zoster vaccines • 750, 000 to 1 million cases in U. S. annually • Lifetime risk – ~ 20% to 30% – 50% of individuals living until 85 years of age Gnann J et al. N Engl J Med. 2002; Katz J et al. Clin Infect Dis. 2004; Ragozzino M et al. Medicine 1982.

Herpes Zoster Epidemiology • Main risk factors – Age – Cellular immune deficiencies • Other risk factors – Race – Sex – Stress – Trauma

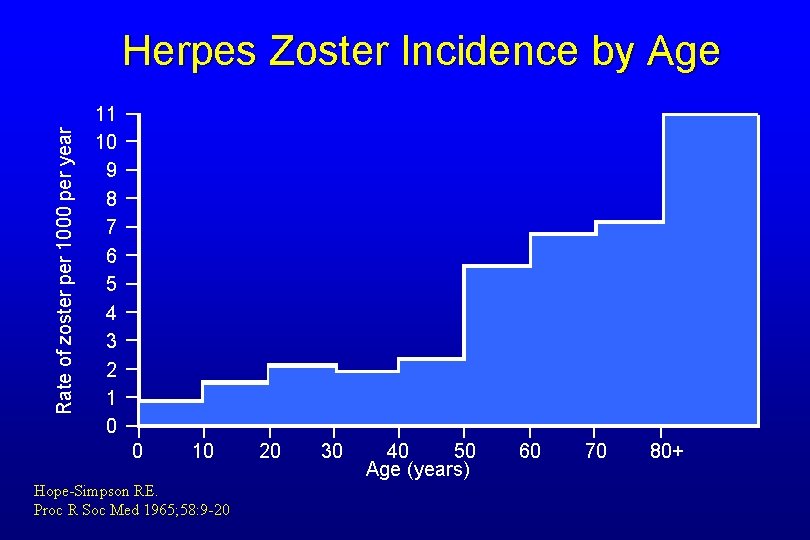
Rate of zoster per 1000 per year Herpes Zoster Incidence by Age 11 10 9 8 7 6 5 4 3 2 1 0 0 10 Hope-Simpson RE. Proc R Soc Med 1965; 58: 9 -20 20 30 40 50 Age (years) 60 70 80+

Complications Common Uncommon • • • Acute neuralgia Postherpetic neuralgia Ocular complications of herpes zoster ophthalmicus • Bacterial superinfection Dissemination Pneumonitis Hepatitis Encephalitis/meningitis Herpetic gangrenosum Motor neuropathies Myelitis Hemiparesis

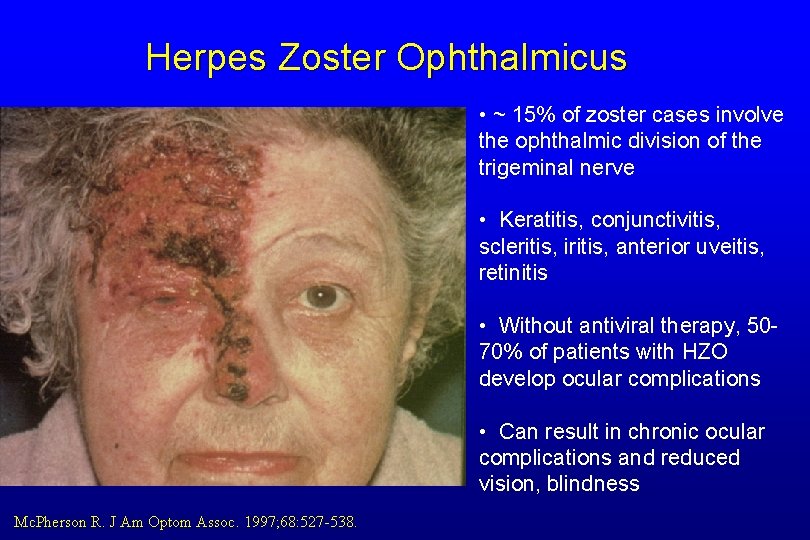
Herpes Zoster Ophthalmicus • ~ 15% of zoster cases involve the ophthalmic division of the trigeminal nerve • Keratitis, conjunctivitis, scleritis, iritis, anterior uveitis, retinitis • Without antiviral therapy, 5070% of patients with HZO develop ocular complications • Can result in chronic ocular complications and reduced vision, blindness Mc. Pherson R. J Am Optom Assoc. 1997; 68: 527 -538.

Herpes Zoster Pain • Can be excruciating (e. g. , like renal colic, childbirth) • Described as aching, burning, stabbing, shock-like • Continuous or paroxysmal • Often associated with: § Altered or painful sensitivity to touch (paresthesia, dysesthesia) § Provoked by trivial stimuli like bed sheets or breeze (allodynia) § Exaggerated, prolonged response to pain (hyperesthesia) § Unbearable itching

Post Herpetic Neuralgia (PHN) • Pain persisting after herpes zoster rash resolves • Variable definitions by time since rash onset and pain intensity • Estimates from antiviral trials (placebo recipients) – – 30 days 60 days 90 days 180 days 68% 60% 55% 40% Cunningham AL, Dworkin RH. BMJ. 2000; 321: 778 -779; Dworkin RH, Portenoy RK. Lancet. 1994; 343: 1648 Arani RB, et al. Stat Med 2001; 20: 2429 -2439; Desmond RA, et al. J Pain Symptom Manage 2002; 23: 510 -516

Risk Factors for Postherpetic Neuralgia • • • Age Severity of acute pain Severity of acute rash Painful prodrome Female sex Jung BF. Neurology. 2004; 62: 1545 -1551.

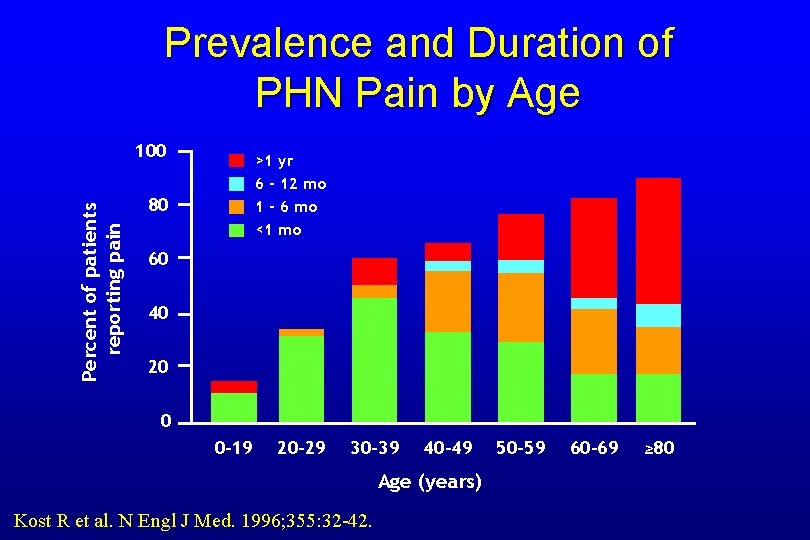
Prevalence and Duration of PHN Pain by Age Percent of patients reporting pain 100 >1 yr 6 - 12 mo 1 - 6 mo <1 mo 80 60 40 20 0 0 -19 20 -29 30 -39 40 -49 Age (years) Kost R et al. N Engl J Med. 1996; 355: 32 -42. 50 -59 60 -69 ≥ 80

Acute and Post Herpetic Neuralgia Impact on Quality of Life Comparable to congestive heart failure, diabetes, and depression • • • Physical Chronic fatigue Anorexia & weight loss Physical inactivity Insomnia Social Fewer social gatherings Change in social role Schmader KE. Clin Infect Dis 2001; 32(10): 1481 -6 Psychological • • • Anxiety Difficulty concentrating Depression, suicidal ideation Functional • Interferes with activities of daily living: dressing, bathing, eating, travel, cooking, shopping

Zoster Vaccine • ZOSTAVAX®, Merck and Co. , Inc • Licensed by FDA in May 2006 • Live, attenuated Oka/Merck VZV vaccine • Minimum dose 19, 400 PFU at expiration [varicella vaccine 1, 350 PFU at expiration] [varicella vaccine

Shingles Prevention Study • • • Randomized, double blind placebo-controlled trial 38, 546 adults ≥ 60 years at 22 study sites Study end points – Primary: Burden of illness (Sum of HZ severity of illness scores ~ pain level and duration) – Secondary: Post herpetic neuralgia (PHN) and HZ Incidence • Prospective, active follow up median 3. 1 years • 94% cases laboratory confirmed Oxman M et al. N Engl J Med. 2005; 352: 2271 -2284.

Shingles Prevention Study Enrollees 38, 546 (59% male) Zoster vaccine 19, 270 Placebo 19, 276 Completed study 18, 359 (95. 3%) 315 HZ cases Completed study 18, 357 (95. 2%) 642 HZ cases 793 (4. 1%) died 57 (0. 3%) withdrew 61 (0. 3%) lost follow up Oxman M et al. N Engl J Med. 2005; 352: 2271 -2284. 792 (4. 1%) died 75 (0. 4%) withdrew 52 (0. 2%) lost follow up

Results

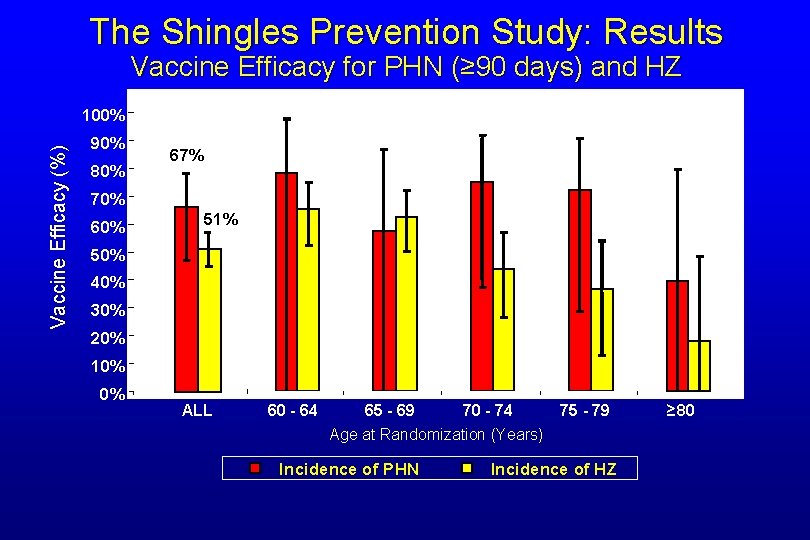
The Shingles Prevention Study: Results Vaccine Efficacy for PHN (≥ 90 days) and HZ Vaccine Efficacy (%) 100% 90% 80% 67% 70% 60% 51% 50% 40% 30% 20% 10% 0% ALL 60 - 64 65 - 69 70 - 74 75 - 79 Age at Randomization (Years) Incidence of PHN Incidence of HZ ≥ 80

Shingles Prevention Study: Results Adverse Events Appears to be safe: • No pattern suggesting a causal link to serious adverse events • No cases of HZ caused by vaccine virus • Excess of mild reactions in vaccine recipients: Zoster Vaccine N=3345 Local injection-site reactions*: *Erythema, pain, swelling, pruritis, warmth 48% Placebo N=3271 17%

FDA Licensure: May 2006 • Indicated for prevention of herpes zoster in persons ≥ 60 years • Not indicated for the treatment of herpes zoster or PHN • Contraindications: § History of anaphylaxis to vaccine components § Primary or acquired immunodeficiency states § Immunosuppressive therapy

Zoster Vaccine Policy Recommendations Advisory Committee Immunization Practices • Recommended for adults 60 years of age and older whether or not they report a prior episode of herpes zoster • Persons with chronic medical conditions may be vaccinated unless a contraindication or precaution exists for their condition • Provisional recommendations available at http: //www. cdc. gov/nip/recs/provisional_recs/zoster-11 -20 -06. pdf

Vaccine Cost and Re-imbursement • • • Wholesale acquisition price = $152 Average wholesale price - $190. 60 CDC contract price = $107. 90 • Covered under Medicare part D • Will be covered under prevention services by private insurers • Merck financial assistance program

Vaccine Storage and Handling • Lyophilized preparation stored frozen at -15º C (+5ºF) or colder • Cannot be stored in refrigerator for any length of time • Reconstituted with diluent provided • Discard if not administered within 30 minutes

Monitoring Implementation and Impact of Vaccine Program

Post Licensure Surveillance Vaccine • Coverage - considering options for using – National data sources e. g. NHIS, Medicare – Modifying NIS [NAIS] • Safety – challenges with age group and co-existing medical conditions – VAERS – VSD rapid cycle analysis – Laboratory testing and VZV strain identification (national. VZVlab@cdc. gov)

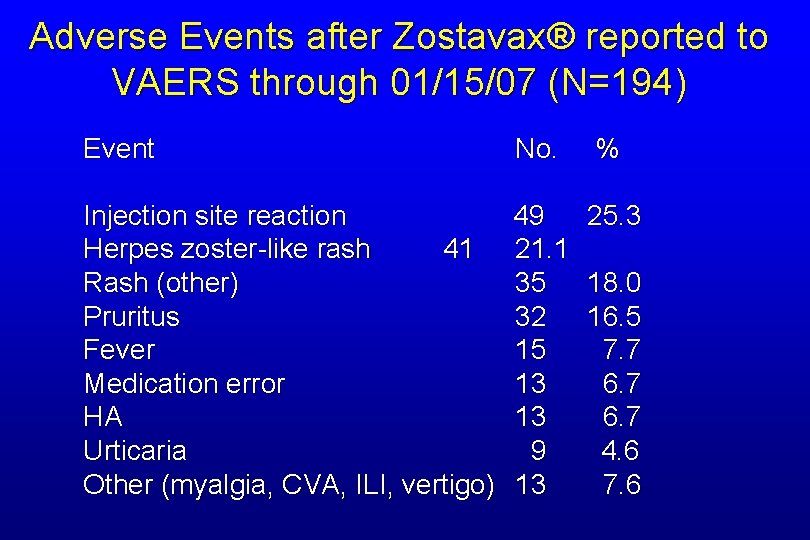
Post Licensure Safety Surveillance Reporting to VAERS through 1/15/07 • 194 reports, 2 serious • Median age = 65 years • Median interval from vaccination to symptom onset = 1 day

Adverse Events after Zostavax® reported to VAERS through 01/15/07 (N=194) Event No. % Injection site reaction 49 25. 3 Herpes zoster-like rash 41 21. 1 Rash (other) 35 18. 0 Pruritus 32 16. 5 Fever 15 7. 7 Medication error 13 6. 7 HA 13 6. 7 Urticaria 9 4. 6 Other (myalgia, CVA, ILI, vertigo) 13 7. 6

Post Licensure Surveillance Disease and Vaccine Effectiveness Herpes Zoster +/- PHN • National data sources e. g. NHIS, Medicare, Med. Stat • Varicella and HZ active surveillance sites Vaccine effectiveness in preventing HZ, PHN • National data sources – Medicare • Active surveillance sites: case control study

Summary • Herpes zoster causes considerable morbidity in elderly persons • Herpes zoster and its complications may now be prevented or modified by vaccination • Persons ≥ 60 years (and their health care providers) should be educated about HZ and offered vaccine

Thank You

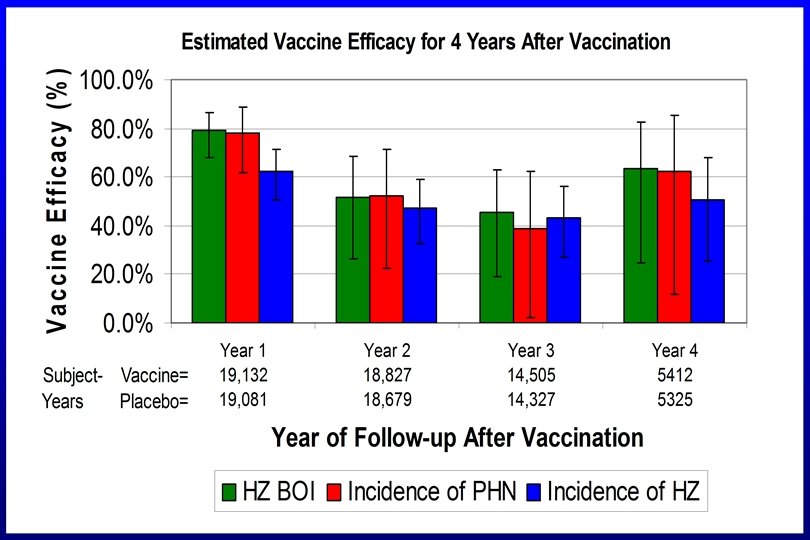
Duration of Protection?


Zoster Vaccine: Safety Study • No cases of herpes zoster caused by vaccine virus among 919 PCR-confirmed cases tested • Vaccine did not induce herpes zoster – During the 30 days postvaccination • Placebo group → 18 cases • Vaccine group → 6 cases Oxman M et al. N Engl J Med. 2005; 352: 2271 -2284

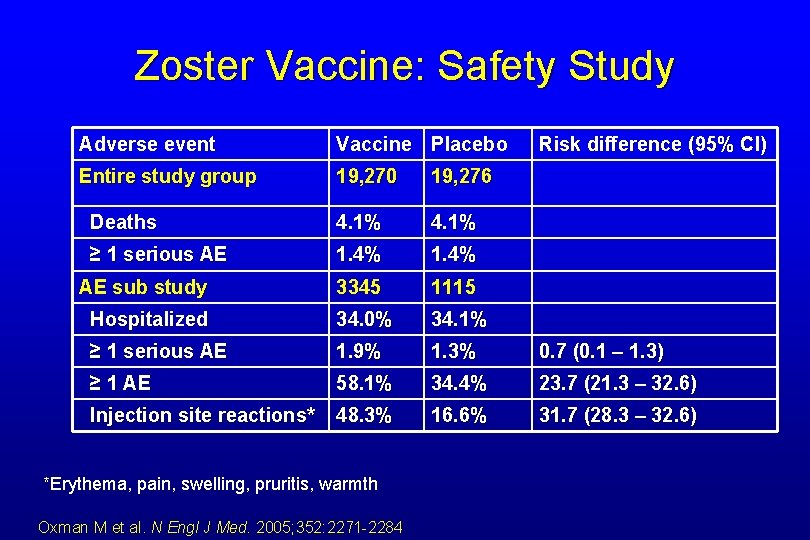
Zoster Vaccine: Safety Study Adverse event Vaccine Placebo Entire study group 19, 270 19, 276 Deaths 4. 1% ≥ 1 serious AE 1. 4% 3345 1115 Hospitalized 34. 0% 34. 1% ≥ 1 serious AE 1. 9% 1. 3% 0. 7 (0. 1 – 1. 3) ≥ 1 AE 58. 1% 34. 4% 23. 7 (21. 3 – 32. 6) Injection site reactions* 48. 3% 16. 6% 31. 7 (28. 3 – 32. 6) AE sub study *Erythema, pain, swelling, pruritis, warmth Oxman M et al. N Engl J Med. 2005; 352: 2271 -2284 Risk difference (95% CI)

The Shingles Prevention Study: Results Vaccine Efficacy against PHN of Varying Duration PHN Defined by Varying Duration (days) Vaccine Efficacy VEPHN (95% CI) 30 60 90 120 180 59% (47, 69) 60% (44, 73) 67% (48, 79) 69% (45, 83) 73% (42, 89)

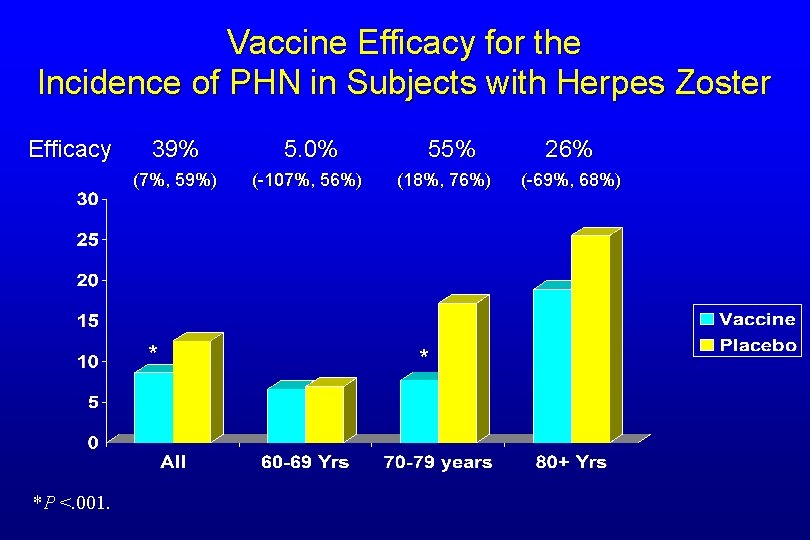
Vaccine Efficacy for the Incidence of PHN in Subjects with Herpes Zoster Efficacy 39% 5. 0% 55% 26% (7%, 59%) (-107%, 56%) (18%, 76%) (-69%, 68%) * *P <. 001. *

Photo provided courtesy of Dr. Kenneth Schmader, Duke University and Durham VA Medical Centers