Herpes Zoster subunit vaccine A Better Shot at

Herpes Zoster subunit vaccine: A Better Shot at Preventing Shingles Kaitlyn Rivard, Pharm. D, BCPS, AAHIVP June 1, 2018

Learning Objectives • Compare and contrast the herpes zoster subunit vaccine and the herpes zoster live vaccine • Summarize ACIP recommendations for herpes zoster immunization • Recommend herpes zoster vaccination for at-risk patients ACIP: Advisory Committee on Immunization Practices
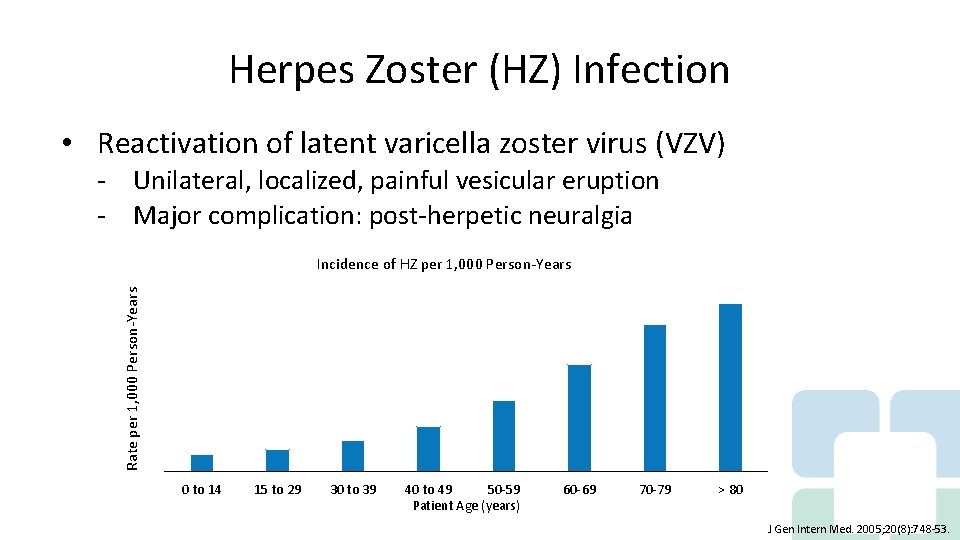
Herpes Zoster (HZ) Infection • Reactivation of latent varicella zoster virus (VZV) - Unilateral, localized, painful vesicular eruption - Major complication: post-herpetic neuralgia Rate per 1, 000 Person-Years Incidence of HZ per 1, 000 Person-Years 12 10, 9 9, 5 10 8 6, 9 6 4, 6 4 2 2, 9 1, 1 1, 4 0 to 14 15 to 29 2 0 30 to 39 40 to 49 50 -59 Patient Age (years) 60 -69 70 -79 > 80 J Gen Intern Med. 2005; 20(8): 748 -53.

Herpes Zoster, A History 1875 1943 1958 1982 1995 2006 2017 • Chickenpox recognized as distinct entity from Smallpox • Inoculation of HZ vesicular fluid into susceptible person results in chickenpox • VZV isolated from culture • Acyclovir approved for treatment of herpesvirus infections • VZV vaccine (Varivax®) approved by FDA • Herpes Zoster, live vaccine (Zostavax®) approved by FDA • Herpes Zoster, subunit vaccine (Shingrix®) approved by FDA Chicken Pox and Herpes Zoster. In: Mandell, Douglas, and Bennett’s Principles of Infectious Disease.

Prevention of Herpes Zoster Infection Varicella Virus Vaccine (Varivax®) Zoster Vaccine Live (Zostavax®) Herpes Zoster subunit vaccine (Shingrix®) CDC abbreviation VAR ZVL RZV Vaccine Type Live Inactivated; Recombinant 1350 PFU VZV 19400 PFU VZV glycoprotein E (g. E): 50 mcg AS 01 adjuvant: 3000 mcg ≥ 12 months ≥ 60 years ≥ 50 years 2 1 2 0, 1 month 1 dose 0, 2 -6 months Sub. Q Intramuscular Storage Freezer Refrigerator Diluent Sterile Water AS 01 adjuvant Vaccine Components Recommended Age Number of Doses Schedule Route PFU = plaque-forming units Varivax [package insert]. Merck: Whitehouse Station, NJ. 2017 Zostavax [package insert]. Merck: Whitehouse Station, NJ. 2018 Shingrix [package insert]. GSK: Research Triangle Park, NC. 2017

ACIP/CDC Recommendations for HZ Immunization • Should ACIP recommend RZV for vaccination of immunocompetent patients 50 years and older? - Yes • Should ACIP recommend RZV for persons who previously received ZVL? - Yes • Should ACIP recommend a preference of RZV over ZVL? - Yes MMWR. 2018; 67(3): 103 -108.

RZV: Vaccine Components Lyophilized recombinant VZV glycoprotein E (g. E): 50 mcg • Required for viral replication and cell-to-cell spread • Primary target of VZV-specific immune response AS 01 adjuvant • Induces production of antigen-specific antibodies and CD 4 cells Measures of efficacy • Humoral immunity: geometric mean concentrations (GMC) of serum anti-g. E and anti-VZV antibody • Cell-mediated immunity: CD 4+ T cells expressing at least 2 activation markers per 10 6 CD 4+ T cells (add 4 markers) • Interferon-γ, interleukin-2, tumor necrosis factor-α, CD 40 ligand • 4 -fold above baseline Hum Vaccin Immunother. 2017; 13(8): 1789 -97 Shingrix [package insert]. GSK: Research Triangle Park, NC. 2017
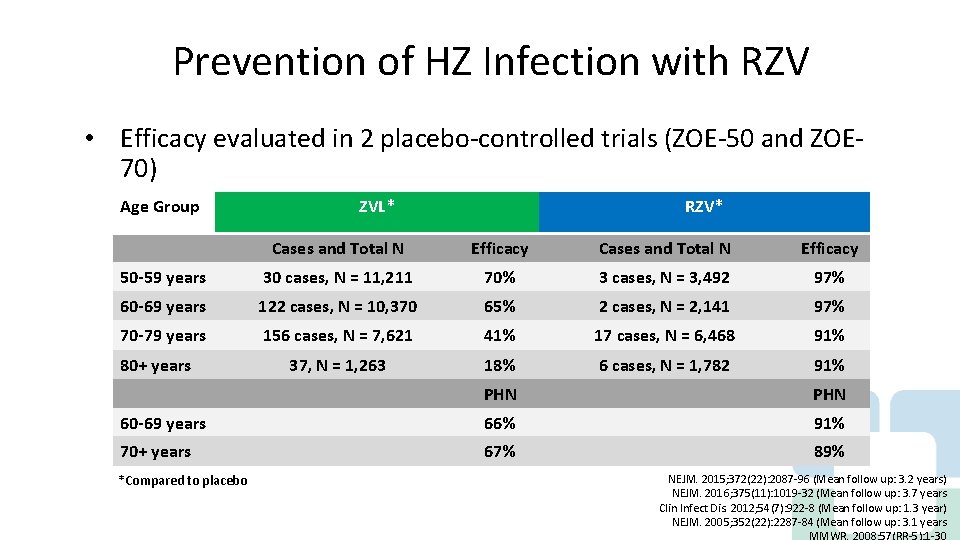
Prevention of HZ Infection with RZV • Efficacy evaluated in 2 placebo-controlled trials (ZOE-50 and ZOE 70) - Group Vaccine Age efficacy (VE) ZVL* in preventing HZ infection and PHN RZV* Cases and Total N Efficacy 50 -59 years 30 cases, N = 11, 211 70% 3 cases, N = 3, 492 97% 60 -69 years 122 cases, N = 10, 370 65% 2 cases, N = 2, 141 97% 70 -79 years 156 cases, N = 7, 621 41% 17 cases, N = 6, 468 91% 37, N = 1, 263 18% 6 cases, N = 1, 782 91% 80+ years PHN 60 -69 years 66% 91% 70+ years 67% 89% *Compared to placebo NEJM. 2015; 372(22): 2087 -96 (Mean follow up: 3. 2 years) NEJM. 2016; 375(11): 1019 -32 (Mean follow up: 3. 7 years Clin Infect Dis. 2012; 54(7): 922 -8 (Mean follow up: 1. 3 year) NEJM. 2005; 352(22): 2287 -84 (Mean follow up: 3. 1 years
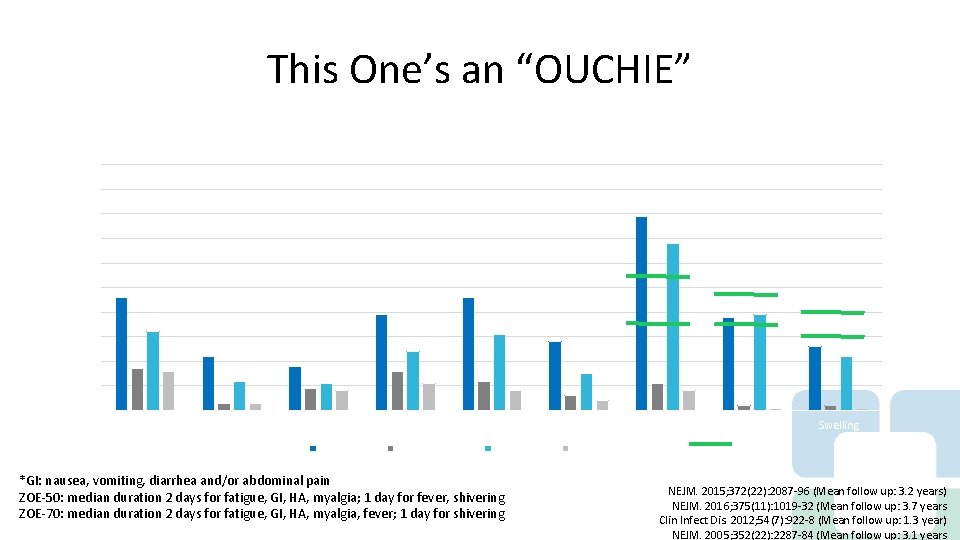
This One’s an “OUCHIE” Systemic Symptoms Reported During 7 days Post-Vaccination Any Grade Overall by Subject: ZOE-50 and ZOE-70 100 90 80 70 60 50 40 30 20 10 0 Fatigue Fever > 37. 5 GI RZV/ZOE-50 Headache Myalgia Placebo/ZOE-50 RZV/ZOE-70 *GI: nausea, vomiting, diarrhea and/or abdominal pain ZOE-50: median duration 2 days for fatigue, GI, HA, myalgia; 1 day for fever, shivering ZOE-70: median duration 2 days for fatigue, GI, HA, myalgia, fever; 1 day for shivering Shivering Placebo/ZOE-70 Pain Redness Swelling ZVL NEJM. 2015; 372(22): 2087 -96 (Mean follow up: 3. 2 years) NEJM. 2016; 375(11): 1019 -32 (Mean follow up: 3. 7 years Clin Infect Dis. 2012; 54(7): 922 -8 (Mean follow up: 1. 3 year) NEJM. 2005; 352(22): 2287 -84 (Mean follow up: 3. 1 years
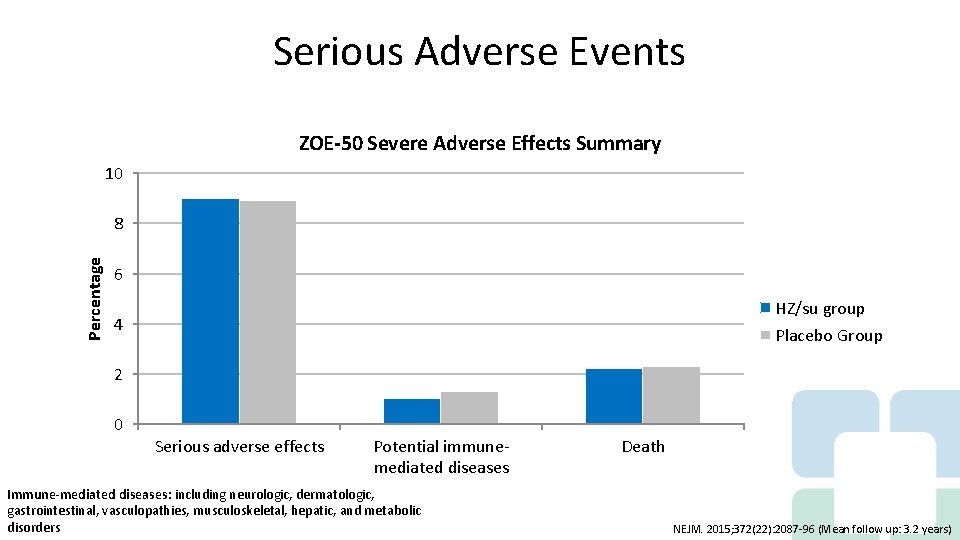
Serious Adverse Events ZOE-50 Severe Adverse Effects Summary 10 Percentage 8 6 HZ/su group 4 Placebo Group 2 0 Serious adverse effects Potential immunemediated diseases Immune-mediated diseases: including neurologic, dermatologic, gastrointestinal, vasculopathies, musculoskeletal, hepatic, and metabolic disorders Death NEJM. 2015; 372(22): 2087 -96 (Mean follow up: 3. 2 years)
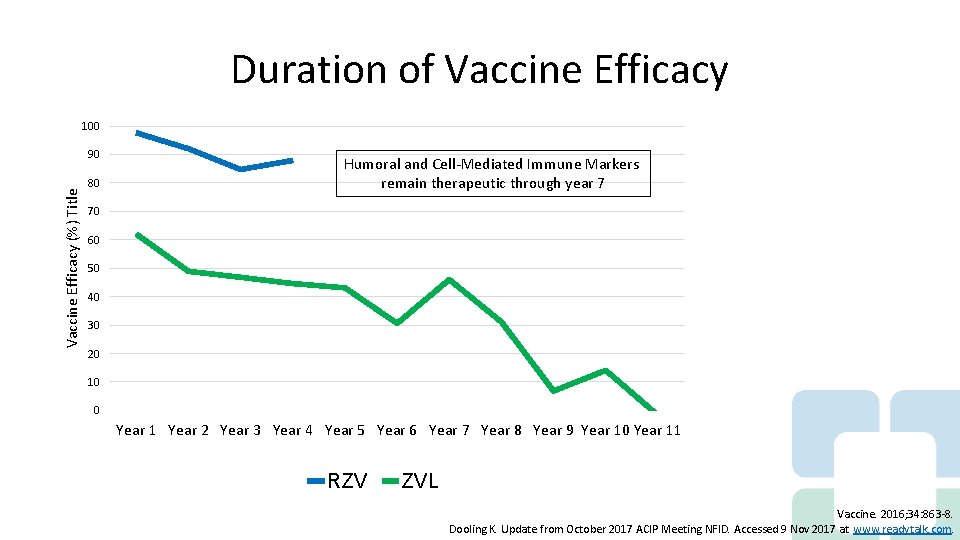
Duration of Vaccine Efficacy 100 Vaccine Efficacy (%) Title 90 80 Humoral and Cell-Mediated Immune Markers remain therapeutic through year 7 70 60 50 40 30 20 10 0 Year 1 Year 2 Year 3 Year 4 Year 5 Year 6 Year 7 Year 8 Year 9 Year 10 Year 11 Axis Title RZV ZVL Vaccine. 2016; 34: 863 -8. Dooling K. Update from October 2017 ACIP Meeting NFID. Accessed 9 Nov 2017 at www. readytalk. com.

Drug Interaction • Co-administration with other vaccinations - PPSV 23 may be administered concurrently Seasonal influenza quadravalent vaccine may be administered concurrently • Immunosuppressive medications - May decrease effectiveness/immune response to RZV vaccine No data when administered to patients on biologic immunomodulators or corticosteroids at dose > 20 mg prednisone equivalents daily Limited data in other immunocompromised patient populations GSK data on file: Immunogenicity and safety of Shingrix when co-administered with PPSV 23. Schwarz TF. J Infect Dis 2017; ahead of print. Shingrix [package insert]. GSK: Research Triangle Park, NC. 2017
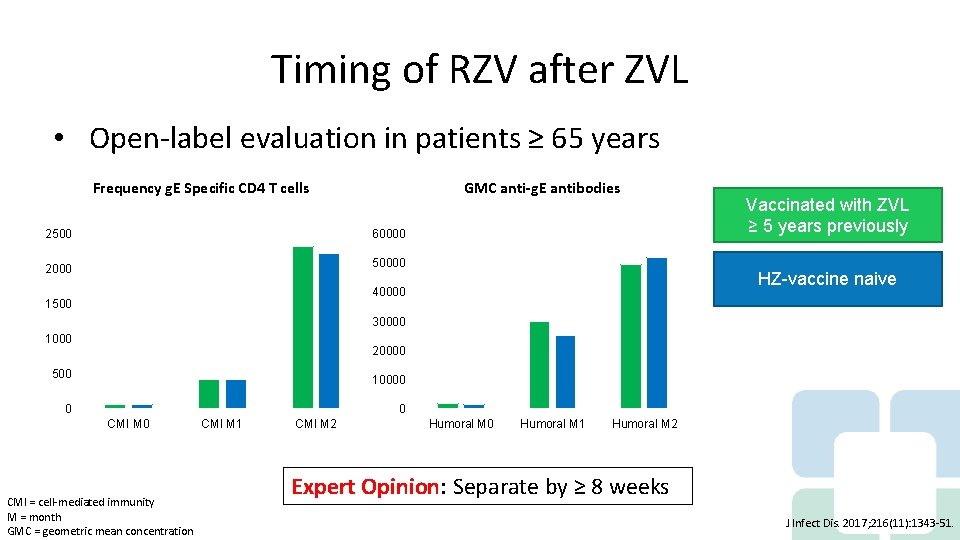
Timing of RZV after ZVL • Open-label evaluation in patients ≥ 65 years Frequency g. E Specific CD 4 T cells GMC anti-g. E antibodies Chart Title 2500 60000 2000 50000 HZ-vaccine naive 40000 1500 Vaccinated with ZVL ≥ 5 years previously 30000 1000 20000 500 10000 0 0 CMI M 1 Axis Title CMI = cell-mediated immunity M = month GMC = geometric mean concentration CMI M 2 Humoral M 0 Humoral M 1 Humoral M 2 Axis Title Expert Opinion: Separate by ≥ 8 weeks J Infect Dis. 2017; 216(11): 1343 -51.

Storage and Inactive Components • Refrigerated vials (36○F to 46○F) - Diluent vial (blue top): AS 01 adjuvant - Vaccine vial (brown top): g. E antigen - Once reconstituted: Stable in refrigeration x 6 hours • Does NOT contain - Natural rubber latex - Preservatives including thimerosal or hen’s egg allergens Key Point: Must be reconstituted with manufacturer supplied diluent Shingrix [package insert]. GSK: Research Triangle Park, NC. 2017

Vaccine Administration • Administer 0. 5 m. L intramuscularly (deltoid) • Vaccine administration outside of the recommended intervals - Best immune response = 0, 2 -6 month schedule • Minimum interval: 4 weeks - Administration at 0, 12 months did not meet non-inferiority compared to 0, 2 month or 0, 6 months - Opportunity to schedule second dose on the same day that first dose is administered Shingrix [package insert]. GSK: Research Triangle Park, NC. 2017 GSK Data on File: Immunogenicity and safety of 2 doses of Shingrix administered 2, 5, or 12 months apart in adults > 50 years of age.
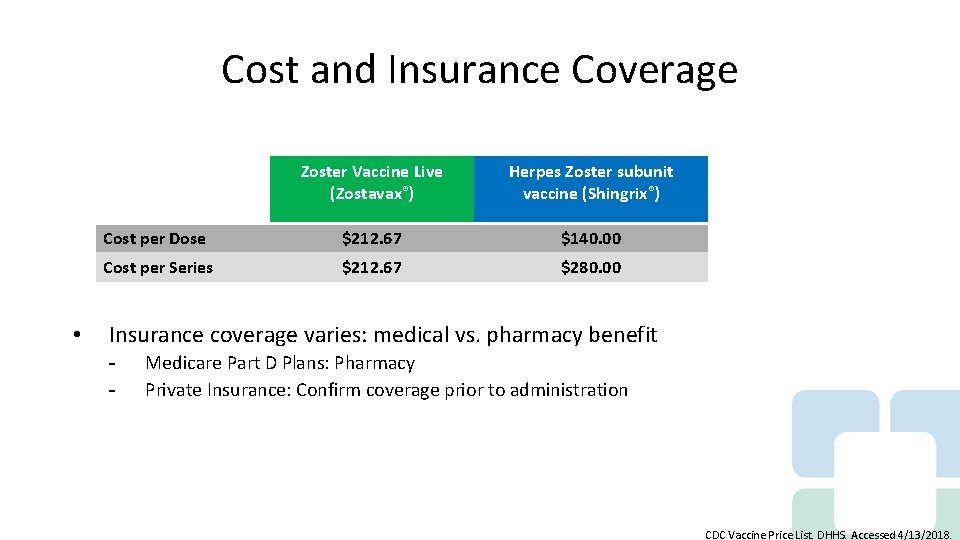
Cost and Insurance Coverage • Zoster Vaccine Live (Zostavax®) Herpes Zoster subunit vaccine (Shingrix®) Cost per Dose $212. 67 $140. 00 Cost per Series $212. 67 $280. 00 Insurance coverage varies: medical vs. pharmacy benefit - Medicare Part D Plans: Pharmacy Private Insurance: Confirm coverage prior to administration CDC Vaccine Price List. DHHS. Accessed 4/13/2018.
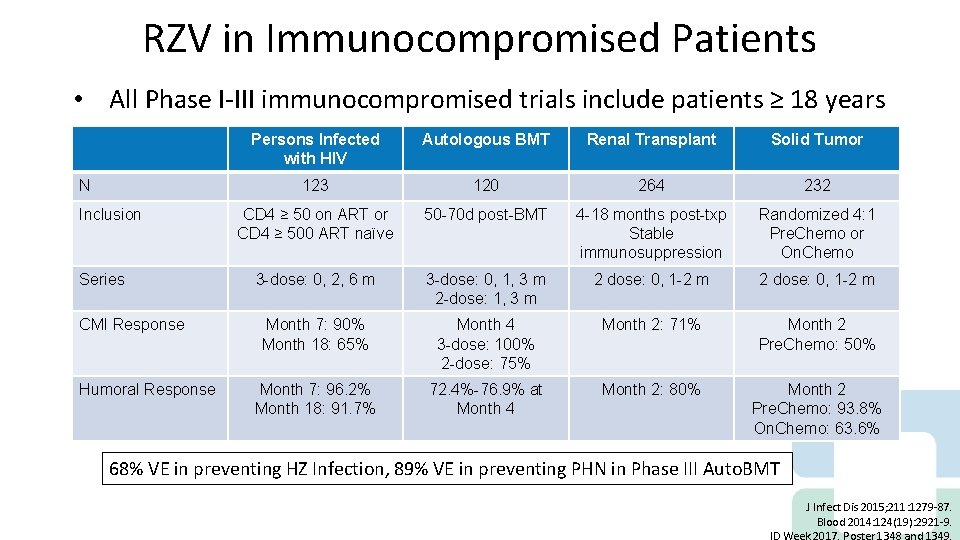
RZV in Immunocompromised Patients • All Phase I-III immunocompromised trials include patients ≥ 18 years N Inclusion Series CMI Response Humoral Response Persons Infected with HIV Autologous BMT Renal Transplant Solid Tumor 123 120 264 232 CD 4 ≥ 50 on ART or CD 4 ≥ 500 ART naïve 50 -70 d post-BMT 4 -18 months post-txp Stable immunosuppression Randomized 4: 1 Pre. Chemo or On. Chemo 3 -dose: 0, 2, 6 m 3 -dose: 0, 1, 3 m 2 -dose: 1, 3 m 2 dose: 0, 1 -2 m Month 7: 90% Month 18: 65% Month 4 3 -dose: 100% 2 -dose: 75% Month 2: 71% Month 2 Pre. Chemo: 50% Month 7: 96. 2% Month 18: 91. 7% 72. 4%-76. 9% at Month 4 Month 2: 80% Month 2 Pre. Chemo: 93. 8% On. Chemo: 63. 6% 68% VE in preventing HZ Infection, 89% VE in preventing PHN in Phase III Auto. BMT J Infect Dis 2015; 211: 1279 -87. Blood 2014: 124(19): 2921 -9.

Final Thoughts • RZV is a 2 -dose inactivated vaccine series for protection against HZ in patients ≥ 50 years of age - RZV preferred vaccine product - ZVL still option for patients ≥ 60 years • RZV has improved efficacy and durability compared to ZVL • Preliminary data in immunocompromised patients suggest RZV is safe and effective - ACIP anticipated to make recommendations in 2018 - Logistic challenges in meantime

Acknowledgements • Andrea Pallotta, Pharm. D, BCPS, AAHIVP • Brian Bohn, Pharm. D

- Slides: 20