Pandemic Influenza Vaccine Monovalent vaccine ACalifornia072009 H 1





- Slides: 6

Pandemic Influenza Vaccine • Monovalent vaccine – A/California/07/2009 (H 1 N 1) • Voluntary vaccination program will begin in early October • Public Health will coordinate vaccine distribution

Vaccine Supply • Federal Government Contract – Five manufacturers • TIV and LAIV • Adjuvanted and un-adjuvanted products • Preservative-free products – Doses purchased: 195 million – FDA approved products: 4 (as of 9/18/09) • Limited supplies, initially – Vaccine to be distributed continuously

Pandemic H 1 N 1 Vaccine Recommendations*, ACIP, July 2009 • Pregnant women • Household and caregiver contacts of children younger than 6 months of age • Health care and emergency medical services personnel • Children and young adults ages 6 months through 24 years • Persons aged 25 through 64 years who have medical conditions which put them at high risk for complications or death from influenza * Unadjuvanted vaccine

ACIP Recommendations Limited Vaccine Supply • Pregnant women • People who live with/care for children <6 mos • Healthcare & EMS workers with direct patient contact • Children 6 months - 4 years • Children 5 – 18 years with chronic medical conditions

Vaccine Administration • Number of doses – Adults • 1 dose (likely) – Children < 10 years • 2 doses (likely) regardless of previous vaccination status • Three week interval (likely)

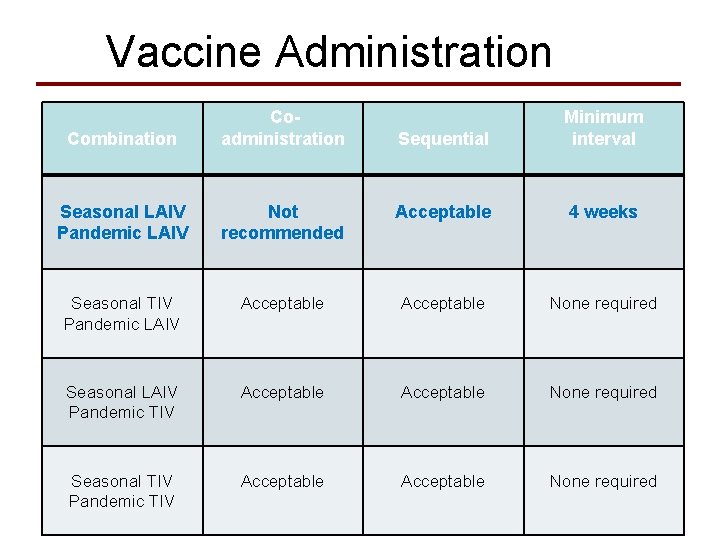
Vaccine Administration Combination Coadministration Sequential Minimum interval Seasonal LAIV Pandemic LAIV Not recommended Acceptable 4 weeks Seasonal TIV Pandemic LAIV Acceptable None required Seasonal LAIV Pandemic TIV Acceptable None required Seasonal TIV Pandemic TIV Acceptable None required











