Massachusetts Department of Public Health Training Module Vaccine

Massachusetts Department of Public Health Training Module Vaccine Management, Storage, and Handling Presented by: Bob Morrison Vaccine Manager Massachusetts Department of Public Health November 16, 2010 10/24/2020 1

Learning Objectives ¨ Summarize principles of vaccine management ¨ Describe current vaccine storage and handling recommendations ¨ Describe preventative measures to maintain refrigerator and freezer temperatures ¨ List steps to protect vaccine in the event of a power outage or emergency 10/24/2020 LA DPH 2
Vaccine Management 10/24/2020 3

Importance of Vaccine Protection o Vaccines are fragile-keep at recommended temperatures at all times to maintain potency o Mishandled vaccines increase the risk that recipients will not be protected o Vaccines are expensive o Vaccine Restitution Policy, effective 1/1/11 10/24/2020 LA DPH 4

Vaccine Management Guidelines ¨ Develop and maintain detailed written protocols and emergency plan ¨See www. mass. gov/dph/imm for sample templates ¨ ¨ Assign responsibilities to one person Designate a back-up person Provide staff training on vaccine management Obtain proper equipment and materials 10/24/2020 LA DPH 5

Vaccine Manager Responsibilities o Order vaccine and oversee inventory o Receive vaccine and refrigerate/freeze immediately o Provide proper storage and handling o Handle damaged, wasted and expired vaccine o Respond when vaccine is out of required temperature range n Contact MDPH Vaccine Unit to determine if vaccine has been damaged (617 -983 -6828) 10/24/2020 LA DPH 6

Vaccine Inventory Management ¨ Limit access to authorized personnel ¨ Order vaccines regularly, do not stockpile ¨ Organize vaccine with shortest shelf life in front ¨ Conduct and log vaccine inventory monthly ¨ Rotate vaccines and monitor expiration dates 10/24/2020 LA DPH 7

Vaccine Inventory Management ¨ Separate by lot numbers and formulations ¨ Keep vaccines in original packaging to protect from light ¨ Never use expired vaccine or diluent ¨ Report vaccine usage if required ¨ Return unused, expired vaccine and diluent if required 10/24/2020 LA DPH 8

Receiving Vaccine ¨ ¨ ¨ ¨ Examine shipment immediately upon arrival Report problems to MDPH within two hours Check contents against packing slip Check vaccine expiration dates Examine contents for damage Check temperature monitor Immediately place in appropriate storage Keep a shipping box on hand to use when returning any damaged or expired vaccine 10/24/2020 9
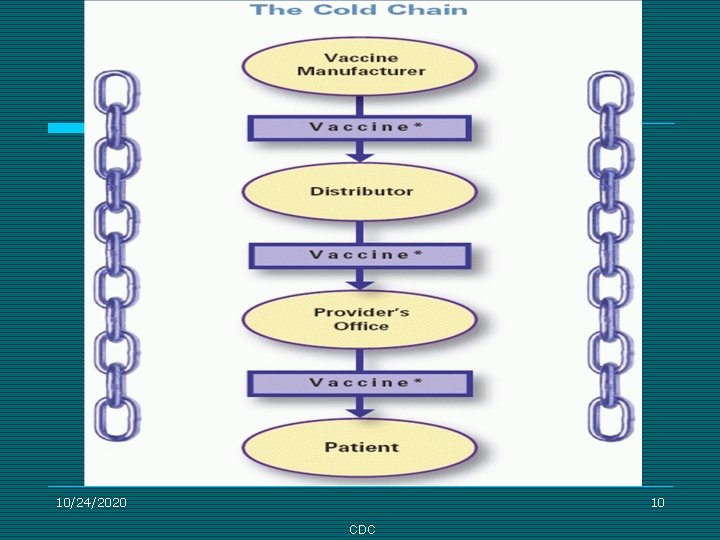
10/24/2020 10 CDC

3 Components of the Cold Chain ¨ Proper transport and storage equipment ¨ Trained personnel ¨ Current vaccine management policies and procedures 10/24/2020 LA DPH 11

Maintain the Cold Chain o Transport vaccine from refrigerator to clinic site in cooler with cold packs (not frozen packs) and thermometer o Refrigerate cold packs for at least 24 hours before use in transport o Keep vaccine on cold packs, but provide a barrier to protect from direct contact with cold pack 10/24/2020 12

Vaccinator Responsibilities at Station o Maintain the cold chain at vaccinator station n Injectable vaccine: max 30 minutes at room temperature n Intranasal vaccine: kept at 2 - 8° C (35 - 46°F) o Any vaccine drawn up must be used by end of clinic or be disposed of o Protect from exposure to light o Label opened unused vials with date and your initials 10/24/2020 13

Vaccine Storage and Handling 10/24/2020 CDC 14

General Vaccine Storage Requirements ¨ Have unit large enough to store your maximum vaccine inventory ¨ Dedicate unit to vaccines/medications only ¨ Monitor refrigerator/freezer temps for at least 3 days prior to storing vaccine ¨ Maintain required temperature range yearround ¨ Make arrangements for vaccine storage, monitoring temperatures and receipt of vaccine during vacations or other lengthy absences 10/24/2020 15

Vaccine Storage Units o CDC recommends separate refrigerator and freezer units for vaccine storage o For combination units, make sure there are separate doors and controls for refrigerator and freezer o It is recommended that units used to store vaccines be less than 10 years old o Have a certified calibrated thermometer inside both the refrigerator and freezer in a central area 10/24/2020 LA DPH 16

Maintain Proper Temperatures HANDLE WITH CARE! Maintain refrigerator temps between 2°-8°C or 35°-46°F Maintain freezer temps at -15 °C or 5°F or colder Protect from light 10/24/2020 17

Vaccine Storage & Handling Tips ¨ Do not store vaccine on unit doors ¨ Do not store biologics with food or drink ¨ Place thermometer and vaccine in central area of the unit ¨ Organize and label vaccine in baskets that allow air circulation ¨ Refer to package insert for specific guidelines regarding the storage and handling of different formulations 10/24/2020 LA DPH 18

Acceptable Vaccine Storage 10/24/2020 CDC 19

Unacceptable Vaccine Storage 10/24/2020 CDC 20

Unacceptable Vaccine Storage 10/24/2020 21

Certified Calibrated Thermometers 10/24/2020 22

Monitor Temperatures ¨ Record temperatures twice daily § First thing in the morning § End of the work day § Temperature logs must be retained for at least 3 years § Monitor for out-of-range temperatures ¨ Place temperature logs on front of unit ¨ Submit temperature logs as required 10/24/2020 LA DPH 23

Out of Range Temperatures o All out of range temperatures require immediate reporting to MDPH Vaccine Unit 617 -983 -6828 or 617 -983 -6800 (24 x 7) 1 -888 -658 -2850 o Actions and results of these actions must be documented on the temperature logs 10/24/2020 LA DPH 24

Emergency Plan for loss of power ¨ Develop an emergency plan ¨ Share the emergency plan ¨ Post the emergency plan ¨ Update and practice your plan annually 10/24/2020 LA DPH 25

Power Failure ¨ If you lose power for extended period, follow your emergency plan: § pack vaccine § transport to prearranged site § notify MDPH Vaccine Unit ¨ When power returns: § do not adjust the temperature control in unit § allow unit to return to proper temperature range § return vaccine to unit 10/24/2020 LA DPH 26

Temperature Adjustment 10/24/2020 27

Temperature Adjustment o Never adjust the refrigerator temperature control with vaccine in unit o Consult with MDPH Vaccine Unit o Remove vaccine to another refrigerator/freezer o Adjust temperature of unit o Wait until you have 3 successive readings one hour apart within range before returning vaccine to the unit 10/24/2020 28 LA DPH

Safeguard Power Supply ¨ Use a plug guard or safety-lock plug ¨ Place “Do Not Unplug” sign on storage unit, outlet and “Do Not Disconnect” on circuit breakers ¨ Consider installing a temperature alarm ¨ Do not use extension cords 10/24/2020 LA DPH 29

Safeguard Power Supply 10/24/2020 CA DPH 30

Safeguard Power Supply 10/24/2020 CA DPH 31

The Effect of “Thermotrauma” on Vaccines o Live vaccine: § Tolerates freezing (Call MDPH Vaccine Management Unit if this occurs: 617 -9836828) § Deteriorates rapidly after removal from refrigeration o Inactivated vaccine: § Damaged by freezing § Tolerates short time out of refrigeration 10/24/2020 32

Vaccine Shelf Life o Multi-dose vials of vaccine contain a bacteriostatic agent. n After opening, can be used until the date of expiration as specified by the manufacturer and package insert, unless visibly contaminated. n Date and initial multi-dose vials after opening and use first. n In the event the refrigerator/freezer temperatures are out-ofrange, this is important in determining the viability of the remaining doses in the vial. n Always check the package insert for shelf life guidelines when opening multi-dose vials. o Single-dose vials do not contain a bacteriostatic agent. n Once opened, they must either be used or discarded at the end of the day. 33

Storing Diluents o Diluents packaged separately from their corresponding vaccines can be stored at room temperature or in the refrigerator. o Diluents packaged with their vaccines should be stored in the refrigerator. o Once vaccines are reconstituted with a diluent, their shelf life is limited. 10/24/2020 34

Take-Home Messages ¨ Colder is NOT better for inactivated vaccines ¨ It is estimated that >$100 million worth of vaccine is exposed to freezing temperatures each year in the United States ¨ Out of range temperature readings require IMMEDIATE action ¨ Remove expired vaccine from refrigerator ¨ Report vaccine usage to MDPH Vaccine Unit: 617 -983 -6828 10/24/2020 35

Vaccine Storage & Handling Questions o MDPH Vaccine Management Unit (617) 983 -6828 o MDPH Immunization/Epidemiology Division (888) 658 -2850 (617) 983 -6800 (24 x 7) o Always consult with MDPH Immunization Program before removing improperly stored vaccine from the storage unit n If the temperature of the vaccine goes out of range, either too high or too low, call the MDPH Vaccine Management Unit immediately! 10/24/2020 36

Please take a look at the other training modules! o Vaccine and Medication Preparation and Administration o Planning a Clinic/EDS/POD o All-Staff Briefing & Just-In-Time Training o Available at: http: //sph. bu. edu/otlt/alliance/training_pu blichealthnursing. php 10/24/2020 37

Online Resources o MDPH Guidelines for Compliance with Federal and State Vaccine Administration Requirements http: //www. mass. gov/Eeohhs 2/docs/dph/cdc/immunization/guidel ines_vaccine_compliance. pdf o MDPH General Protocols for Vaccine Storage, Administration, Standing Orders, and Mass Immunization Clinics, Sept 2009 http: //www. mass. gov/Eeohhs 2/docs/dph/cdc/immunization/mso_ protocols_general. pdf o MMWR: General Recommendations on Immunization, Recommendations of the Advisory Committee on Immunization Practices (ACIP), December 2006 http: //www. cdc. gov/mmwr/pdf/rr/rr 5515. pdf o MDPH Vaccine Management Guidelines for Vaccine Provider http: //www. mass. gov/Eeohhs 2/docs/dph/cdc/immunization/vaccin e_management_checklist. pdf 10/24/2020 38

Online Resources o MDPH Immunization Program http: //www. mass. gov/dph/imm o Immunization Action Coalition www. immunize. org o Epidemiology and Prevention of Vaccine. Preventable Diseases, The Pink Book: Course Textbook (11 th Edition), May 2009 http: //www. cdc. gov/vaccines/pubs/pinkbook/default. htm# download o World Health Organization http: //www. who. int/en 10/24/2020 39

Online Resources o MDPH Sample Vaccine Management-Standards Operating Procedures (SOP) http: //www. mass. gov/Eeohhs 2/docs/dph/cdc/immuniz ation/vaccine_management_sop_sample. pdf o Emergency Response Worksheet for vaccine storage http: //immunize. org/catg. d/p 3051. pdf o Pediatrics “Standards for Child and Adolescent Immunization Practices” National Vaccine Advisory Committee, Pediatrics 2003: 112: 958 -963 http: //www. pediatrics. org/cgi/content/full/112/4/958 10/24/2020 40

Online Resources o CDC: Vaccine Storage and Handling Toolkit http: //www 2 a. cdc. gov/vaccines/ed/shtoolkit/default. htm o CDC: Vaccine Management Business Improvement Project (VMBIP); New Centralized Vaccine Ordering and Storage Requirements http: //www. cdc. gov/vaccines/programs/vmbip/default. htm o MDPH: Vaccine Management Business Improvement Plan: Basic Guidelines http: //mass. gov/EOHHS 2/docs/dph/cdc/immunization/vmbip_basic_gu idelines. pdf o Los Angeles County Immunization Program, Vaccine Storage and Handling presentation http: //www. lapublichealth. org/ip/trainconf/Vaccine%20 Storage%20 Ha ndling. pdf 10/24/2020 41

QUESTIONS? Thank You ! 10/24/2020 42
- Slides: 42