Matter Outline Properties of Matter States of matter




























- Slides: 39

Matter

Outline �Properties of Matter States of matter Physical properties Chemical properties �Changes in Matter Physical changes Chemical changes Law of Conservation of Mass �Mixtures, Elements and Compounds Heterogeneous, Homogeneous mixtures Separating Mixtures Elements and Compounds

What is matter? �Everything in the universe is made of matter. �Matter is made of tiny particles called atoms.

States of Matter �All matter exists as one of the three physical forms, which are called the states of matter. Solid Definite Shape Definite Volume Particles held tightly together Liquid Gas

Physical Properties �A physical property is a characteristic of matter that can be observed directly or measured without changing the substance’s composition. �Examples of Physical Properties are: �Boiling point Ductility �Melting point Elasticity �Density Brittleness �Color �Odor �Malleability

Chemical Properties �Characteristic of matter that can only be observed when one substance changes into a different substance �Examples of Chemical Properties: �Flammability �Reactivity with acids �Ability to rust

Identifying Changes �Physical Changes Physical state • Chemical Changes

Types of Change Type of Change Products of Change Physical Change No new substance, but physical state is changed. Example: ___________ Chemical Change New substances are formed. Also called a chemical reaction. Example: ___________

Chemical Reactions �A chemical reaction is a process in which atoms of one or more substances rearrange to form one or more new substances. �When a new substance(s) is formed, there are indications. • • Color Change • Temperature change Change in odor • Light Formation of Bubbles Formation of Precipitate

Conservation of Mass �The Law of Conservation of Mass states that mass is neither created nor destroyed during a chemical reaction. Mass is always conserved in a chemical reaction.

Chemical Reactions and Chemical Equations �Chemical reactions are written in the form of a chemical equation. �The starting substances are called the reactants. �The substances produced, or formed, are called the products. �Reactant + Reactant Product + Product �Eg.

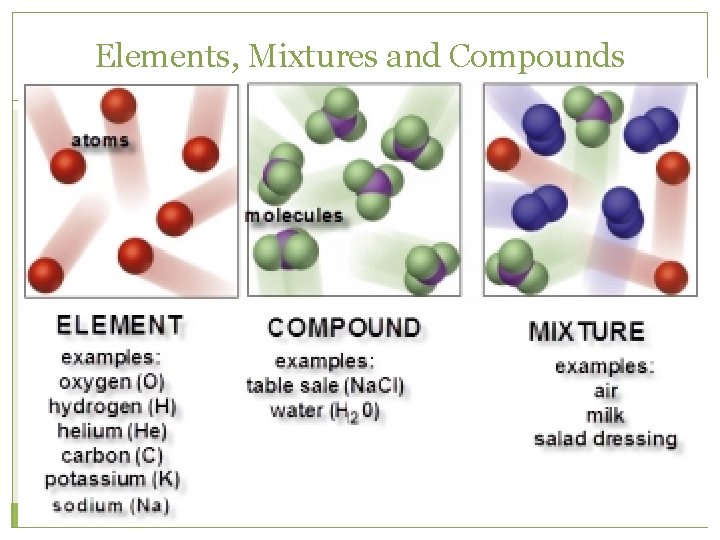
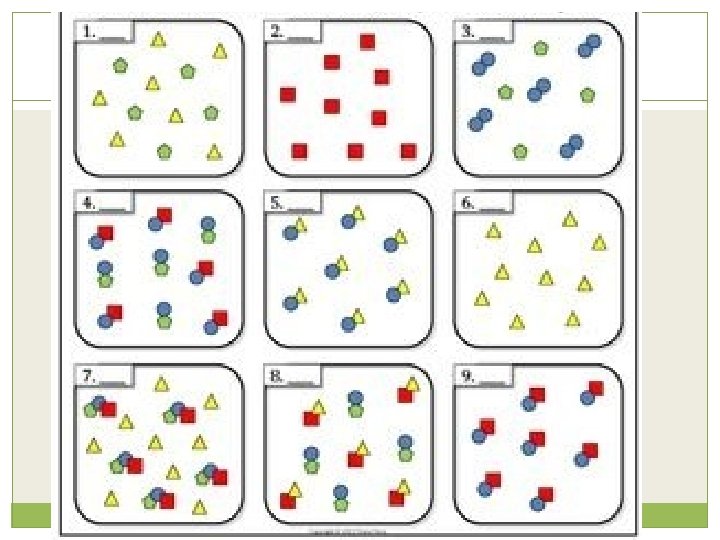
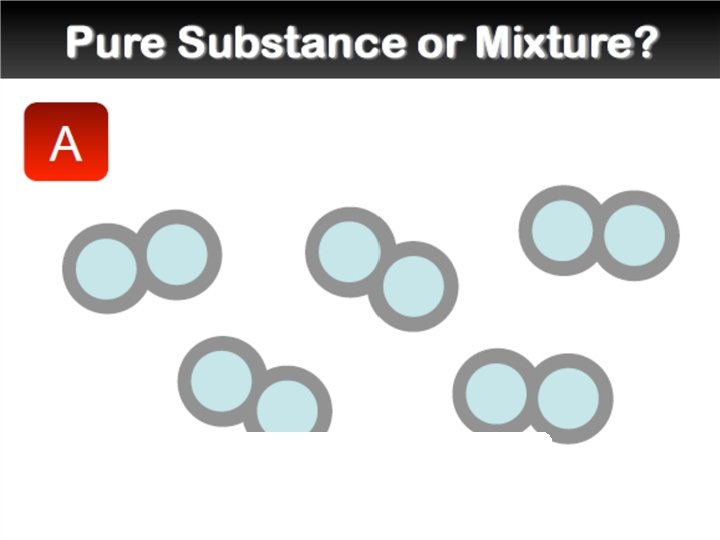
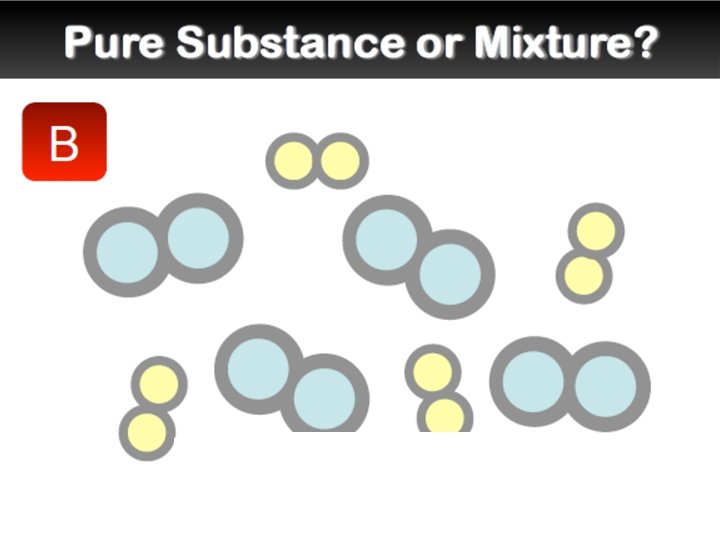
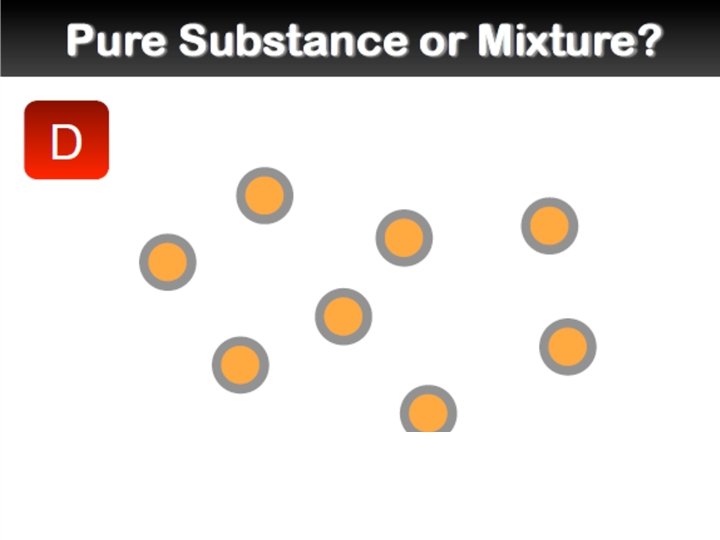
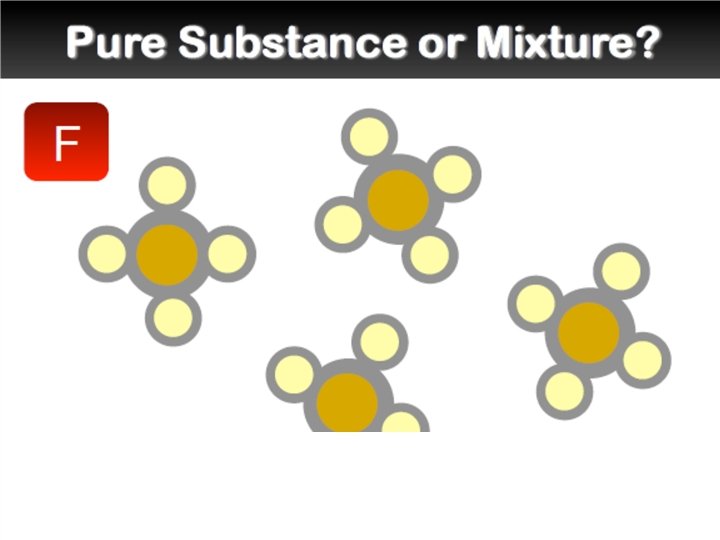
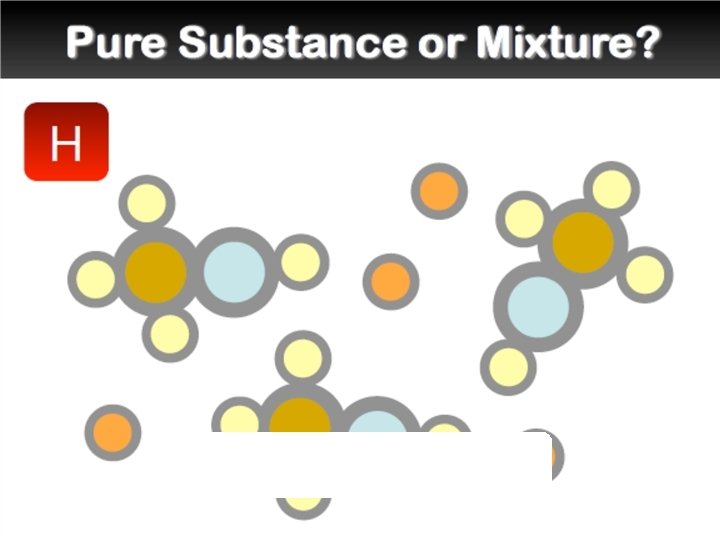
Elements and Compounds �An element is a pure substance that cannot be separated into simpler substances by any means. Each element has a name and a chemical symbol. Elements are organized in the periodic table. �A compound is made up of two or more different elements that are combined chemically.

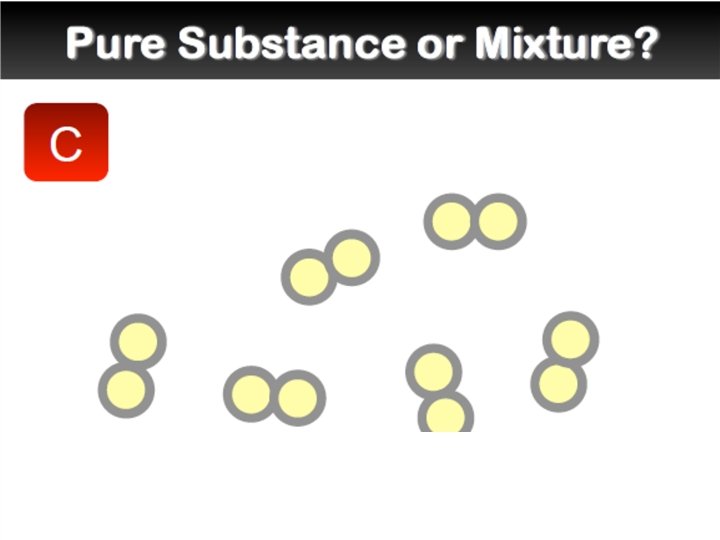
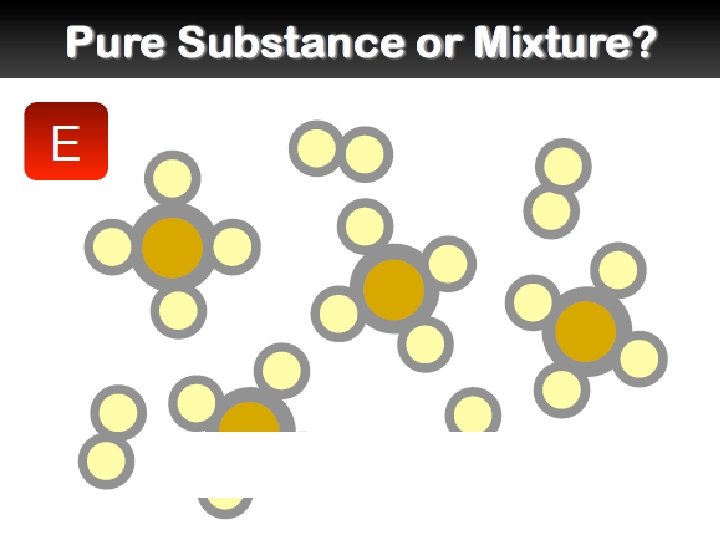
Mixtures �A mixture is a combination of two or more pure substances. Each substance in a mixture keeps its chemical properties. �A pure substance is a substance that is the same throughout. Example:

Compounds: Law of Definite Proportions �A compound is always composed of the same elements in the same proportion by mass. �Mass of a compound = sum of the masses of the elements that make up the compound

Compounds: Law of Multiple Proportions �When different compounds are formed by a combination of the same elements, �If two elements form more than one compound between them, then the ratios of the masses of the second element (which combine with a fixed mass of the first element) will be ratios of small whole numbers �Example:

Elements, Mixtures and Compounds













Mixtures �Types of Mixtures: Homogeneous: “the same” A mixture that has constant composition throughout. Example: Heterogeneous: “different” A mixture that does not blend smoothly and has a non-uniform composition Example:



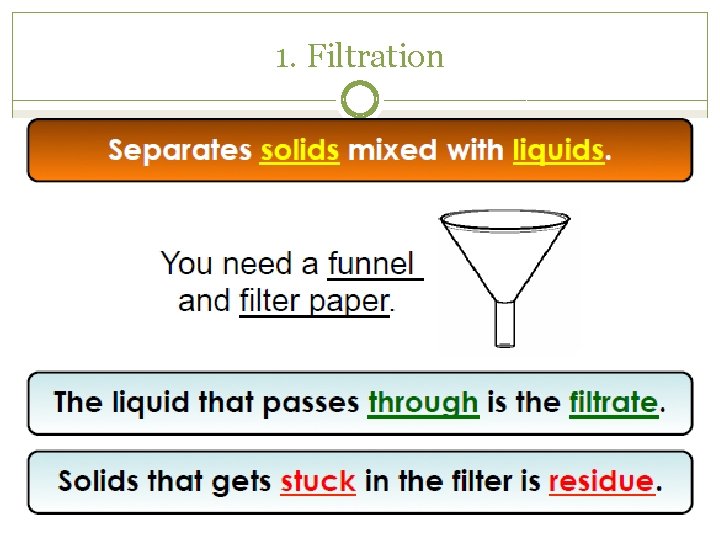
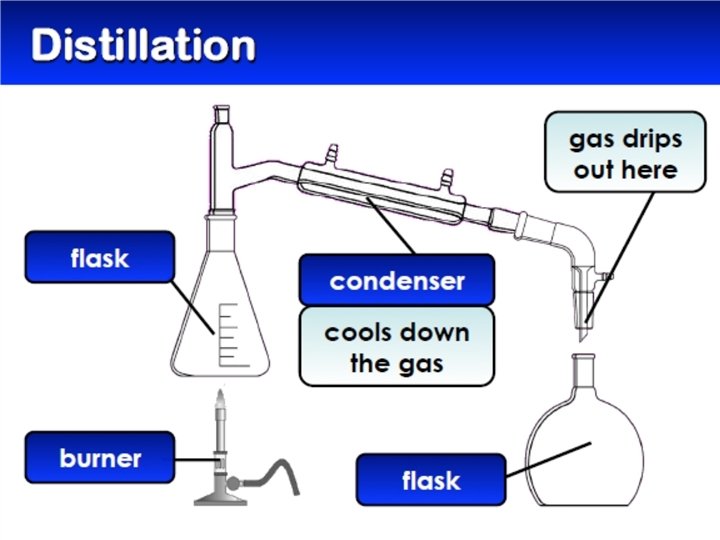
Separating Mixtures �Because substances in a mixture are physically combined, the processes used to separate a mixture are physical (no need for chemical reactions) �Filtration �Distillation �Crystallization �Sublimation �Chromatography

1. Filtration

2. Distillation


3. Crystallization �Formation of pure solid particles of a substance from a solution containing the dissolved substance.

4. Sublimation or Evaporation �Sublimation: Solid changes to vapor without melting �Eg. �Evaporation: Liquid changes to gas �Eg.


5. Chromatography