Whats the Matter foldable Chemistry Chemistry is the

What’s the Matter foldable

Chemistry • Chemistry is the study of matter and the changes matter undergoes.

Matter • Matter is anything that takes up space and has mass.

General Properties • mass • weight • volume • density • shape • color • texture

Properties of Matter v How It Looks (Shiny , Dull, Color, etc. ) v How It Feels (Hard, Soft, Rough , Smooth, etc. ) v How It Smells (Sweet, Sharp, Terrible, No Smell, etc. ) v How It Sounds (Loud, Soft, Echo, No Sound, etc. ) v What It Does (Bounce, Stretch, Tear, Break, Magnetism etc. )

SOLIDS

Solids Characteristics vdefinite shape vdefinite volume vobjects do not pass through easily v. Examples: paperclip, bones, desk, car, chair, etc…

Solids v. Molecules are very close vmolecules move very slowly in place.

Liquids

Liquids Characteristics v no definite shape v definite volume v takes shape of container v has surface tension v example: water, soda, glue, oil, etc…

Liquids v molecules have some space between them. v molecules move around a bit faster.

GAS

GAS Characteristics vno definite shape vno definite volume vexpands to fill container vexamples: oxygen, hydrogen, steam, air, carbon dioxide, etc…

GAS v. Molecules have lots of space. vmolecules very fast.

Metals Characteristics v. Most are solid at room temp. Except Mercury. v. Most elements are metals v. Located to the left of the zigzag line. v. Shiny, conduct heat & electricity, and Malleable.

Non-Metals Characteristics v. Located on the right side of the zigzag line. v. More then ½ are gases at room temperature. vdull, DO NOT conduct heat or electricity.

Metalloids Characteristics v. Border the zigzag line v. Have properties of both metals and nonmetals. v. Called “semi-conductors”.

Compound • Molecule made of more then one type of atom, chemically combined in a fixed proportion. • CAN NOT be separated easily by physical means. • Properties of the compound are much different than the elements that make them up.
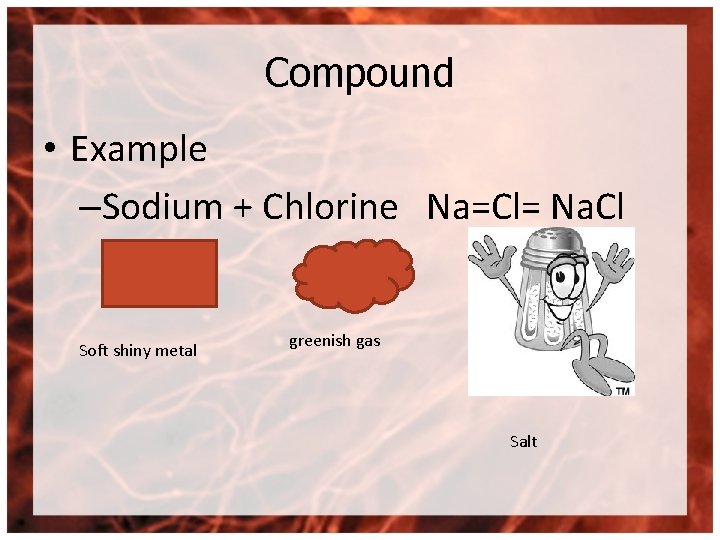
Compound • Example –Sodium + Chlorine Na=Cl= Na. Cl Soft shiny metal greenish gas Salt

Mixture • Made up of different types of particles, they are NOT chemically combined. – tossed salad • CAN BE separated easily by physical means. • 2 Types of mixtures • Homogeneous-the same appearance and composition throughout.

Mixtures • Heterogeneous-made up of different visible substances. Example oil and vinegar • Example; Chocolate chip cookie recipe Recipe ingredients+ baking=cookie

Element • Molecules made up of only one type of atom

Solutions • Special Mixture where a substance dissolves in another. • particles are not big enough to be seen. • Particles spread out evenly – Example + H 2 O =

Physical Property Characteristic of matter that can be seen through direct observation without changing the identity of the substance.

Density: mass/volume. Brittleness: cracks/breaks. Transparency: light passes through. Boiling point: temperature liquid to gas. Freezing point: temp from liquid to solid. Melting Point: temp from solid to liquid. Ductility: bends without breaking. Elasticity: how much it can be stretched.

Physical Change v Change in which the identity of the substance does not change! v Atoms do not rearrange v only physical properties change, chemical do not change v No energy is produced. v Physical changes are generally easy to reverse.

Chemical Property v. Characteristics of a substance indicating that it can change chemically. It changes one substance into another.

properties v. Flammability- ability to burn v. Combustible- explode v. Rust- reacts with oxygen v. React with Vinegar-produce new substance. v. Oxidation- produces something new v. All chemical changes include physical change

Chemical Change How the particles of matter react with other kinds of matter and change. v color v temperature v bubble formation v can not be reversed v ex burning eggs

Chemical Change v Atoms are rearranged into different molecules v Both physical and chemical properties have changed. v Energy is often produced (heat) v CHANGES ARE NOT REVERSIBLE WITHOUT ANOTHER REACTION!

ENDOTHERMIC reaction v. Absorbs energy vsolid to liquid- melting vex-ice-water v hot

Exothermic v releases energy v liquid to solid- freezing v water to ice v cold
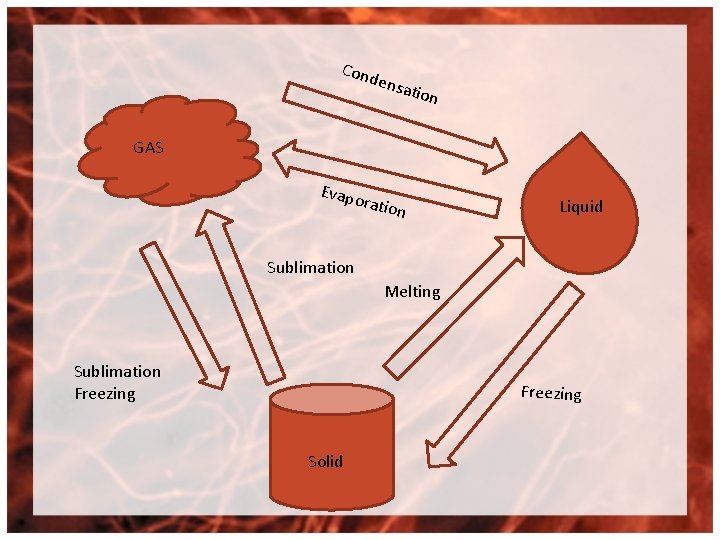
Cond ensa tion GAS Evapo ration Liquid Sublimation Melting Sublimation Freezing Solid

Solid liquid melting (ICE to water) Solid gas- Sublimation ex: Dry ice CO 2 Liquid Gas – Evaporation H 2 O steam Liquid Solid- Freezing H 2 O ice Gas Liquid- Condensation Steam H 2 O Gas Solid- Sublimation freezing CO 2 dry ice

p. H Scale A range showing the strength of acids and bases 1 Acid 7 Neutral 14 Base

Acid- A substance that is sour, reacts with metals has H+ ions Examples: lemon juice, orange juice, vinegar, perfume, Alcohol , pineapple. (picture) Base- A substance that is bitter, slippery, and has OH¯ ions. Examples- Hairspray, furniture polish, bleach, (non-scented) soap. (picture)

Solubility= The ability of one substance to dissolve in another at a given temperature & pressure.

Solubility Soluble- able to dissolve in another substance Insoluble- Not able to dissolve in another substance

Solubility Insoluble can equal a precipitate. • a substance formed when two or more substances are mixed and a solid “falls” out of the liquid and settles on the bottom.
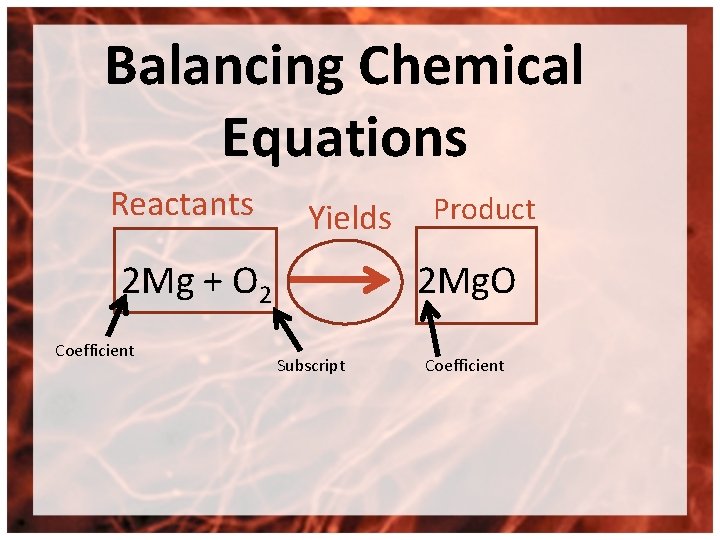
Balancing Chemical Equations Reactants Yields 2 Mg + O 2 Coefficient Product 2 Mg. O Subscript Coefficient

Balancing Chemical Equations Steps 1. Determine number of atoms on each side. 2. If not equal pick one element you need to work with. 3. Add a coefficient in front of formula adjust counts to see if balanced. 4. If not keep adding to get each side equal.
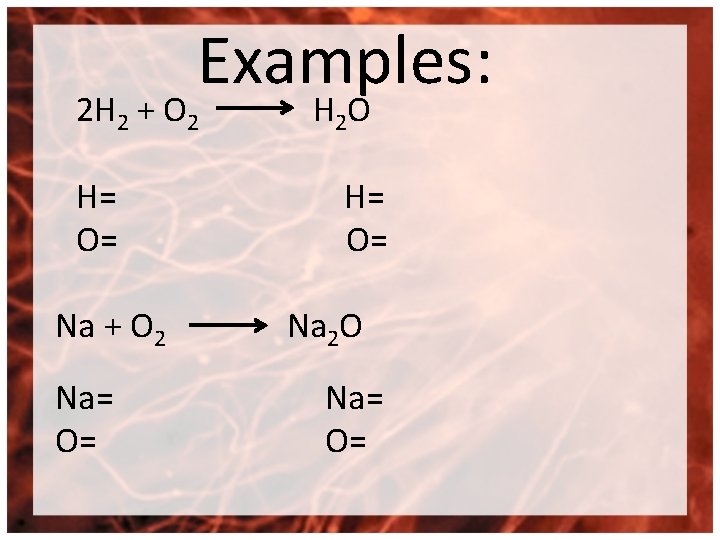
Examples: 2 H 2 + O 2 H= O= Na + O 2 Na= O= H 2 O H= O= Na 2 O Na= O=
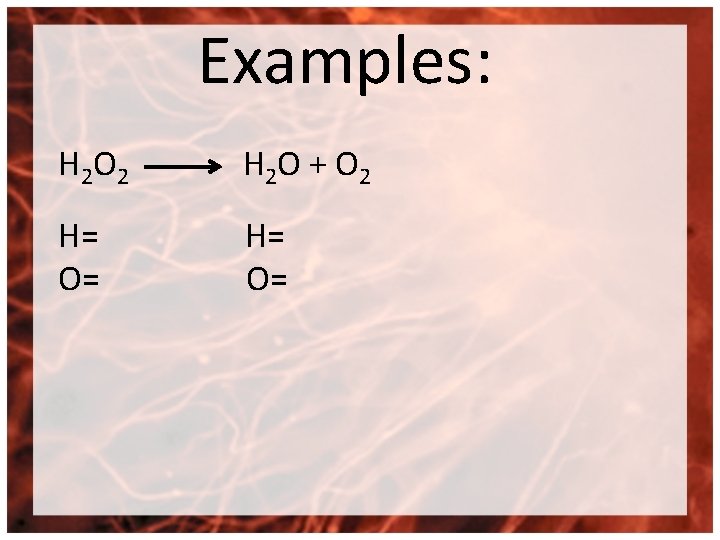
Examples: H 2 O 2 H 2 O + O 2 H= O=
- Slides: 43