States of Matter Gases States of Matter The



















- Slides: 22

States of Matter & Gases

States of Matter The three states of matter include: Solid: Liquid: Gas State Volume Shape Solid definite Liquid definite indefinite Gas indefinite

Energy and phase change • When Heated: Solid liquid gas • When Cooled: Gas liquid solids

Energy & phase change

Heating Curve & phase changes • Substances can exist in three states. They are solid, liquid, and gas. They (the substances) change when the kinetic energy of a substance changes. • When a substance changes from a solid to a liquid, it is called melting. • When a substance changes from a liquid to a gas, it is called vaporization. • Gas particles have the most kinetic energy of the three states, but they also have the lowest potential energy. This is opposite of a solid which has high potential energy but low kinetic energy. • There is one special phase change which is not common. The change is from a solid to a gas. This is called sublimation.

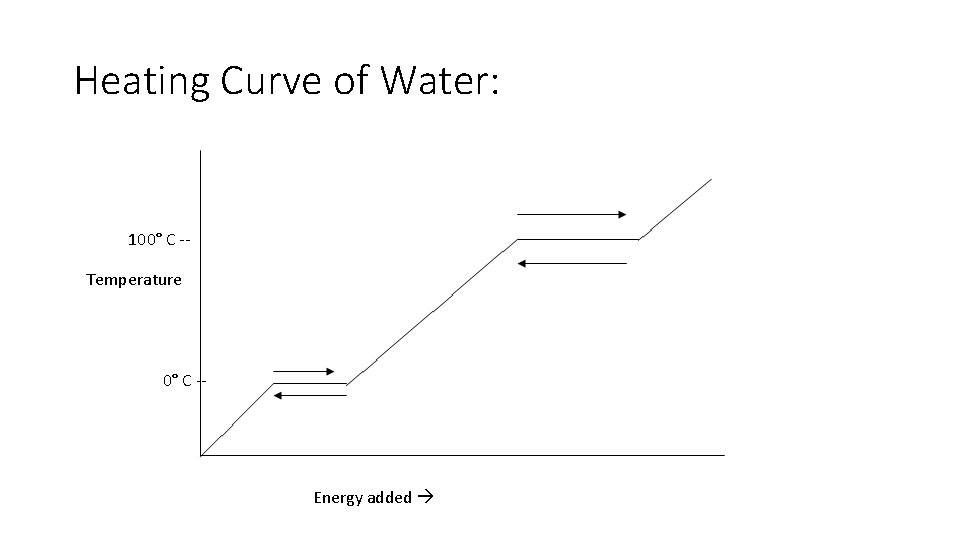
Heating Curve of Water: 100° C -Temperature 0° C -- Energy added

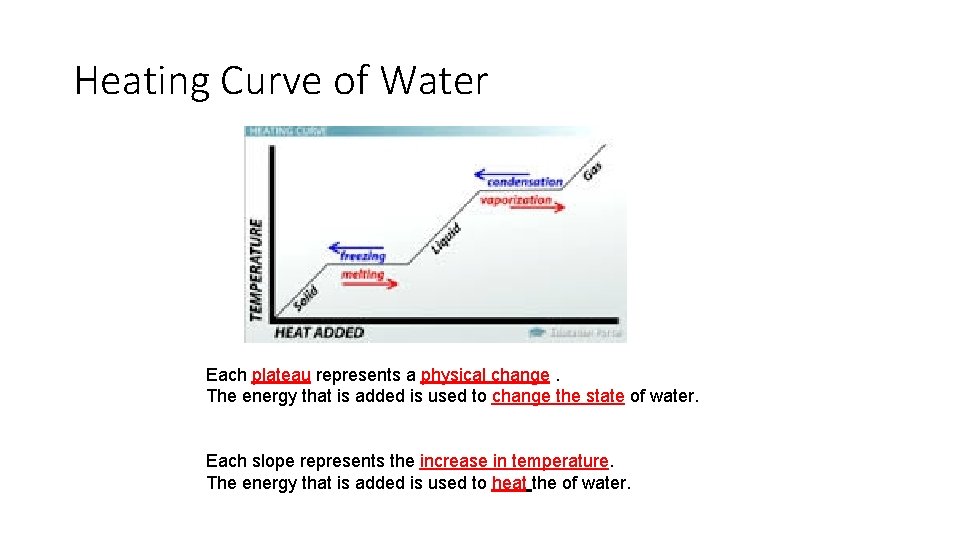
Heating Curve of Water Each plateau represents a physical change. The energy that is added is used to change the state of water. Each slope represents the increase in temperature. The energy that is added is used to heat the of water.

Gases: • Parameters used to describe Gases: • The variables used to describe the properties of a gas include: • • P = Pressure V = Volume T= Temperature n = moles (amount)

Temperature: • Temperature (T): must be described using the unit Kelvin (K). K = °C + 273 • You try: °C = K – 273 1. 25 °C = _______ K 298 K 2. 450 K = _____ °C 177 °C

Pressure: • Pressure (P): measures the force/area in units of atm. , mm Hg or k. Pa 1 atm = 760 mm Hg = 101. 3 k. Pa Now you try! 1. Convert 0. 95 atm to mm Hg 722 mm. Hg 2. Convert 0. 95 atm to k. Pa 96. 2 mm. Hg 3. Convert 100. 5 k. Pa to mm. Hg 754 mm. Hg

Volume: • Volume (V): is defined using milliliters (m. L) or liters (L) 1000 m. L = 1 L Now you try! 1. 150 m. L = _____ L. 150 L 2. 34. 5 L = _____ m. L 34, 500 m. L

Atmospheric Pressure: • Atmospheric pressure: is the pressure exerted by the weight of air in the atmosphere. Traditionally it is measure using a barometer. • https: //www. youtube. com/watch? v=jm. Q 8 FWn. M 0 f. A

Does atmospheric pressure really exist? Collapsing Can experiment: • Objective- to prove that atmospheric pressure exists. Fill in the remaining part of this experiment together in class: • Background to experiment: • Observations: • Interpretation of findings: • https: //www. youtube. com/watch? v=Jso. E 4 F 2 Pb 20

Gas Laws: • Boyle’s Law Describes the inverse ( )relationship of pressure: volume P 1 V 1 = P 2 V 2

Sample calculation: Boyle’s Law • A gas has an initial pressure of 1. 0 atm and a volume of 1. 5 L. What is the new volume when the pressure is increased to 1. 2 atm? P 1 = 1. 0 atm V 1 = 1. 5 L P 2= 1. 2 atm V 2 = ? Answer: V 2= 1. 25 L

Gas Laws: • Charle’s Law Describes the direct relationship between temperature and volume ( ). V 1 = V 2 T 1 T 2 Remember: Temperature is defined by Kelvin (K = °C + 273)

Sample calculation: Charles's Law • A gas has an initial temperature of 275 K and a volume of 0. 45 L. What is the new volume when the temperature is increased to 300 K? • • V 1 = 0. 45 L T 1 = 275 K T 2 = 300 K V 2 = ? Answer : 0. 49 L

The combined gas law • The combined gas law pulls together the inverse relationship of P: V and the direct relationship of V: T • The combined gas law:

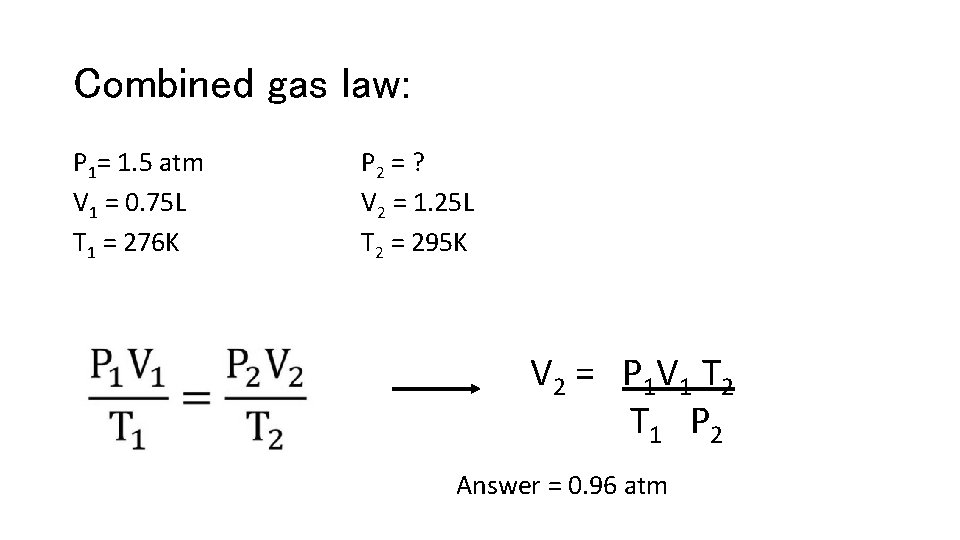
Combined gas law: P 1= 1. 5 atm V 1 = 0. 75 L T 1 = 276 K P 2 = ? V 2 = 1. 25 L T 2 = 295 K V 2 = P 1 V 1 T 2 T 1 P 2 Answer = 0. 96 atm

The Ideal Gas Law: • Includes the amount of a gas in moles (n) PV = n. RT P= pressure (atm) V= volume (L) n = moles R = gas constant: 0. 08206 L atm/ mole K T = temperature (K)

Practice problem: • Calculate the pressure of 0. 500 moles nitrogen gas in a 10. 0 L container, held at 298 K. P= ? V = 10. 0 L n = 0. 500 mole T = 298 K R = 0. 08206 L atm/mole K P = n. RT V Answer = 1. 22 atm

Gas law applications: Mountaineering Scuba Diving Hot Air balloons Tire pressure
 Giọng cùng tên là
Giọng cùng tên là Voi kéo gỗ như thế nào
Voi kéo gỗ như thế nào Thế nào là mạng điện lắp đặt kiểu nổi
Thế nào là mạng điện lắp đặt kiểu nổi Thể thơ truyền thống
Thể thơ truyền thống Lời thề hippocrates
Lời thề hippocrates đại từ thay thế
đại từ thay thế Thế nào là hệ số cao nhất
Thế nào là hệ số cao nhất Vẽ hình chiếu vuông góc của vật thể sau
Vẽ hình chiếu vuông góc của vật thể sau Ng-html
Ng-html Sự nuôi và dạy con của hổ
Sự nuôi và dạy con của hổ Dạng đột biến một nhiễm là
Dạng đột biến một nhiễm là Cách giải mật thư tọa độ
Cách giải mật thư tọa độ Vẽ hình chiếu đứng bằng cạnh của vật thể
Vẽ hình chiếu đứng bằng cạnh của vật thể Tư thế worm breton
Tư thế worm breton Quá trình desamine hóa có thể tạo ra
Quá trình desamine hóa có thể tạo ra Thiếu nhi thế giới liên hoan
Thiếu nhi thế giới liên hoan điện thế nghỉ
điện thế nghỉ Các châu lục và đại dương trên thế giới
Các châu lục và đại dương trên thế giới Thế nào là sự mỏi cơ
Thế nào là sự mỏi cơ Bổ thể
Bổ thể Sơ đồ cơ thể người
Sơ đồ cơ thể người Thế nào là số nguyên tố
Thế nào là số nguyên tố Làm thế nào để 102-1=99
Làm thế nào để 102-1=99