States of Matter 3 States of Matter n









- Slides: 18

States of Matter

3 States of Matter n Solids Most Dense ___________________ n Liquids ___________________ n Gases ___________________ Least Dense

The 4 th State of Matter n What happens if you raise the temperature to super-high levels…between 1000°C and 1, 000, 000°C? PLASMA!! § § § A plasma is an ionized gas. A plasma is a very good conductor of electricity and is affected by magnetic fields. Plasma, like gases have an indefinite shape and an indefinite volume.

Nature of Gases n n Minimal attractive forces between particles Particles have lots of energy and move freely Kinetic Molecular Theory: = a model or theory used to describe the behavior of gases 1. ) The particles in a gas are considered to be __________________________________________ 2. ) The motion of the particles in a gas is ___________________________________________ 3. ) All collisions between particles in a gas are perfectly elastic _________________________________________________________

Gas Pressure n Gas pressure = _______________________________________ **atmospheric pressure = ___________________________ measured with a barometer Units: kilopascal (k. Pa) mm. Hg atm torr 1 atm = 101. 3 k. Pa = 760 mm. Hg = 760 torr

Nature of Liquids n Particles are free to flow from one locations to another n BUT. . . The particles are attracted to each other via intermolecular forces.

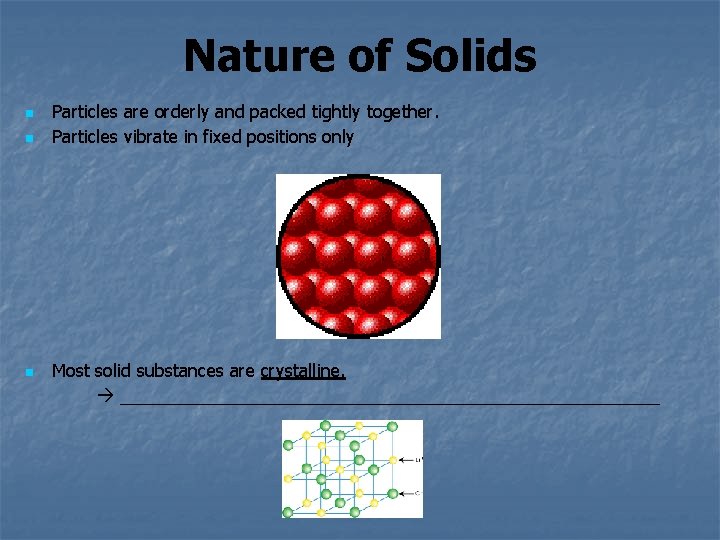
Nature of Solids n n n Particles are orderly and packed tightly together. Particles vibrate in fixed positions only Most solid substances are crystalline. ____________________________

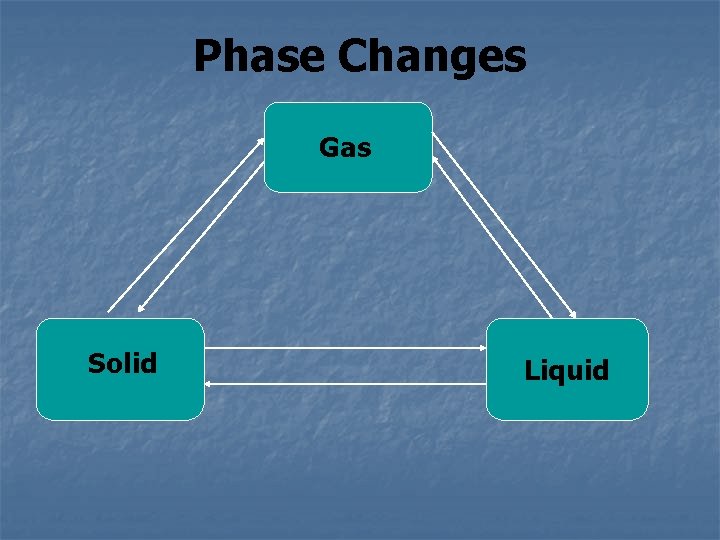
Phase Changes Gas Solid Liquid

Temperature = a measure of the average _________ of particles = ___________ **At a given temperature, the particles of all substances, regardless of physical state, have the same average kinetic energy. ** Kelvin Scale =________________________ boiling point of water = ____ K freezing pt of water = ____ K Reference Tables: absolute zero = ____ K - Table A (STP) - Table T (degrees Celcius to Kelvin

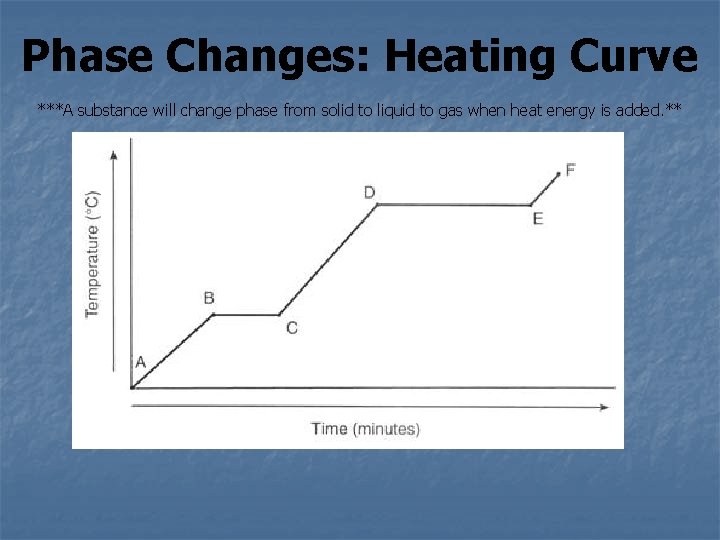
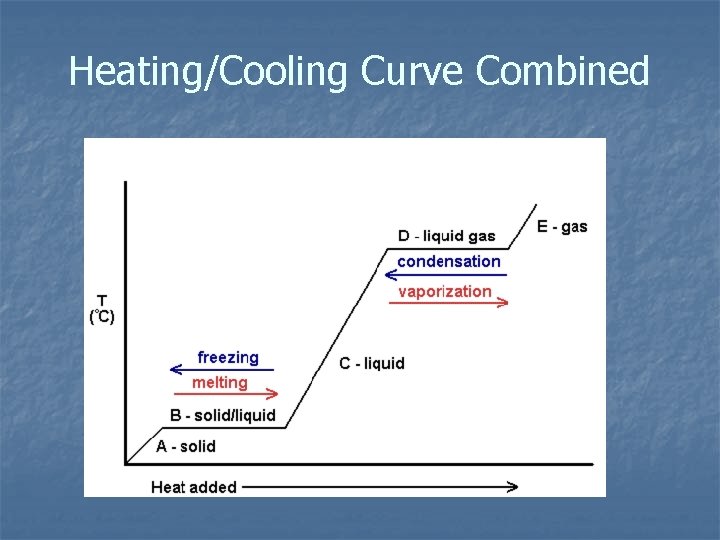
Phase Changes: Heating Curve ***A substance will change phase from solid to liquid to gas when heat energy is added. **

Phase Changes: Cooling Curve ***A substance will change phase from gas to liquid to solid when heat energy is lost. **

Heating/Cooling Curve Combined


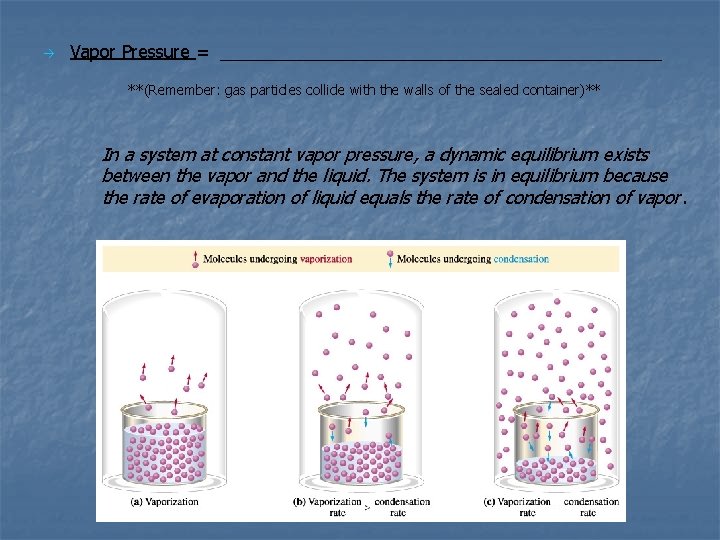
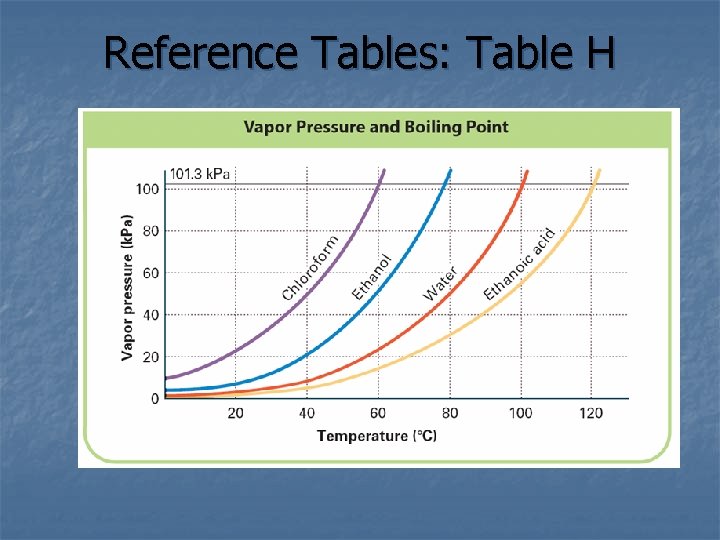
Vapor Pressure = _______________________ **(Remember: gas particles collide with the walls of the sealed container)** In a system at constant vapor pressure, a dynamic equilibrium exists between the vapor and the liquid. The system is in equilibrium because the rate of evaporation of liquid equals the rate of condensation of vapor.

Vapor Pressure & Temperature: How will temperature affect vapor pressure? _______________________ Why? _______________________________________________ **The higher the vapor pressure of a substance, the more volatile it is. volatility = _______________

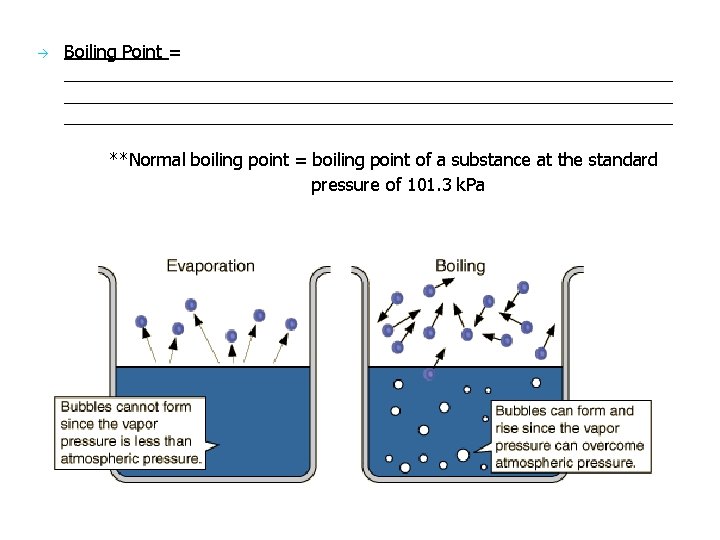
Boiling Point = ______________________________________________________________ **Normal boiling point = boiling point of a substance at the standard pressure of 101. 3 k. Pa

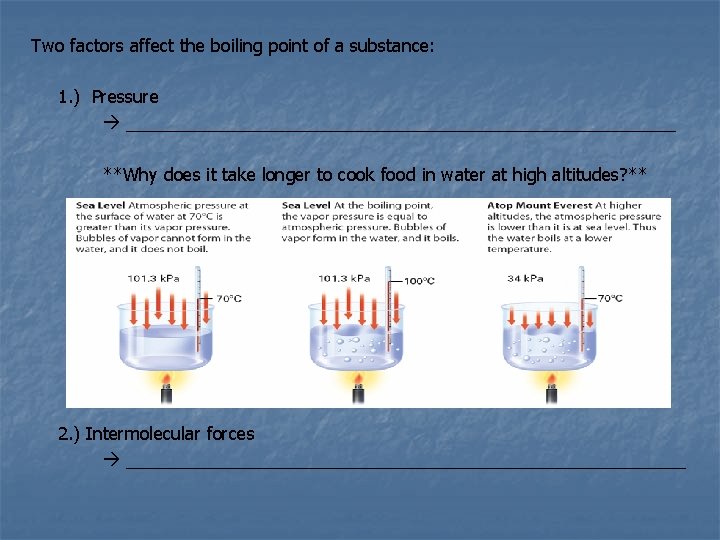
Two factors affect the boiling point of a substance: 1. ) Pressure ____________________________ **Why does it take longer to cook food in water at high altitudes? ** 2. ) Intermolecular forces _____________________________

Reference Tables: Table H