Matter Mixtures Solutions Matter and Energy The Big

Matter Mixtures Solutions

Matter and Energy

The Big Bang

Formation of Matter • • • Big Bang Time, Space, Energy – Light Quarks and Gluons (E=MC 2) 10 -6 seconds Hadrons – Protons, Neutrons 105 years Atoms - Hydrogen, Helium Billions of years – Stars, Heavier Elements, Molecules (Carbon, Oxygen, Water) 1010 years Substances – Our Sun, Earth, Rocks DNA, Proteins Life – Plants , Animals 13, 500, 000 years - Us

Four States of Matter Solid Liquid Gas Plasma

Solid A solid has a definite shape and volume. Examples of solids include ice (solid water), a bar of steel, and dry ice (solid carbon dioxide).

Liquid A liquid has a definite volume, but takes the shape of its container. Examples of liquids include water and oil.

Gas A gas has neither a definite volume nor a definite shape. Examples of gases are air, oxygen, and helium.

Plasma has neither a definite volume nor a definite shape. Plasma often is seen in ionized gases. Plasma is distinct from a gas because it possesses unique properties. Free electrical charges (not bound to atoms or ions) cause plasma to be electrically conductive. Plasma may be formed by heating and ionizing a gas. Stars are made of plasma. Lightning is plasma. You can find plasma inside fluorescent lights and neon signs.
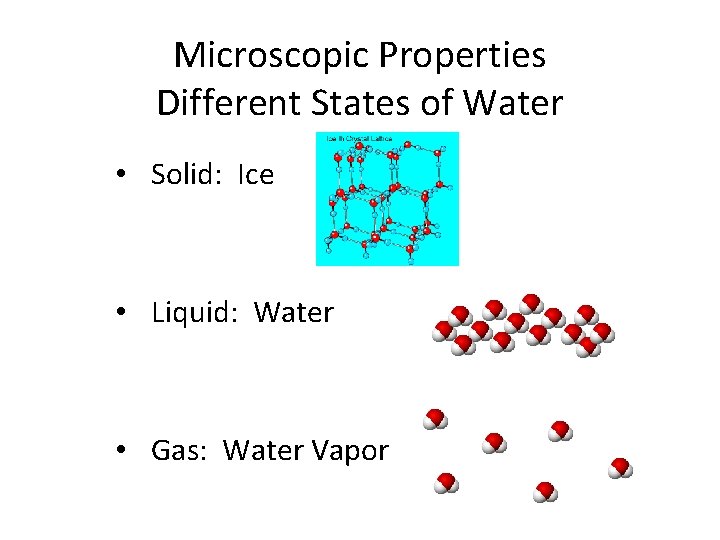
Microscopic Properties Different States of Water • Solid: Ice • Liquid: Water • Gas: Water Vapor

Mixtures • Combination of two or more substances in which the individual substances do not lose their physical or chemical properties. • Individual components can be physically separated.

Solutions A homogeneous mixture of two or more substances, which may be solids, liquids, gases, or a combination of these. Homogeneous: of uniform structure or composition throughout

Solution Components Solute: Substance dissolved in a given solution Solvent: Substance that dissolves a solute
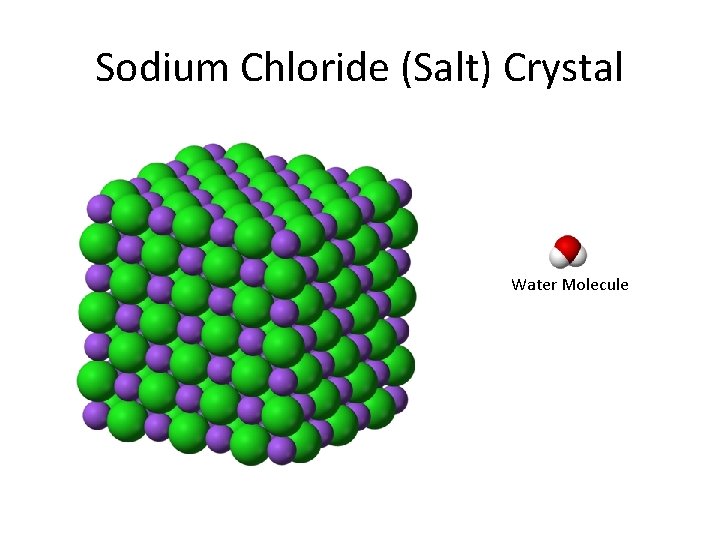
Sodium Chloride (Salt) Crystal Water Molecule

Solubility • The amount of a substance that can be dissolved in a given amount of solvent. • Exothermic process: Gives of heat • Endothermic process: Requires heat input
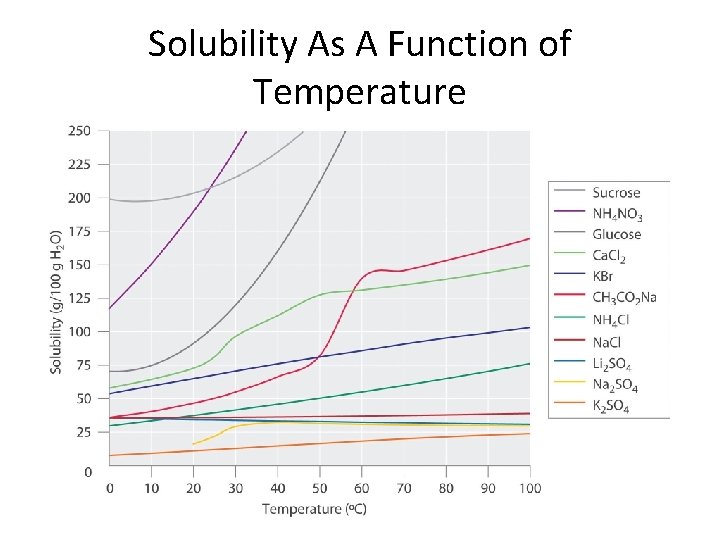
Solubility As A Function of Temperature

Solutions – Separation of Solute from Solvent
- Slides: 17