STATES OF MATTER The Four States of Matter



























- Slides: 30


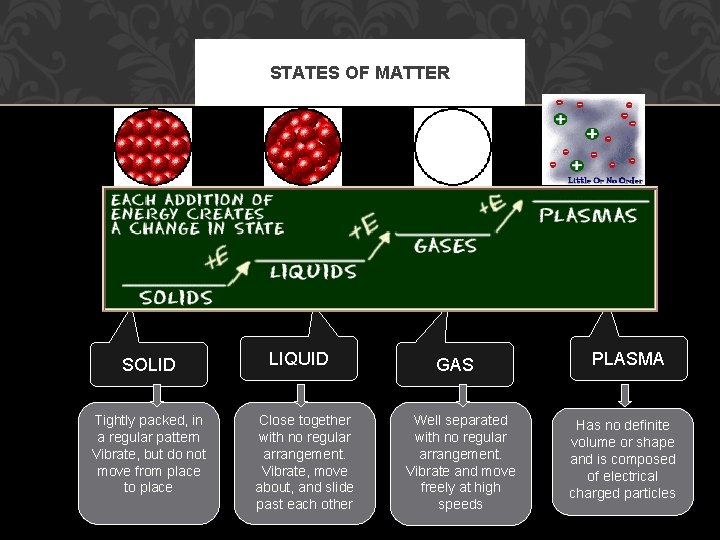
STATES OF MATTER The Four States of Matter Four States Solid Liquid Gas Plasma

STATES OF MATTER ØBased upon particle arrangement ØBased upon energy of particles ØBased upon distance between particles

KINETIC THEORY OF MATTER Matter is made up of particles which are in continual random motion.

STATES OF MATTER SOLIDS • Particles of solids are tightly packed, vibrating about a fixed position. • Solids have a defiite shape and a definite volume. Heat

STATES OF MATTER LIQUID §Particles of liquids are tightly packed, but are far enough apart to slide over one another. §Liquids have an indefinite shape and a definite volume. Heat

STATES OF MATTER GAS §Particles of gases are very far apart and move freely. §Gases have an indefinite shape and an indefinite volume. Heat

F. PHASE CHANGES – PHYSICAL Evaporation = Liquid -> Gas Condensation = Gas -> Liquid Melting = Solid -> Liquid Freezing = Liquid -> Solid Sublimation = Solid -> Gas

PHASE CHANGES Description of Phase Change Solid to liquid Term for Phase Change Melting Liquid to Freezing solid Heat Movement During Phase Change Heat goes into the solid as it melts. Heat leaves the liquid as it freezes.

PHASE CHANGES Description of Phase Change Liquid to gas Term for Phase Change Vaporization, which includes Heat goes into the boiling and liquid as it vaporizes. evaporation Gas to liquid Condensation Solid to gas Heat Movement During Phase Change Sublimation Heat leaves the gas as it condenses. Heat goes into the solid as it sublimates.

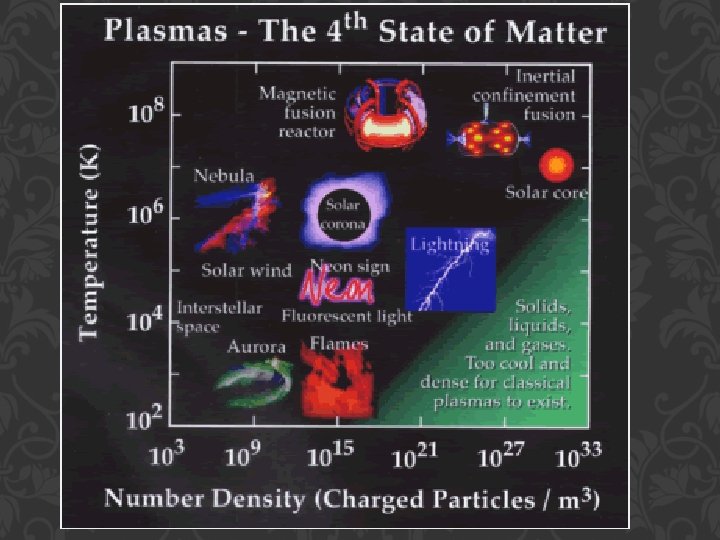
BUT WHAT HAPPENS IF YOU RAISE THE TEMPERATURE TO SUPERHIGH LEVELS… BETWEEN 1000°C AND 1, 000, 000°C ? Will everything just be a gas?

STATES OF MATTER PLASMA §A plasma is an ionized gas. §A plasma is a very good conductor of electricity and is affected by magnetic fields. §Plasmas, like gases • Plasma is the have an indefinite common state shape and an of matter indefinite volume.

STATES OF MATTER SOLID Tightly packed, in a regular pattern Vibrate, but do not move from place to place LIQUID Close together with no regular arrangement. Vibrate, move about, and slide past each other GAS Well separated with no regular arrangement. Vibrate and move freely at high speeds PLASMA Has no definite volume or shape and is composed of electrical charged particles

SOME PLACES WHERE PLASMAS ARE FOUND… 1. Flames

2. Lightning

3. Aurora (Northern Lights)

The Sun is an example of a star in its plasma state


G. CHEMICAL CHANGES Signs of a Chemical Change change in color or odor formation of a gas formation of a precipitate (solid) change in light or heat

H. PHYSICAL VS. CHEMICAL CHANGES Examples: rusting iron dissolving in water burning a log melting ice grinding spices chemical physical

LEARNING CHECK S 1 Match: (1) solid, (2) liquid, or (3) gas. ____ A. Has a definite volume, but shape of the container. ____ B. Its particles are moving rapidly. ____ C. Fills the volume of a container. ____ D. Particles are in a fixed structure. ____ E. Particles are close together, but mobile. 21

HEAT CALCULATION FOR FUSION Heat = g water x = g water 80. cal g water x 334 J g water How much heat in calories is needed to melt 15. 0 g of water? 15. 0 g water x 80. cal 22 = 1200 cal 1 g water

LEARNING CHECK CS 2 A. Why does the temperature of ice stay at 0°C while the ice melts? B. How much heat in joules is needed to melt 1. 00 g of ice of 0°C? 1) 1. 00 J 2) 100 J 3) 334 J C. How many joules of heat are released when 100. 0 g of water at 0°C freezes (0°C)? 1) 3. 34 J 2) 3340 J 3) 33, 400 J 23

SOLUTION CS 2 A. Why does the temperature of ice stay at 0°C while the ice melts? Energy goes into the change of state. B. How much heat in joules is needed to melt 1. 00 g of ice of 0°C? 3) 334 J C. How many joules of heat are released when 100. 0 g of water at 0°C freezes (0°C)? 3) 33, 400 J 24

HEAT OF VAPORIZATION Amount of heat needed to change 1 gram of liquid to gas at its boiling point Boiling (Condensing) Point of Water = 100°C Heat of Vaporization (water) = 2260 J/g 25

LEARNING CHECK CS 4 A. Ice cubes in a warm drink will 1) melt 2) freeze 3) not change B. The liquid drink _____ energy. 1) loses 2) gains change 3) does not C. The ice ______energy. 1) loses 2) gains 3) does not change D. The final temperature of the ice and liquid is 1) the same 26 2) different

SOLUTION CS 4 A. Ice cubes in a warm drink will 1) melt B. The liquid drink 1) loses energy. C. The ice 2) gains energy. D. The final temperature of the ice and liquid is 1) the same 27

I. LAW OF CONSERVATION OF MASS REVIEW FROM BALANCING CR CH. 11 Although chemical changes occur, mass is neither created nor destroyed in a chemical reaction Mass of reactants equals mass of products massreactants = massproductsts A+B C

I. CONSERVATION OF MASS In an experiment, 10. 00 g of red mercury (II) oxide powder is placed in an open flask and heated until it is converted to liquid mercury and oxygen gas. The liquid mercury has a mass of 9. 26 g. What is the mass of the oxygen formed in the reaction? GIVEN: WORK: 10. 00 g = 9. 86 g + moxygen Mercury (II) oxide mercury + oxygen Mercury (II) oxide M mercury + oxygen Mmercury(II) oxide = 10. 00 g = (10. 00 g – 9. 86 oxygen Mmercury = 9. 86 g Mmercury(II) oxide = 10. 00 g Moxygen =? Mmercury = 9. 26 Moxygen = 0. 14 g Moxygen = ? massreactants = massproducts g)

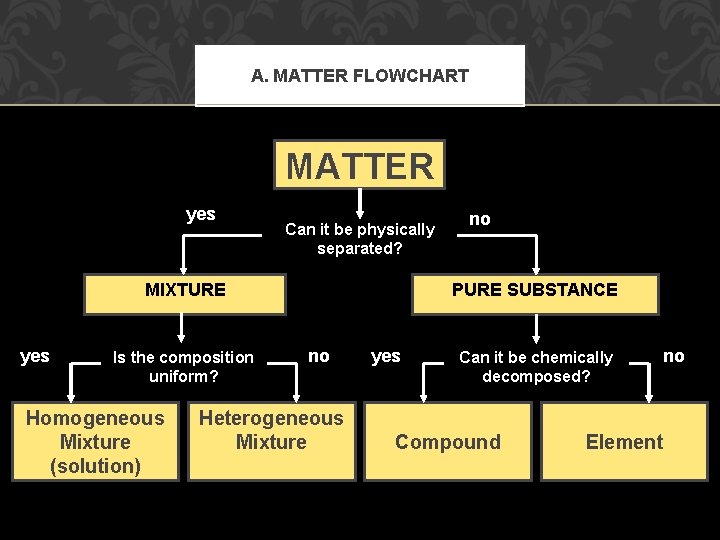
A. MATTER FLOWCHART MATTER yes Can it be physically separated? PURE SUBSTANCE MIXTURE yes Is the composition uniform? Homogeneous Mixture (solution) no no Heterogeneous Mixture yes Can it be chemically decomposed? Compound no Element