Jeopardy Freezing Melting Heat and Its Meas Vapor




































- Slides: 53

Jeopardy Freezing Melting Heat and Its Meas. Vapor Pressure Triple Point Potential Energy Q $100 Q $100 Q $200 Q $200 Q $300 Q $300 Q $400 Q $400 Q $500 Q $500 Final Jeopardy

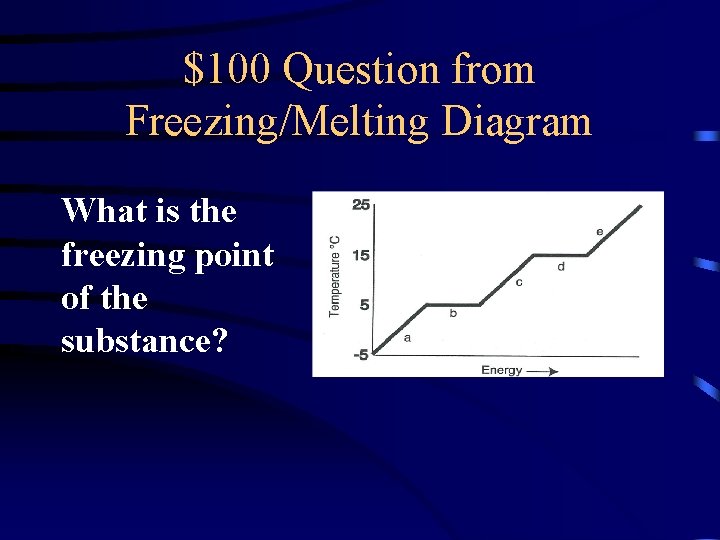
$100 Question from Freezing/Melting Diagram What is the freezing point of the substance?

$100 Answer from Freezing/Melting Diagram o 5 C

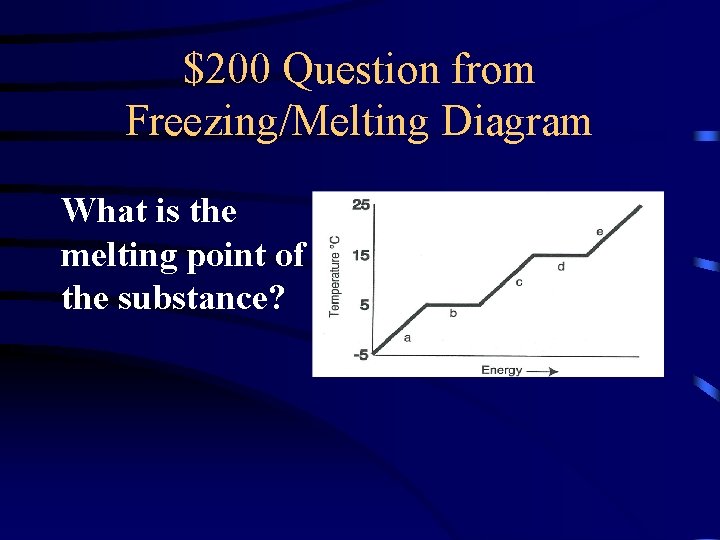
$200 Question from Freezing/Melting Diagram What is the melting point of the substance?

$200 Answer from Freezing/Melting Diagram o 5 C

$300 Question from Freezing/Melting Diagram What letter represents the range where the solid is being warmed?

$300 Answer from Freezing/Melting Diagram A

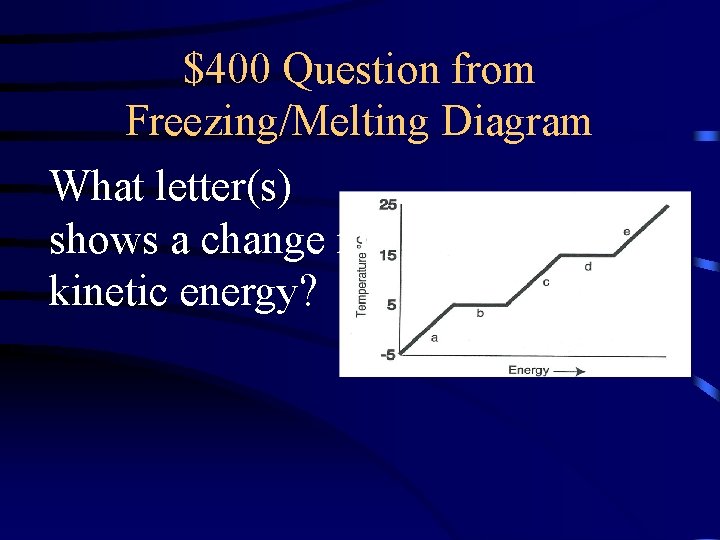
$400 Question from Freezing/Melting Diagram What letter(s) shows a change in kinetic energy?

$400 Answer from Freezing/Melting Diagram A, C, E

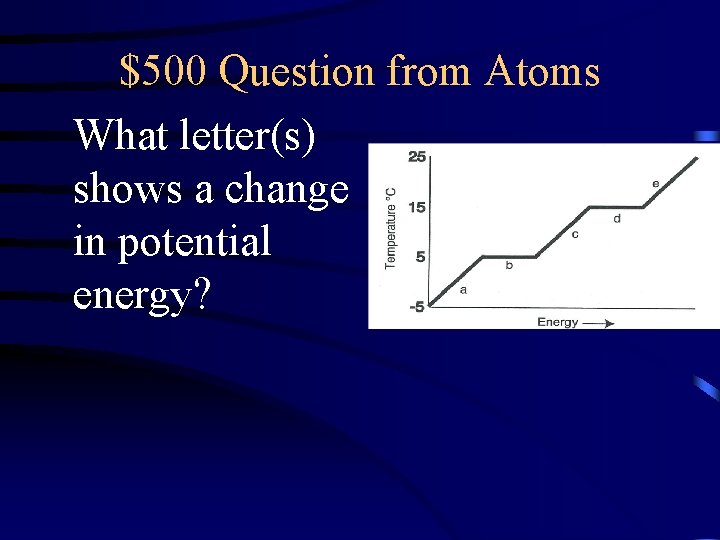
$500 Question from Atoms What letter(s) shows a change in potential energy?

$500 Answer from Freezing/Melting Diagram B, D

$100 Question from Heat and Its Measurement How many joules of heat are given off when 3. 5 g of water cool from 75 o. C to 25 o. C?

$100 Answer from Heat and Its Measurement -730 J

$200 Question from Heat and Its Measurement How many joules does it take to melt 25 g of ice at 0 o. C? .

$200 Answer from Heat and Its Measurement 8400 J

$300 Question from Heat and Its Measurement How many joules are given off when 120 g of steam condense to liquid water?

$300 Answer from Heat and Its Measurement ans: -270000 J

$400 Question from Heat and Its Measurement How many joules of heat are necessary to raise the temperature of 40 g of water from o o 20 C to 90 C?

$400 Answer from Heat and Its Measurement ans: 12000 J

$500 Question from Heat and Its Measurement How much heat is released when 75 g of water at 0 o. C freezes?

$500 Answer from Heat and Its Measurement -25000 J

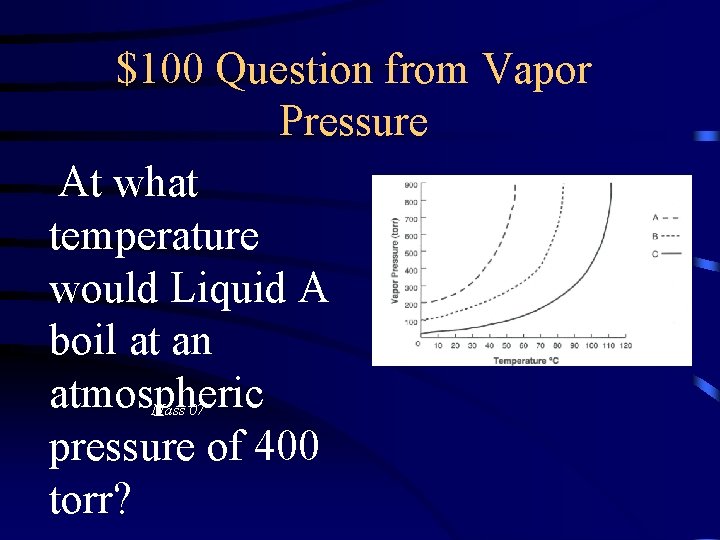
$100 Question from Vapor Pressure At what temperature would Liquid A boil at an atmospheric pressure of 400 torr? Mass 07

$100 Answer from Vapor Pressure 32 o. C

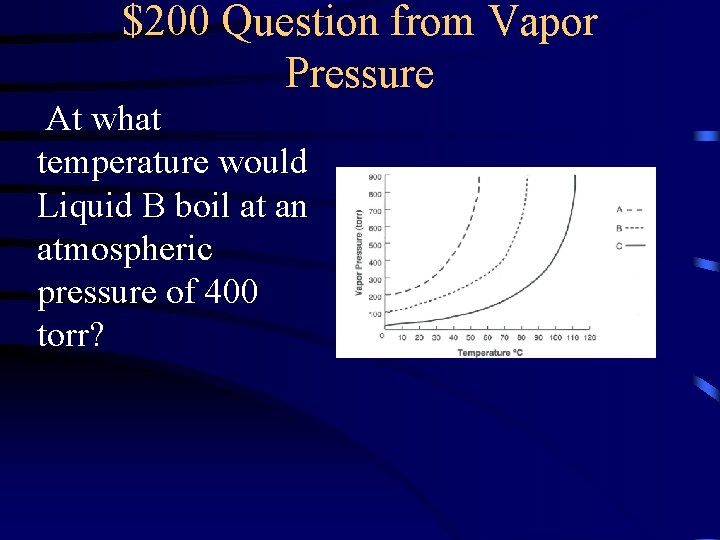
$200 Question from Vapor Pressure At what temperature would Liquid B boil at an atmospheric pressure of 400 torr?

$200 Answer from Vapor Pressure 70 o. C

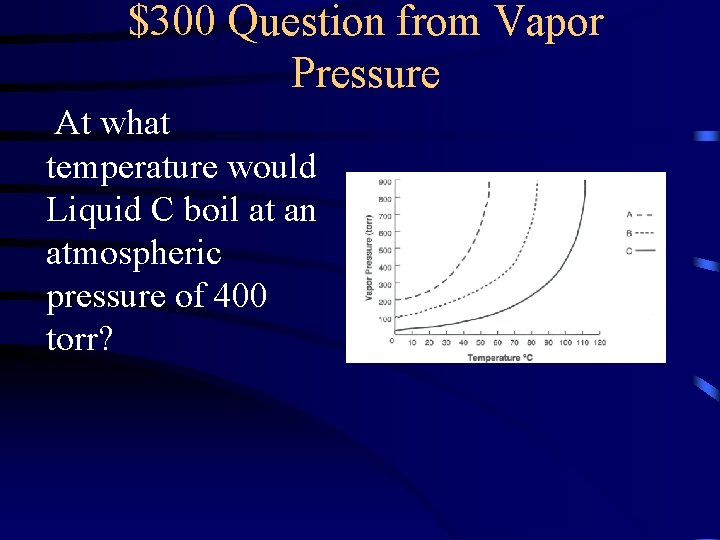
$300 Question from Vapor Pressure At what temperature would Liquid C boil at an atmospheric pressure of 400 torr?

$300 Answer from Vapor Pressure 95 o. C

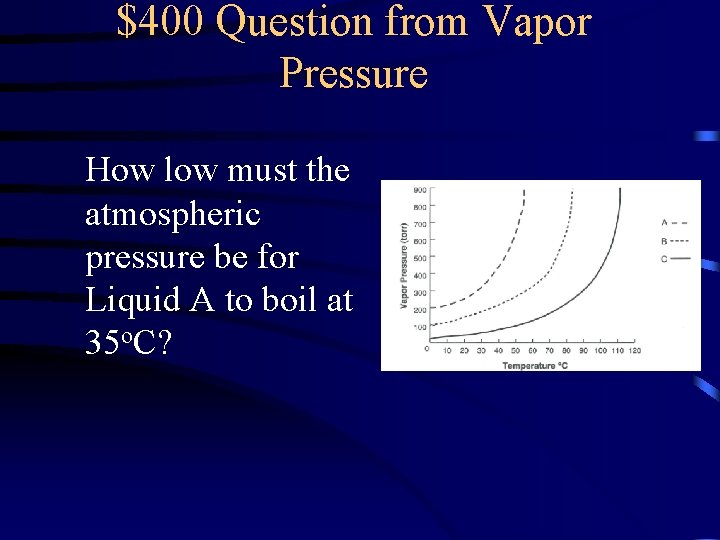
$400 Question from Vapor Pressure How low must the atmospheric pressure be for Liquid A to boil at 35 o. C?

$400 Answer from Vapor Pressure 550 torr

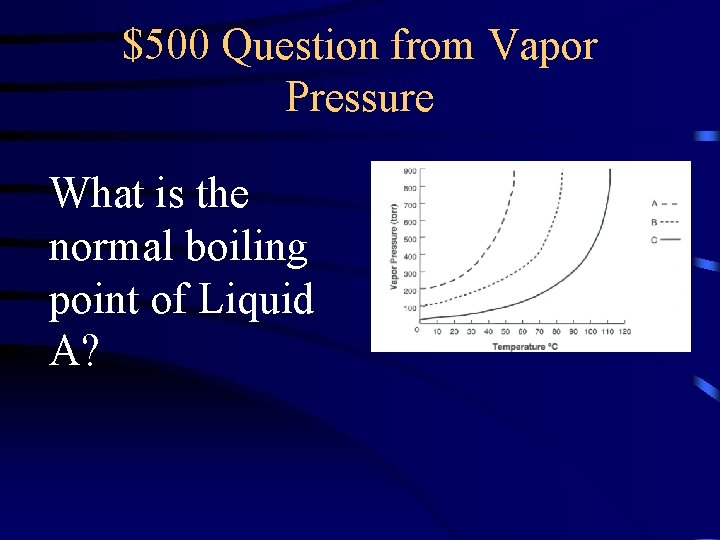
$500 Question from Vapor Pressure What is the normal boiling point of Liquid A?

$500 Answer from Vapor Pressure o 43 C

$100 Question from Triple Point Diagram What section represents the gas phase?

$100 Answer from Triple Point Diagram B

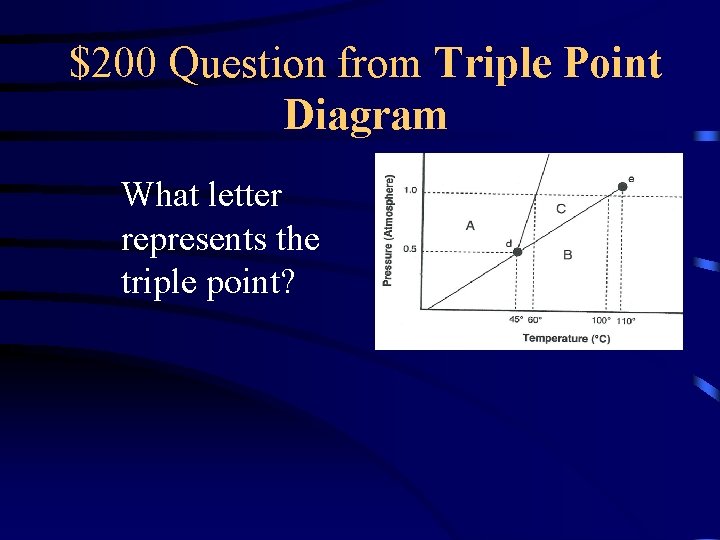
$200 Question from Triple Point Diagram What letter represents the triple point?

$200 Answer from Triple Point Diagram d

$300 Question from Triple Point Diagram What is this substance’s normal melting point?

$300 Answer from Triple Point Diagram 60 o. C

$400 Question from Triple Point Diagram What will happen to the melting point as the pressure is increased from 0. 5 to 1. 0 atm?

$400 Answer from Triple Point Diagram It increases from o 45 to 60 C

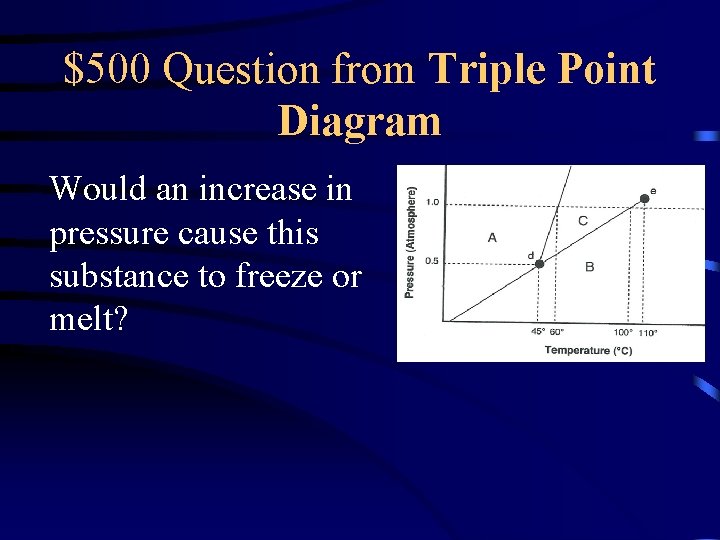
$500 Question from Triple Point Diagram Would an increase in pressure cause this substance to freeze or melt?

$500 Answer from Triple Point Diagram freeze

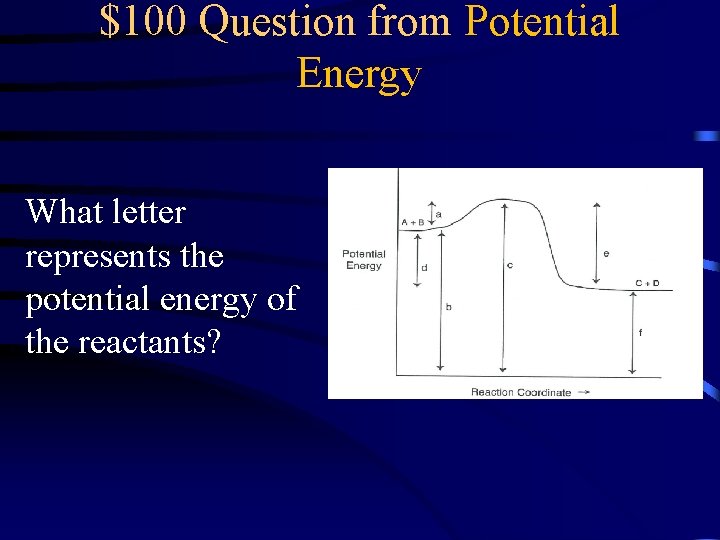
$100 Question from Potential Energy What letter represents the potential energy of the reactants?

$100 Answer from Potential Energy B

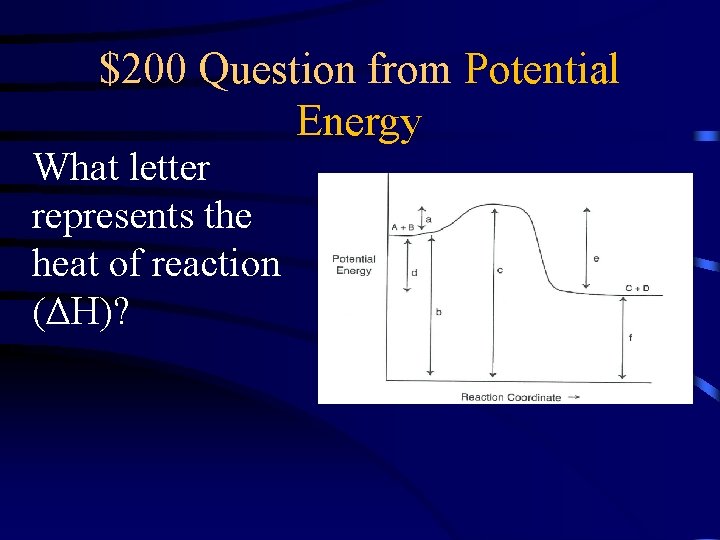
$200 Question from Potential Energy What letter represents the heat of reaction (ΔH)?

$200 Answer from Potential Energy D

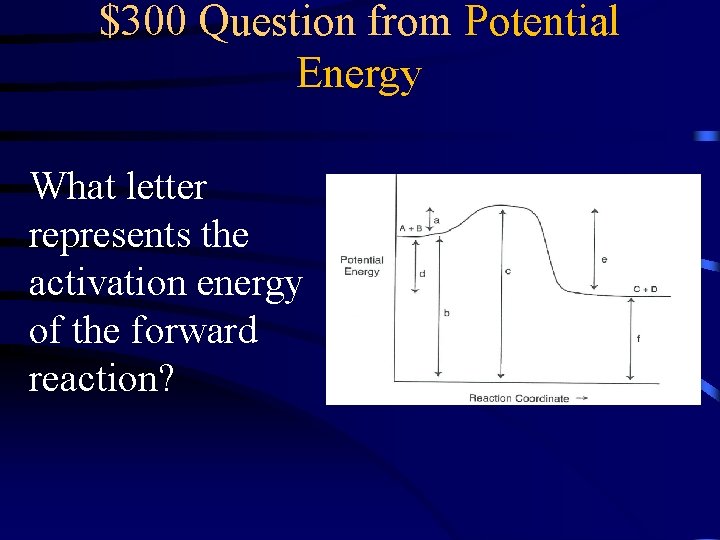
$300 Question from Potential Energy What letter represents the activation energy of the forward reaction?

$300 Answer from Potential Energy A

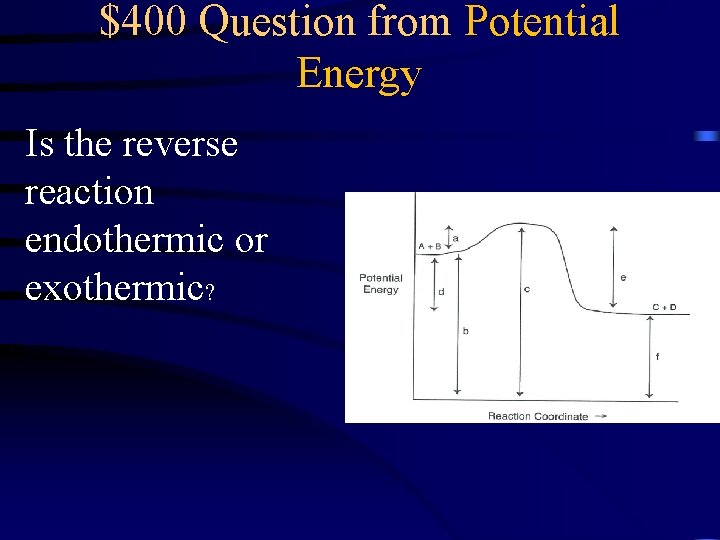
$400 Question from Potential Energy Is the reverse reaction endothermic or exothermic?

$400 Answer from Potential Energy Endothermic

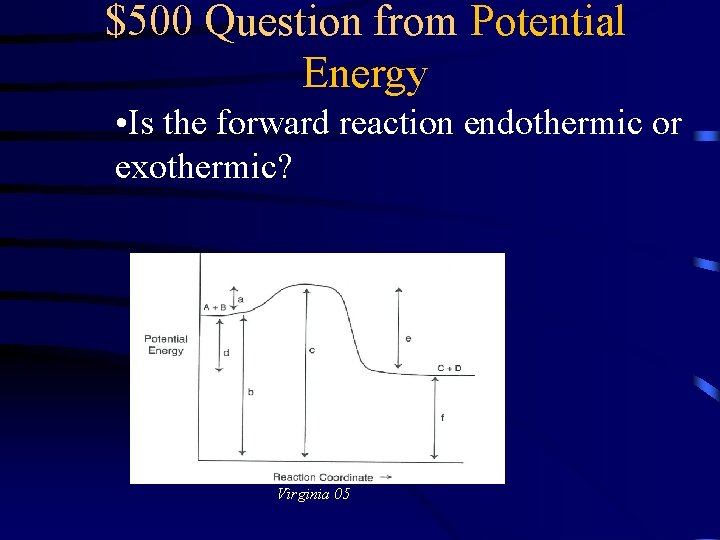
$500 Question from Potential Energy • Is the forward reaction endothermic or exothermic? Virginia 05

$500 Answer from Potential Energy Exothermic

Final Jeopardy Determine the heat required to convert 62. 0 grams of ice at -10. 3 °C to water at 100. 0 °C. The specific heat capacity of ice is 2. 05 J/g °C.

Final Jeopardy Answer Ans. 4. 79 E 4 J 47 900 J