Chapter 2 Atomic Structure Interatomic Bonding ISSUES TO

Chapter 2: Atomic Structure & Interatomic Bonding ISSUES TO ADDRESS. . . • What promotes bonding? • What types of bonds are there? • What properties are inferred from bonding? Chapter 2 - 1

Gecko’s toe pads Extremely large number of microscopically small hairs gecko-inspired bandage http: //web. mit. edu/newsoffice/2008/adhesive-0218. html http: //robotics. eecs. berkeley. edu/~ronf/Gecko/index. html Chapter 2 -

Atomic Structure (Freshman Chem. ) • atom – electrons – 9. 11 x 10 -31 kg protons -27 kg 1. 67 x 10 neutrons } • atomic number = # of protons in nucleus of atom = # of electrons of neutral species • A [=] atomic mass unit = amu = 1/12 mass of 12 C Atomic wt = wt of 6. 022 x 1023 molecules or atoms 1 amu/atom = 1 g/mol C 12. 011 H 1. 008 etc. Chapter 2 - 3
Atomic Structure • Valence electrons determine all of the following properties 1) 2) 3) 4) Chemical Electrical Thermal Optical Chapter 2 - 4

Electronic Structure • Electrons have wavelike and particulate properties. – This means that electrons are in orbitals defined by a probability. – Each orbital at discrete energy level is determined by quantum numbers. Quantum # Designation n = principal (energy level-shell) l = subsidiary (orbitals) ml = magnetic K, L, M, N, O (1, 2, 3, etc. ) s, p, d, f (0, 1, 2, 3, …, n -1) 1, 3, 5, 7 (-l to +l) ms = spin ½, -½ Chapter 2 - 5
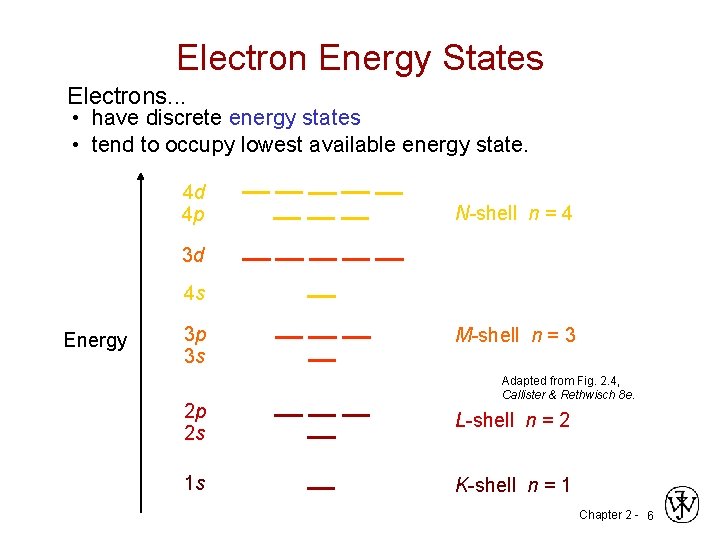
Electron Energy States Electrons. . . • have discrete energy states • tend to occupy lowest available energy state. 4 d 4 p N-shell n = 4 3 d 4 s Energy 3 p 3 s M-shell n = 3 Adapted from Fig. 2. 4, Callister & Rethwisch 8 e. 2 p 2 s L-shell n = 2 1 s K-shell n = 1 Chapter 2 - 6

SURVEY OF ELEMENTS • Most elements: Electron configuration not stable. Element Atomic # Electron configuration Hydrogen 1 1 s 1 Helium 2 1 s 2 (stable) Lithium 3 1 s 2 2 s 1 Beryllium 4 1 s 2 2 s 2 Adapted from Table 2. 2, Boron 5 1 s 2 2 p 1 Callister & Rethwisch 8 e. Carbon 6 1 s 2 2 p 2. . . Neon 10 1 s 2 2 p 6 (stable) 1 s 2 2 p 6 3 s 1 Sodium 11 Magnesium 12 1 s 2 2 p 6 3 s 2 Aluminum 13 1 s 2 2 p 6 3 s 2 3 p 1. . . Argon 18 1 s 2 2 p 6 3 s 2 3 p 6 (stable). . Krypton 36 1 s 2 2 p 6 3 s 2 3 p 6 3 d 10 4 s 2 4 p 6 (stable) • Why? Valence (outer) shell usually not filled completely. Chapter 2 - 7

Electron Configurations • Valence electrons – those in unfilled shells • Filled shells more stable • Valence electrons are most available for bonding and tend to control the chemical properties – example: C (atomic number = 6) 1 s 2 2 s 2 2 p 2 valence electrons Chapter 2 - 8
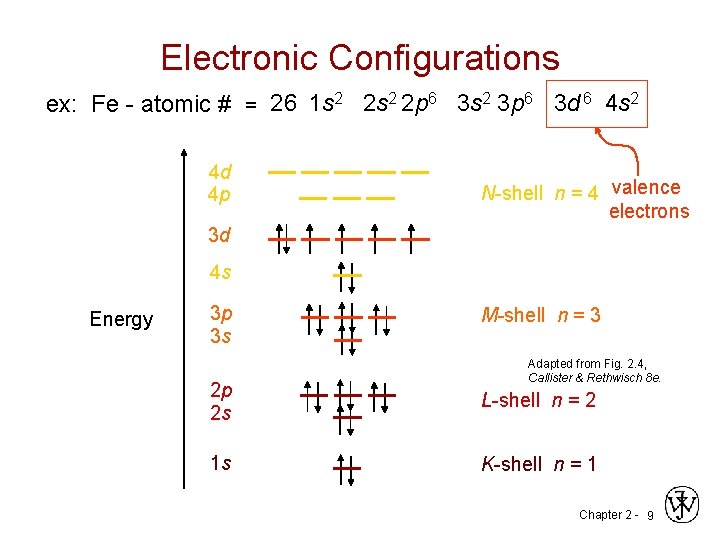
Electronic Configurations ex: Fe - atomic # = 26 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 3 d 6 4 s 2 4 d 4 p N-shell n = 4 valence electrons 3 d 4 s Energy 3 p 3 s M-shell n = 3 Adapted from Fig. 2. 4, Callister & Rethwisch 8 e. 2 p 2 s L-shell n = 2 1 s K-shell n = 1 Chapter 2 - 9

give up 1 egive up 2 egive up 3 e- • Columns: Similar Valence Structure accept 2 eaccept 1 einert gases The Periodic Table H He Li Be O F Ne Na Mg S Cl Ar K Ca Sc Rb Sr Y Cs Ba Se Br Kr Te I Adapted from Fig. 2. 6, Callister & Rethwisch 8 e. Xe Po At Rn Fr Ra Electropositive elements: Readily give up electrons to become + ions. Electronegative elements: Readily acquire electrons to become - ions. Chapter 2 - 10
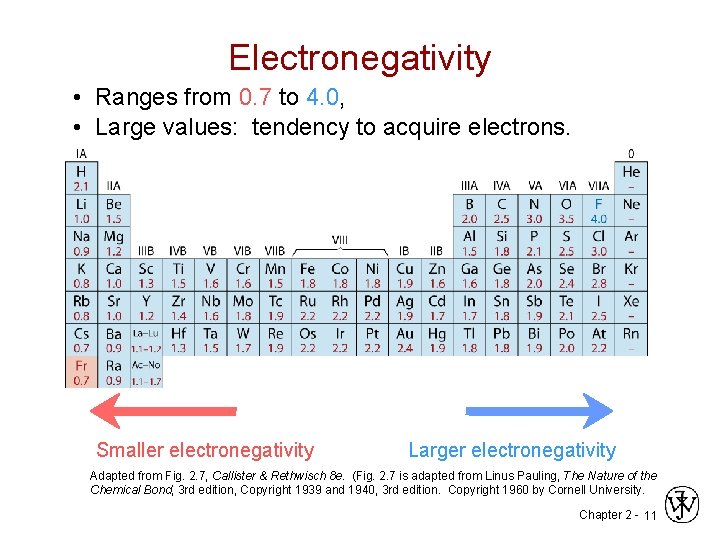
Electronegativity • Ranges from 0. 7 to 4. 0, • Large values: tendency to acquire electrons. Smaller electronegativity Larger electronegativity Adapted from Fig. 2. 7, Callister & Rethwisch 8 e. (Fig. 2. 7 is adapted from Linus Pauling, The Nature of the Chemical Bond, 3 rd edition, Copyright 1939 and 1940, 3 rd edition. Copyright 1960 by Cornell University. Chapter 2 - 11
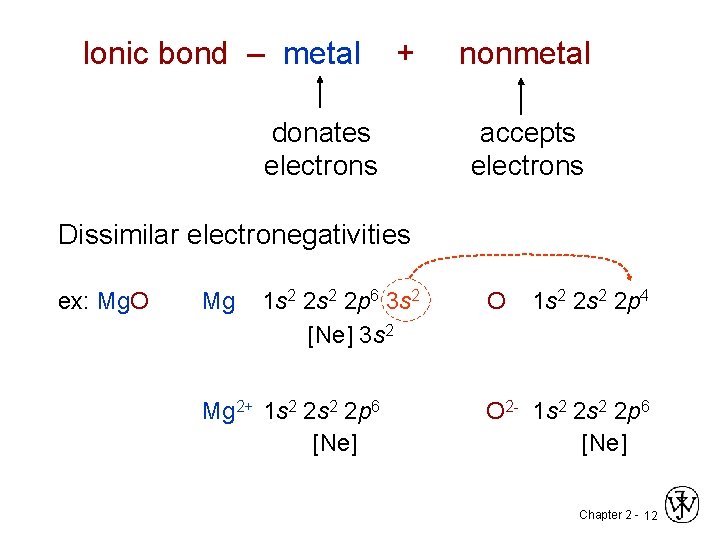
Ionic bond – metal + nonmetal donates accepts electrons Dissimilar electronegativities ex: Mg. O Mg 1 s 2 2 p 6 3 s 2 O 1 s 2 2 p 4 [Ne] 3 s 2 Mg 2+ 1 s 2 2 p 6 [Ne] O 2 - 1 s 2 2 p 6 [Ne] Chapter 2 - 12
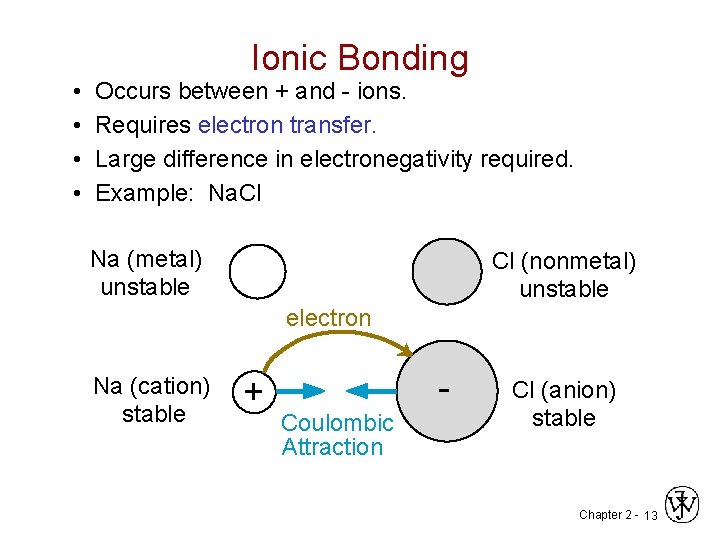
Ionic Bonding • Occurs between + and - ions. • Requires electron transfer. • Large difference in electronegativity required. • Example: Na. Cl Na (metal) unstable Cl (nonmetal) unstable electron Na (cation) stable + Coulombic Attraction - Cl (anion) stable Chapter 2 - 13
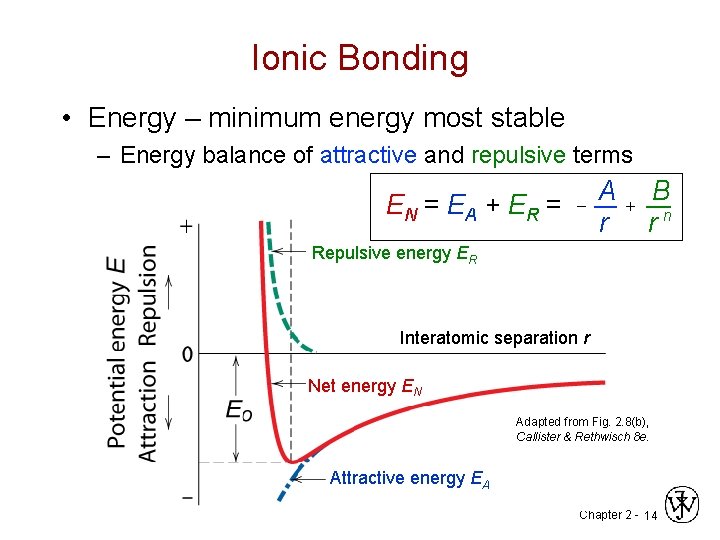
Ionic Bonding • Energy – minimum energy most stable – Energy balance of attractive and repulsive terms EN = EA + ER = - A r + B rn Repulsive energy ER Interatomic separation r Net energy EN Adapted from Fig. 2. 8(b), Callister & Rethwisch 8 e. Attractive energy EA Chapter 2 - 14
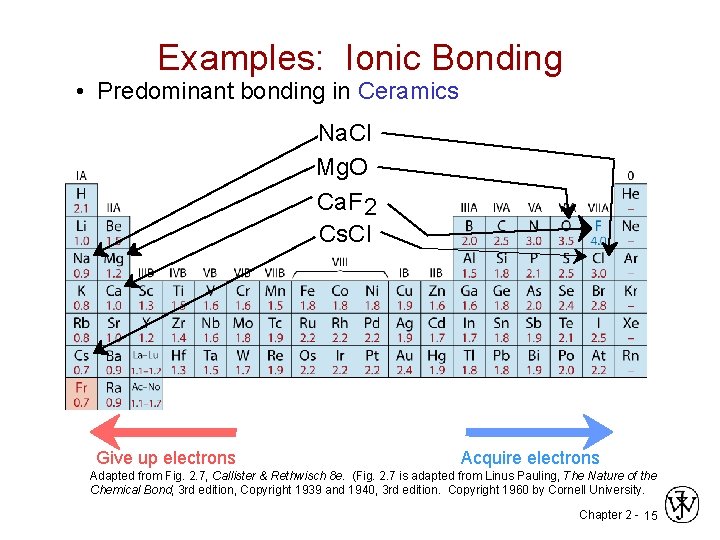
Examples: Ionic Bonding • Predominant bonding in Ceramics Na. Cl Mg. O Ca. F 2 Cs. Cl Give up electrons Acquire electrons Adapted from Fig. 2. 7, Callister & Rethwisch 8 e. (Fig. 2. 7 is adapted from Linus Pauling, The Nature of the Chemical Bond, 3 rd edition, Copyright 1939 and 1940, 3 rd edition. Copyright 1960 by Cornell University. Chapter 2 - 15
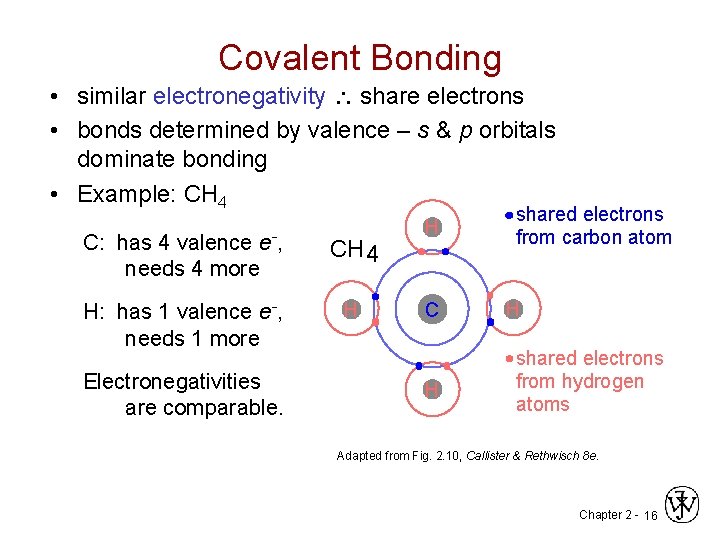
Covalent Bonding • similar electronegativity share electrons • bonds determined by valence – s & p orbitals dominate bonding • Example: CH 4 C: has 4 valence e-, needs 4 more CH 4 H: has 1 valence e-, needs 1 more H Electronegativities are comparable. H C H shared electrons from carbon atom H shared electrons from hydrogen atoms Adapted from Fig. 2. 10, Callister & Rethwisch 8 e. Chapter 2 - 16

Primary Bonding • Metallic Bond -- delocalized as electron cloud • Ionic-Covalent Mixed Bonding % ionic character = x (100%) where XA & XB are Pauling electronegativities Ex: Mg. O XMg = 1. 2 XO = 3. 5 Chapter 2 - 17
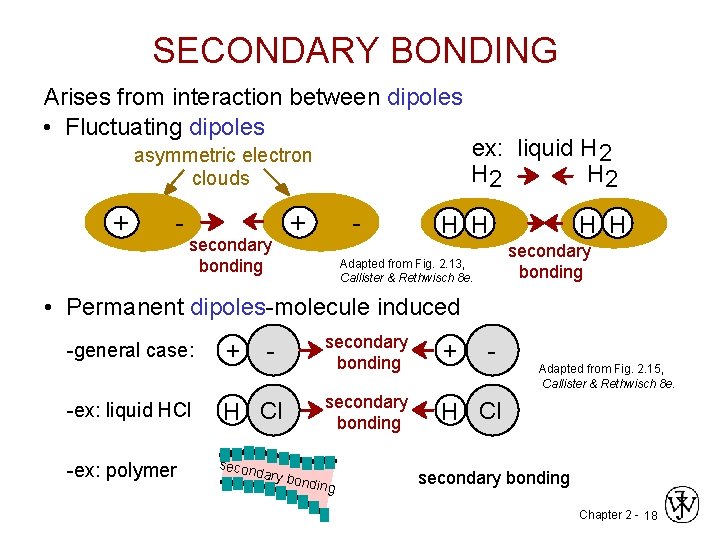
SECONDARY BONDING Arises from interaction between dipoles • Fluctuating dipoles asymmetric electron clouds + - secondary bonding + - ex: liquid H 2 H 2 H H secondary bonding Adapted from Fig. 2. 13, Callister & Rethwisch 8 e. • Permanent dipoles-molecule induced -general case: -ex: liquid HCl -ex: polymer + - H Cl secon dary b secondary bonding + secondary bonding H Cl ondin g - Adapted from Fig. 2. 15, Callister & Rethwisch 8 e. secondary bonding Chapter 2 - 18

Summary: Bonding Comments Type Bond Energy Ionic Large! Nondirectional (ceramics) Covalent Variable large-Diamond small-Bismuth Directional (semiconductors, ceramics polymer chains) Metallic Variable large-Tungsten small-Mercury Nondirectional (metals) Secondary smallest Directional inter-chain (polymer) inter-molecular Chapter 2 - 19
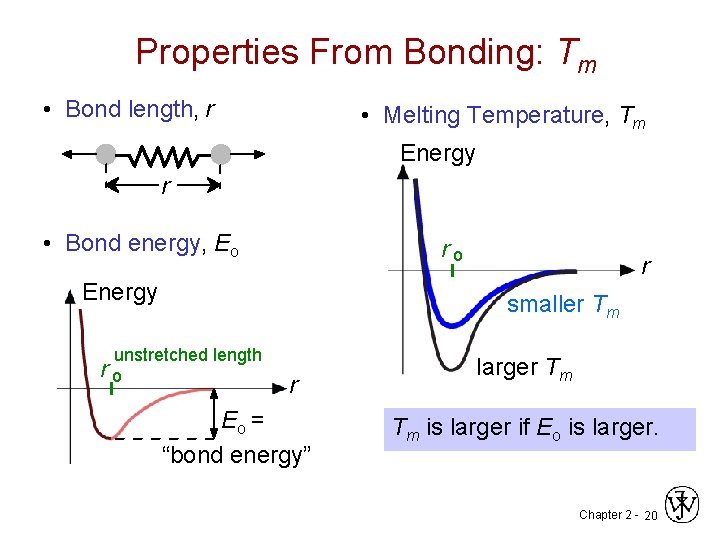
Properties From Bonding: Tm • Bond length, r • Melting Temperature, Tm Energy r • Bond energy, Eo ro Energy r smaller Tm unstretched length r o r Eo = “bond energy” larger Tm Tm is larger if Eo is larger. Chapter 2 - 20
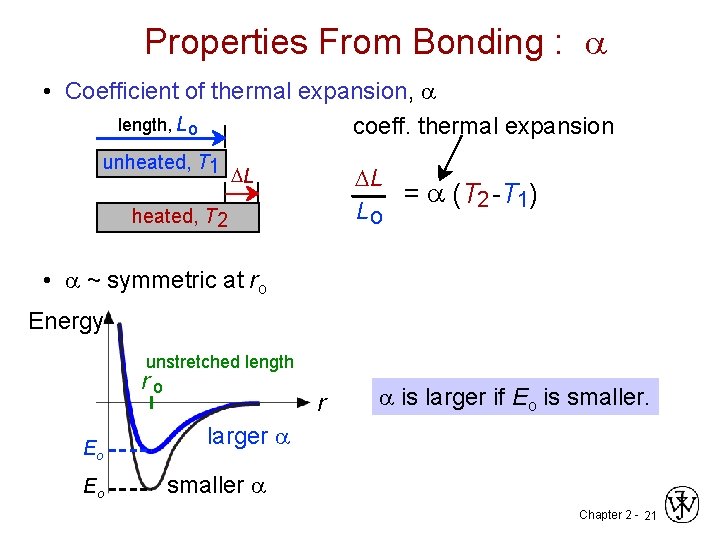
Properties From Bonding : a • Coefficient of thermal expansion, a length, L o coeff. thermal expansion unheated, T 1 DL DL = a (T 2 -T 1) Lo heated, T 2 • a ~ symmetric at ro Energy unstretched length ro Eo Eo r a is larger if Eo is smaller. larger a smaller a Chapter 2 - 21
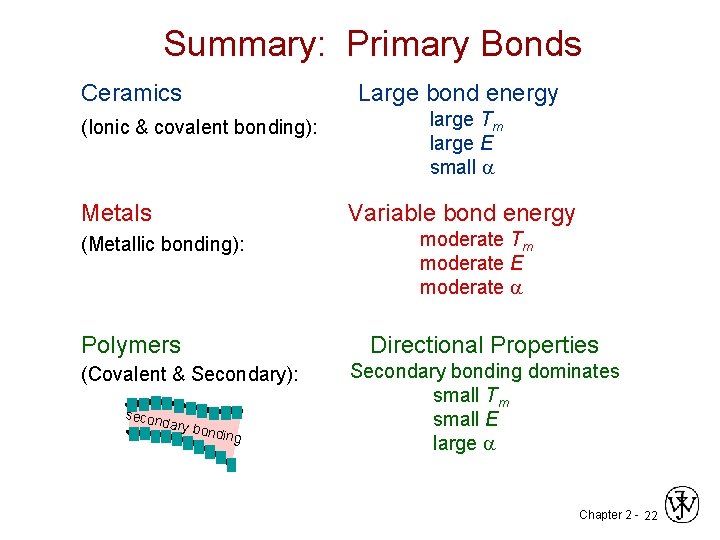
Summary: Primary Bonds Ceramics Large bond energy (Ionic & covalent bonding): Metals large Tm large E small a Variable bond energy (Metallic bonding): moderate Tm moderate E moderate a Polymers Directional Properties (Covalent & Secondary): Secondary bonding dominates small Tm small E large a secon dary b o nding Chapter 2 - 22

Water (Its Volume Expansion Upon Freezing) Chapter 2 - 23

ANNOUNCEMENTS Reading: Chapter-2 Self-Assessment Exercise for Chap-2 Homework for Chap-2: 1, 4, 7, 8, 14, 19. Chapter 2 - 24
- Slides: 24