Biochemistry By Alex Caroline Reed Chemistry Element a

Biochemistry By: Alex & Caroline Reed

Chemistry ● Element : a substance that cannot be broken down into other substances by chemical reactions. ● Elements form compounds , which consist of 2 or more different elements combined in a fixed ratio. ○ These compounds have characteristics that differ from those of the element ■ Ex. Na (soft metal, explodes in water) + Cl (poisonous gas) = Na. Cl (table salt)

Structure = Function ● An element’s properties depend on the structure of its atoms. ● An atom is the smallest unit of matter that still retains the properties of an element. ● They are composed of subatomic particles including… ○ ○ Neutrons (no electrical charge) Protons (positive charge) Electrons (negative charge) Both protons and neutrons reside within a very reside in the electron cloud. dense nucleus while the electrons

More Vocab Atomic Number & Atomic Mass ● An element's atomic number is the number or protons in the nucleus. ● An element’s mass number is the sum of the protons and the neutrons in the nucleus. ● Atomic mas s, the atom’s total mass, can be approximated by the mass number. ○ It is actually represented by an average molecular mass that is based on the abundance of various isotopes.

Isotopes ● All atoms of an element have the same number of protons but may differ in the number of neutrons. ● Isotopes are two atoms of the same element that differ in the number of neutrons. ● Radioactive Isotopes are isotopes that decay spontaneously, giving off particles and energy. ○ These radioactive isotopes are used for… ■ Dating fossils (C-14) ■ Diagnosing medical disorders

Electrons & Electron Clouds ● Energy is the capacity to cause change, perhaps by doing work ● Potential energy is energy that matter has because of its location or structure. ○ An atom’s electrons differ in amounts of potential energy. ● An electron’s state of potential energy is called its energy level or Electron Shell.
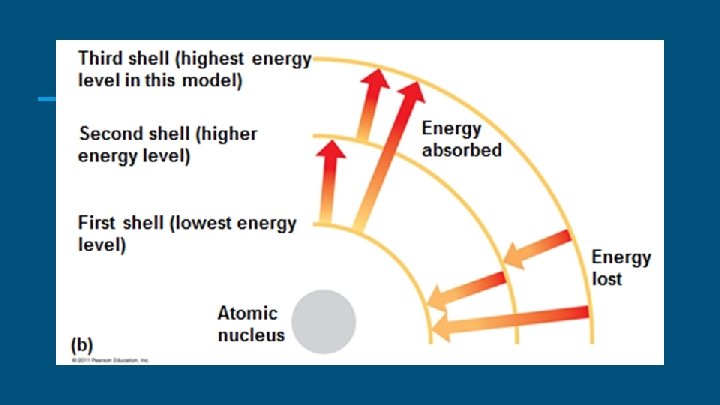

Valence Shells ● The distribution of electrons throughout an atom’s electron energy levels and sublevels determines the chemical behavior of the atom. ● Valence electrons are those in the outermost energy level, or valence sublevel ● Elements with a full valence shell are chemically inactive. ● Atoms with incomplete valence shells can share or transfer valence electrons with certain other atoms. ● These interactions usually result in atoms staying close together, held by attractions called chemical bonds.

Covalent Bonds ● A covalent bond is the sharing of valence electrons by two atoms. ● These electrons count as part of each atom’s valence shell ● A covalent bond is formed between shared pairs of electrons: ○ ○ ○ 1 pair - a single bond 2 pairs - a double bond 3 pairs - a triple bond

Electronegativity ● Atoms is a molecule attracts electrons to varying degrees. ● Electronegativity is an atom’s attraction for the electrons in a covalent bond. ● The more electronegative an atom, the more strongly it pulls shared electrons toward itself.
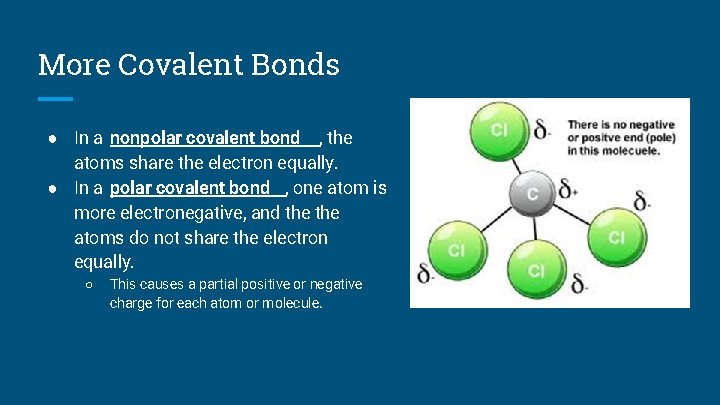
More Covalent Bonds ● In a nonpolar covalent bond , the atoms share the electron equally. ● In a polar covalent bond , one atom is more electronegative, and the atoms do not share the electron equally. ○ This causes a partial positive or negative charge for each atom or molecule.

Ionic Bonds ● Atoms sometimes strip electrons from their bonding partners. ○ Ex. The transfer of electrons between sodium and chlorine. ● A charged atom (or molecule) is called an ○ ○ ion. A cation is a positively charged ion An anion is a negatively charged ion ● An ionic bond is an attraction between an anion and a cation. ○ Compounds formed by ionic bonds are called ionic compounds, or salts.

Intermolecular Forces ● Intermolecular forces : forces of electrostatic attraction “between molecules” ○ Not the same as sharing a pair of electrons within a molecule ● IMFs are intermolecular(between the molecules) whereas chemical bonds are intramolecular (wi thin the molecule). ● London Dispersion Forces (LDFs) - every molecule has these since every molecule has moving valence electrons. The electrons are essential in constant motion and are not always evenly distributed. ● If the electrons are not evenly dispersed, the molecule is now referred to as a dipole.

Dipole-Dipole Forces ● In dipole-induced dipole , there is a permanent dipole (electronegativity difference enough to make a permanent (+) and (-) end of the molecule) that induces a nonpolar molecule to become a dipole. ● Now the two are more attracted to each other then they were before the induction occurred.
Hydrogen Bonding ● A special case of dipole-dipole IMFs. ● Hydrogen bonding occurs when the H of one molecule is attracted to a highly electronegative molecule on an adjacent molecule. ● Makes molecule more attracted to each other, and thus more energy is required to separate them. ● This results in… ○ Higher MP, BP, heat of vaporization and enhanced solubility of substances.
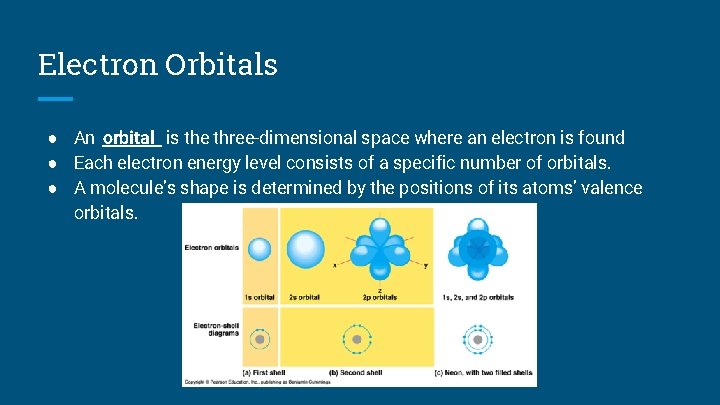
Electron Orbitals ● An orbital is the three-dimensional space where an electron is found ● Each electron energy level consists of a specific number of orbitals. ● A molecule’s shape is determined by the positions of its atoms’ valence orbitals.

Chemical Reactions Let’s get some things straight before we continue… 1. Energy must be added to a system to BREAK a chemical bond. 2. Energy is released when chemical bonds are MADE. ● Chemical reactions are the making and breaking of chemical bonds. ● The starting molecules of the reaction are known as the reactants and the final molecules of the reaction are known as the products.

Chemical Equilibrium ● All chemical reactions are REVERSIBLE!!! ○ The products of the forward reaction become the reactants for the reverse reaction. ● Chemical equilibrium is reached when the rate of the forward reaction is equal to the rate of the reverse reaction. ○ Does NOT mean “equal” amounts of reactants and products, but rather that their concentrations have stabilized in a constant ratio.

Water ● The abundance of water is the main reason that Earth is habitable. ● There are FOUR emergent properties of water that contribute to Earth’s sustainability for life: 1. Water’s cohesive & adhesive behavior 2. Water’s ability to moderate temperature 3. Water expansion upon freezing 4. Water’s versatility as a solvent.

Cohesion and Adhesion ● Cohesion: when water molecules stick to ● Adhesion : when water molecules stick to substance. ○ each other. some other type of Ex. plant cell walls. ● Surface tension: a measure of how hard it is to break the surface of a liquid. ○ This is caused by hydrogen bonding between water molecules.

Moderation of Temperature ● Water has a high heat capacity. ● Heat capacity is the amount of heat required to raise 1 gram of water by 1 o Celsius (1 calorie = 4. 184 J) ● This means that water can absorb large quantities of heat without much change in its own temperature. ● As a result, it is a good thermo regulator. ● This too is a result of hydrogen bonding.

Evaporation ● Evaporation is the transformation of a substance from liquid to vapor. ● Heat of vaporization is the heat a liquid must absorb for 1 gram to be converted to vapor. ● As a liquid evaporates, its remaining surface cools, a process called evaporative cooling. ● This evaporative cooling helps stabilize temperatures in organisms and bodies of water.

Expansion Upon Freezing ● Ice floats in liquid water because hydrogen bonds in ice are more “ordered” forming a hexagonal shape with a hole in the middle, making ice less dense. Ice: stable hydrogen bonds Liquid water: transient hydrogen bonds

Water’s Versatility as a Solvent ● Water is a versatile solvent due to its polarity , which allows it to form hydrogen bonds easily. ● When an ionic compound is dissolved in water, each ion is surrounded by a sphere of water molecules called a hydration shell. ● Water can also dissolve compounds made of nonionic polar molecules .

Vocabulary A solution is a liquid that is a homogeneous mixture of substances A solvent is the dissolving agent of a solution The solute is the substance that is dissolved An aqueous solution is one in which water is the solvent. A hydrophilic substance is one that has an affinity for water. A hydrophobic substance is one that does not have an affinity for water (ex. oil) ● A colloid is a stable substance of fine particles in liquid (ex. milk) ● ● ●

Acids, Bases, and Buffers ● Changes in the concentration of H+ and OH- ions can greatly affect the chemistry of a cell. ● An acid , is any substance that increases the H+ of a solution. ● A base , is any substance that reduces the H+ concentration of a solution. ● Buffers are substances that RESIST changes in concentrations of H+ and OH- in a solution and therefore RESIST changes in p. H.

p. H Calculations ● In any aqueous solution at 25°C the product of H + and OH – is constant and can be written as the autoionization constant of water [H +][OH – ] = 10– 14 ● The p. H of a solution is defined by the negative logarithm of H concentration, written as. . . p. H = –log [H +] +
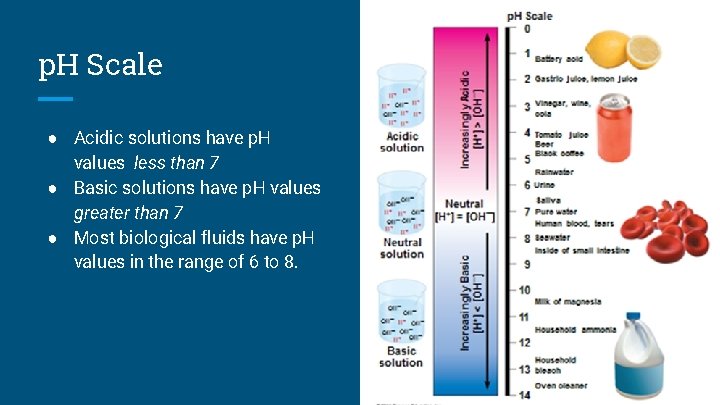
p. H Scale ● Acidic solutions have p. H values less than 7 ● Basic solutions have p. H values greater than 7 ● Most biological fluids have p. H values in the range of 6 to 8.

Carbon ● Living organisms consist mostly of carbon-based compounds. ● Proteins, DNA, carbohydrates, and other molecules that distinguish living matter are all composed of carbon compounds. ● Organic Chemistry

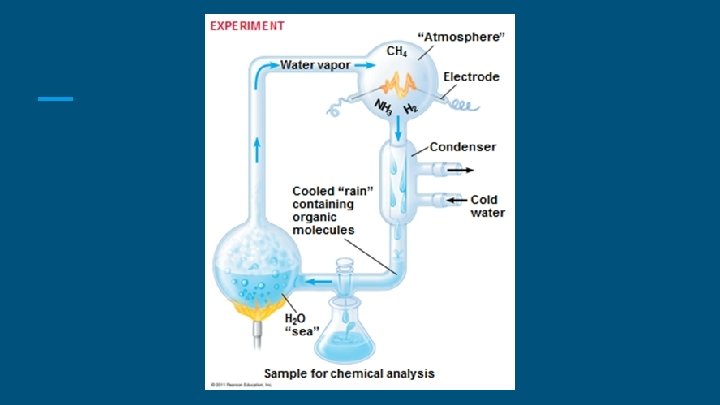
The Miller-Urey Experiment ● Demonstrated the abiotic synthesis of organic compounds. ● Water (H 2 O), methane (CH 4), ammonia (NH 3), and hydrogen (H 2) were all sealed inside a sterile array of glass tubes and flasks connected in a loop, with one flask half-full of liquid water and another flask containing a pair of electrodes. ● The liquid water was heated to induce evaporation, sparks were fired between the electrodes to simulate lightning through the atmosphere and water vapor, and then the atmosphere was cooled again so that the water could condense and trickle back into the first flask in a continuous cycle.

The Miller-Urey Experiment ● Within a day, the mixture had turned pink in color, and at the end of two weeks of continuous operation, Miller and Urey observed that as much as 10– 15% of the carbon within the system was now in the form of organic compounds. ● Two percent of the carbon had formed amino acids that are used to make proteins in living cells, with glycine as the most abundant. Nucleic acids were not formed within the reaction. But the common 20 amino acids were formed, in various concentrations.

Carbon Structure ● Carbon has four valence electrons, and therefore, can form four covalent bonds with a variety of atoms. ● This allows carbon to create large, complex molecules. ● Often takes on a tetrahedral shape.
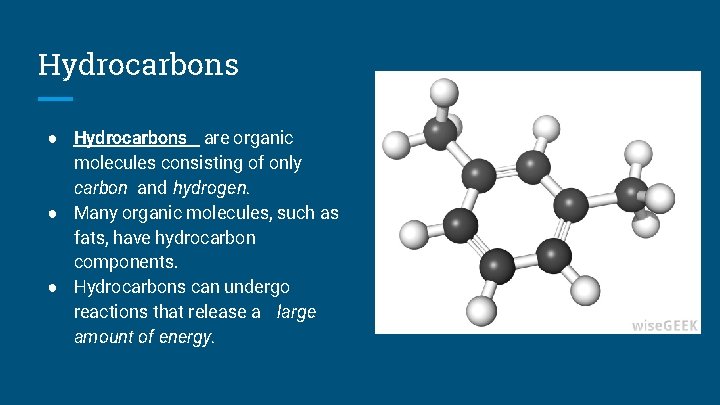
Hydrocarbons ● Hydrocarbons are organic molecules consisting of only carbon and hydrogen. ● Many organic molecules, such as fats, have hydrocarbon components. ● Hydrocarbons can undergo reactions that release a large amount of energy.
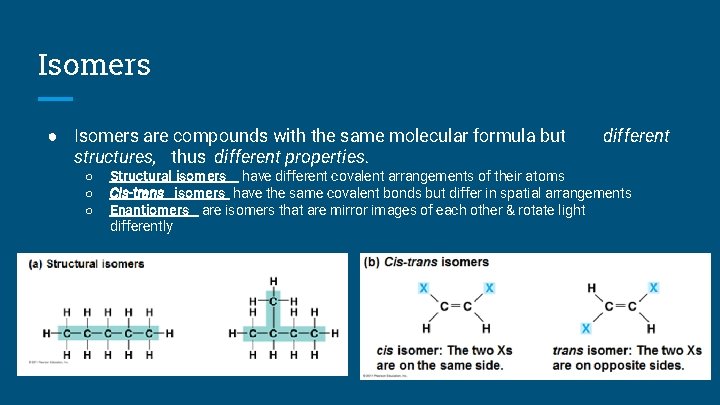
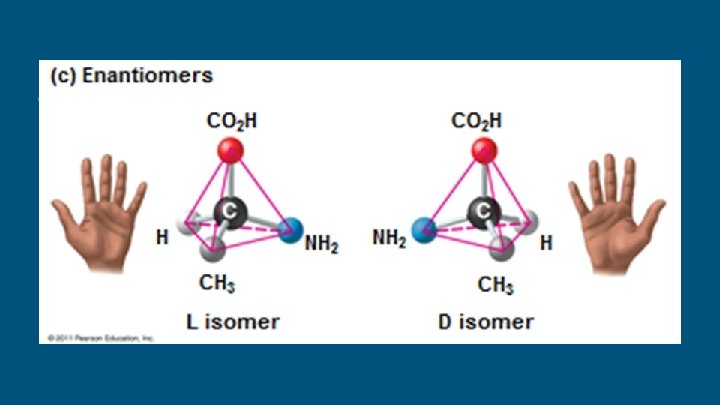
Isomers ● Isomers are compounds with the same molecular formula but structures, thus different properties. ○ ○ ○ different Structural isomers have different covalent arrangements of their atoms Cis-trans isomers have the same covalent bonds but differ in spatial arrangements Enantiomers are isomers that are mirror images of each other & rotate light differently
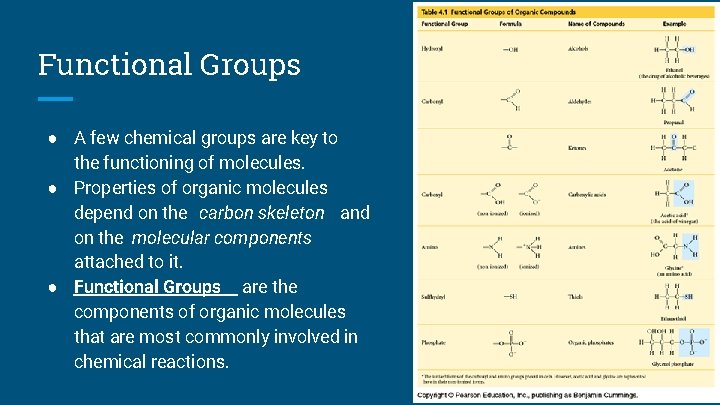
Functional Groups ● A few chemical groups are key to the functioning of molecules. ● Properties of organic molecules depend on the carbon skeleton and on the molecular components attached to it. ● Functional Groups are the components of organic molecules that are most commonly involved in chemical reactions.

BIOMOLECULES There are 4 major biomolecules ● ● Carbohydrates Lipids Proteins Nucleic acids

Building blocks of macromolecules ● Macromolecules are composed of various types of monomers ● A polymer is created from many monomers Only three biomolecules have macromolecules 1. Carbohydrates 2. Proteins 3. Nucleic acids
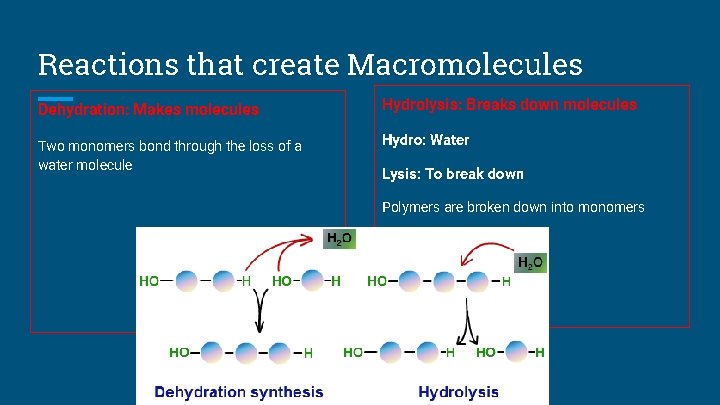
Reactions that create Macromolecules Dehydration: Makes molecules Hydrolysis: Breaks down molecules Two monomers bond through the loss of a water molecule Hydro: Water Lysis: To break down Polymers are broken down into monomers

Carbohydrates Monosaccharides: Simple sugars (Glucose, Fructose) 1. Have formulas that are multiples of CH 2 O 2. Classified by: a. Location of carbonyl group b. Number of carbons
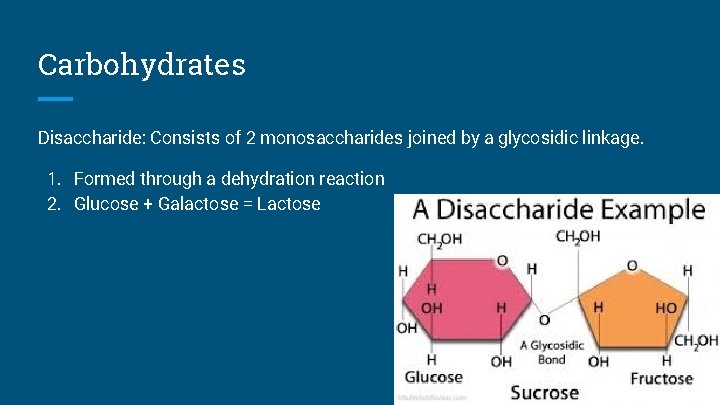
Carbohydrates Disaccharide: Consists of 2 monosaccharides joined by a glycosidic linkage. 1. Formed through a dehydration reaction 2. Glucose + Galactose = Lactose

Carbohydrates Polysaccharides: More than two monosaccharides combined Four examples: ● ● Starch Glycogen Cellulose Chitin
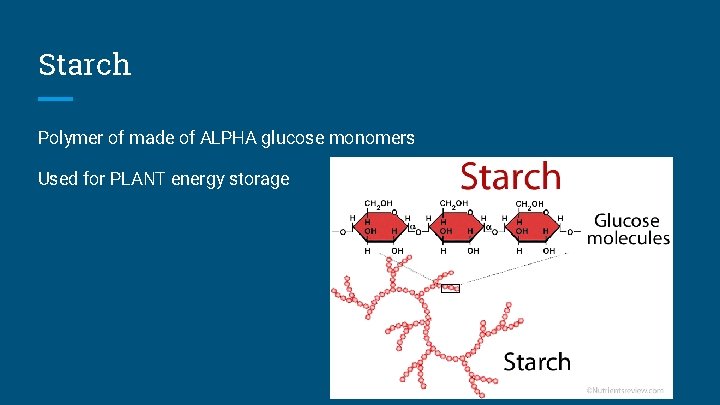
Starch Polymer of made of ALPHA glucose monomers Used for PLANT energy storage
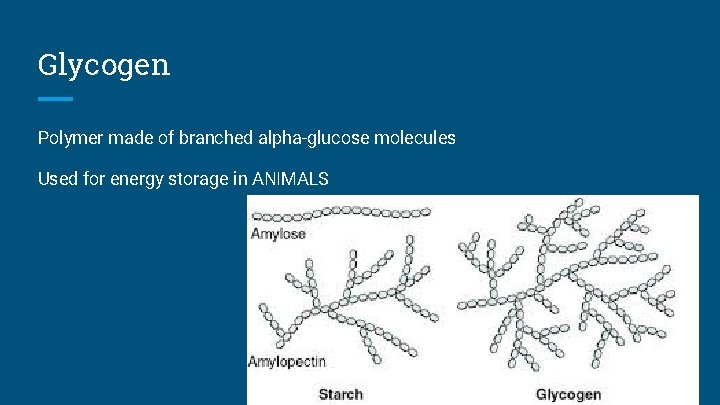
Glycogen Polymer made of branched alpha-glucose molecules Used for energy storage in ANIMALS
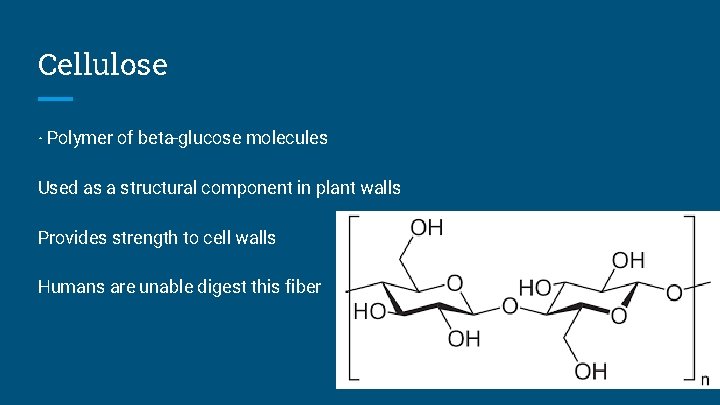
Cellulose · Polymer of beta-glucose molecules Used as a structural component in plant walls Provides strength to cell walls Humans are unable digest this fiber

Chitin Very similar to cellulose Used for the exoskeleton of insects, and gives support to fungi

Lipids Hydrophobic molecule ● Only molecule that does NOT form polymers ● Consist of mostly hydrocarbons: Form nonpolar covalent bonds 1. Fats 2. Phospholipids 3. Steroids

Lipids: Uses Long term energy source: Fats Insoluble in water 3 groups Triglycerides Phospholipids Steroids
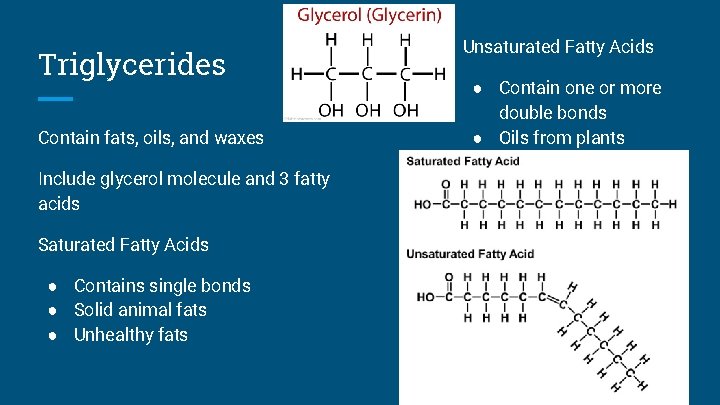
Triglycerides Contain fats, oils, and waxes Include glycerol molecule and 3 fatty acids Saturated Fatty Acids ● Contains single bonds ● Solid animal fats ● Unhealthy fats Unsaturated Fatty Acids ● Contain one or more double bonds ● Oils from plants

Phospholipids - Consist of glycerol molecule + 2 fatty acids + phosphate group - Fatty acid “tails” are nonpolar and hydrophobic - Phosphate “head” is polar and hydrophilic - An amphipathic hydrophobic regions molecule = both hydrophilic and - Bilayered structures - provide structural foundations of cell membranes
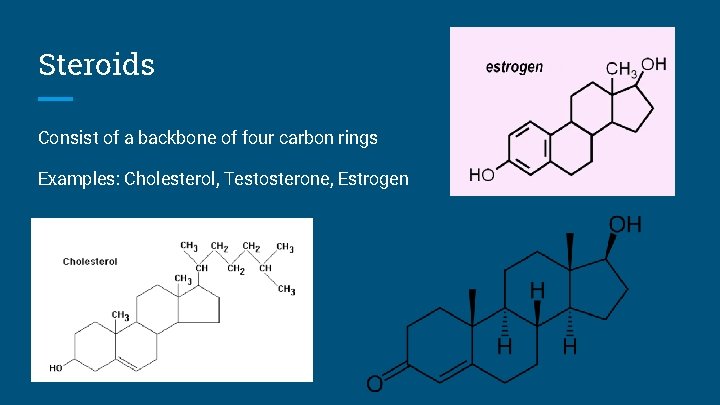
Steroids Consist of a backbone of four carbon rings Examples: Cholesterol, Testosterone, Estrogen

Proteins Uses for proteins: ● ● ● ● Growth and repair : Proteins repair muscles and tissues Signaling : Hormones coordinate an organism’s activities Defense : Antibodies inactivate proteins Catalyzing chemical reactions Enzymes : Speed up chemical reactions Storage : Store amino acids Structure : Provide support for hair, horns, feathers, etc

Protein Basics The subunit for proteins are Amino Acids : Contain carboxyl and amino groups, 23 amino acids and they all contain different R groups which give them different properties Polypeptides : Unbranched polymers built from amino acids Protein : Consists of one or more proteins Peptide bonds : Link amino acids

Proteins Structure of Proteins 1. Primary: The linear order of amino acids 2. Secondary: 3 D arrangement that is caused by HYDROGEN BONDING a. Results in alpha helices or beta sheets 3. Tertiary: Additional 3 D shaping that is caused by more HYDROGEN BONDING and DISULFIDE BONDING 4. Quaternary: Results when a protein consists of many polypeptide chains
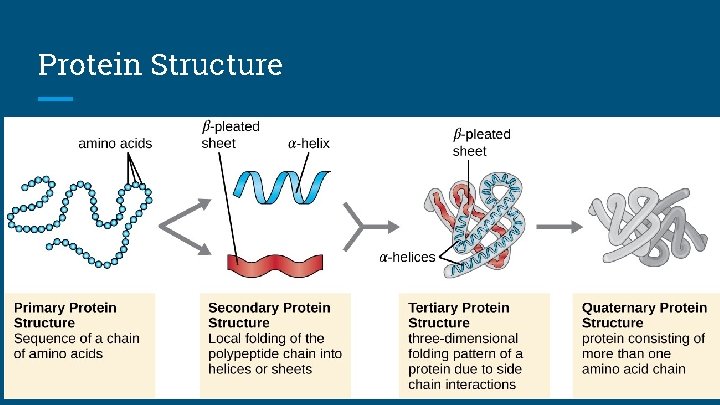
Protein Structure
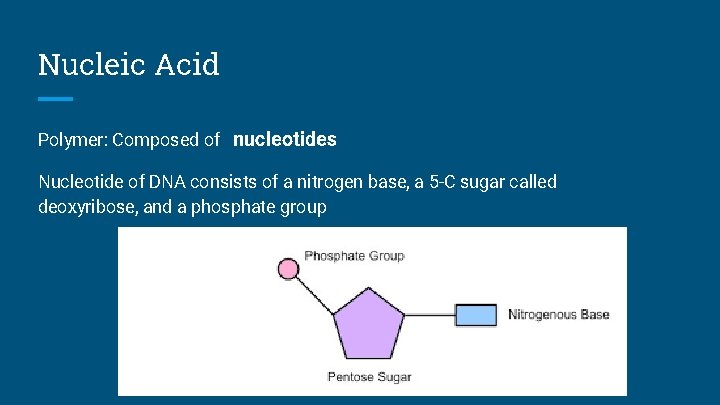
Nucleic Acid Polymer: Composed of nucleotides Nucleotide of DNA consists of a nitrogen base, a 5 -C sugar called deoxyribose, and a phosphate group

Nucleic Acids: Two major types Deoxyribonucleic Acid (DNA) ● ● ● Has its own directions for replication Directs synthesis of m. RNA and controls protein synthesis Has a DOUBLE HELIX structure Ribonucleic Acid (RNA) ● ● Sugar is ribose Does not have thymine and has uracil Single stranded Codes for proteins
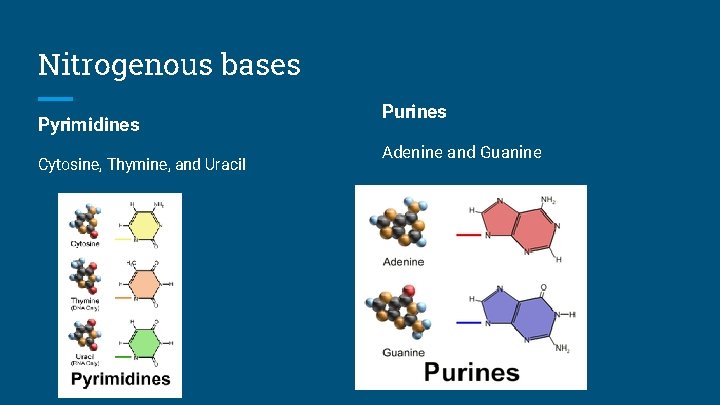
Nitrogenous bases Pyrimidines Cytosine, Thymine, and Uracil Purines Adenine and Guanine

Vocabulary Denaturation: Loss of a protein’s structure Sickle-Cell disease: An inherited blood disorder that come from a substitution in a single amino acid Enzyme: Catalysts that speed up chemical reactions, and almost never get degraded Increase reaction rate by lowering the activation energy
- Slides: 61