General Chemistry M R NaimiJamal Faculty of Chemistry














- Slides: 57

General Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology


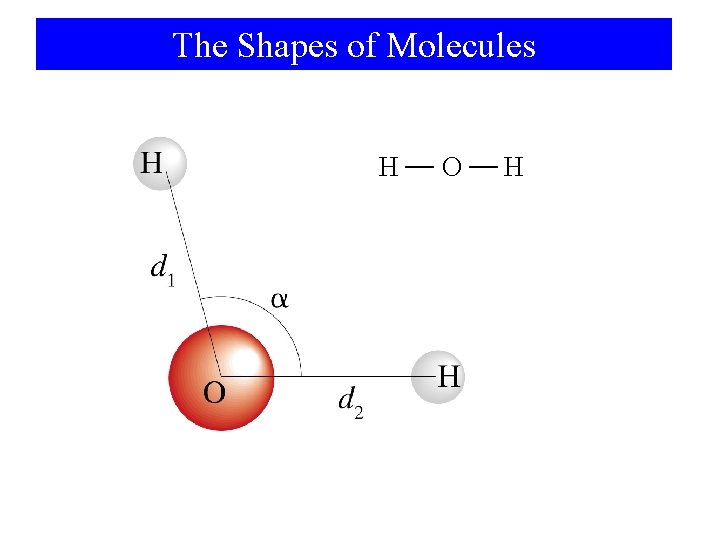
The Shapes of Molecules H O H

Terminology VSEPR Theory Valence Shell Electron Pair Repulsion theory Electron pairs repel each other whether they are in chemical bonds (bond pairs) or unshared (lone pairs). Electron pairs assume orientations about an atom to minimize repulsions.

Molecular Geometry • Electron group geometry – distribution of e- pairs. • Molecular geometry – distribution of nuclei. • Most important factor in determining geometry is relative repulsion between electron pairs.

Balloon Analogy

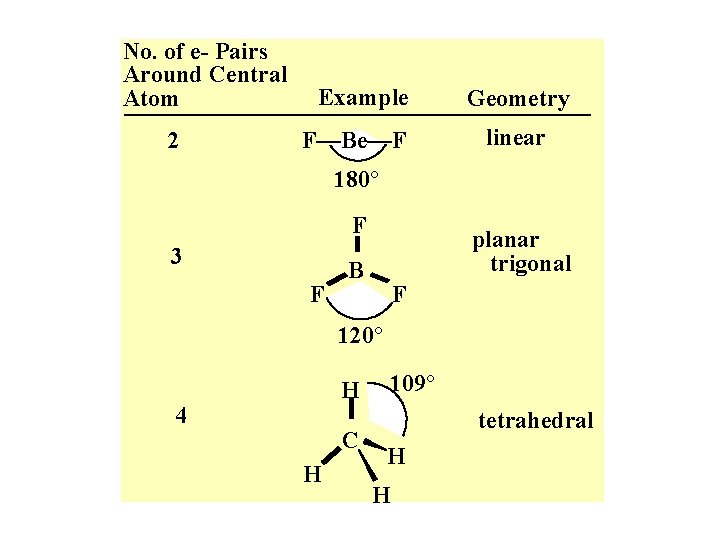
No. of e- Pairs Around Central Atom 2 Example F—Be—F Geometry linear 180° F 3 F planar trigonal B F 120° H 4 C H 109° tetrahedral H H

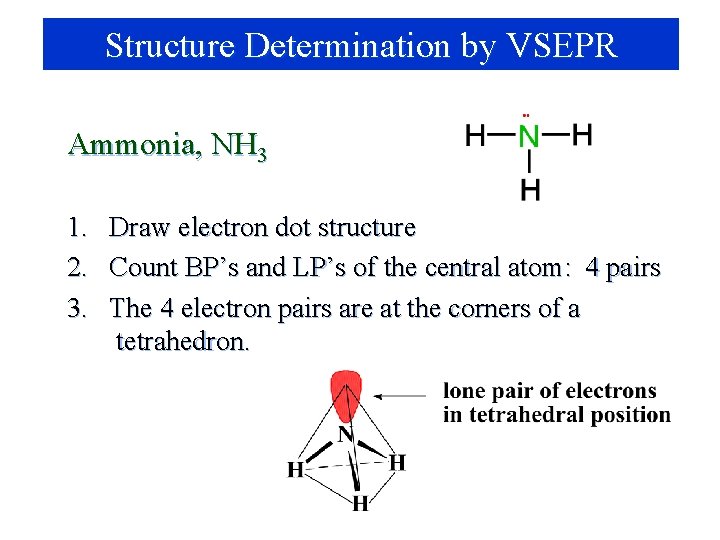
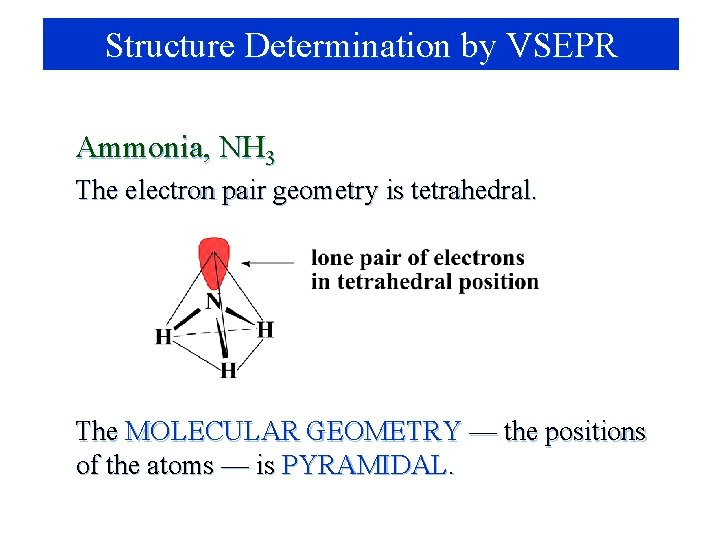
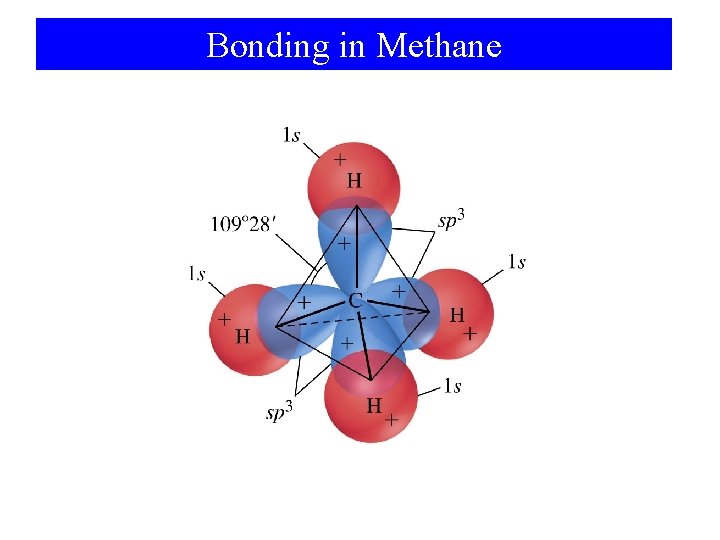
Structure Determination by VSEPR Ammonia, NH 3 1. 2. 3. Draw electron dot structure Count BP’s and LP’s of the central atom: 4 pairs The 4 electron pairs are at the corners of a tetrahedron.

Structure Determination by VSEPR Ammonia, NH 3 The electron pair geometry is tetrahedral. The MOLECULAR GEOMETRY — the positions of the atoms — is PYRAMIDAL.

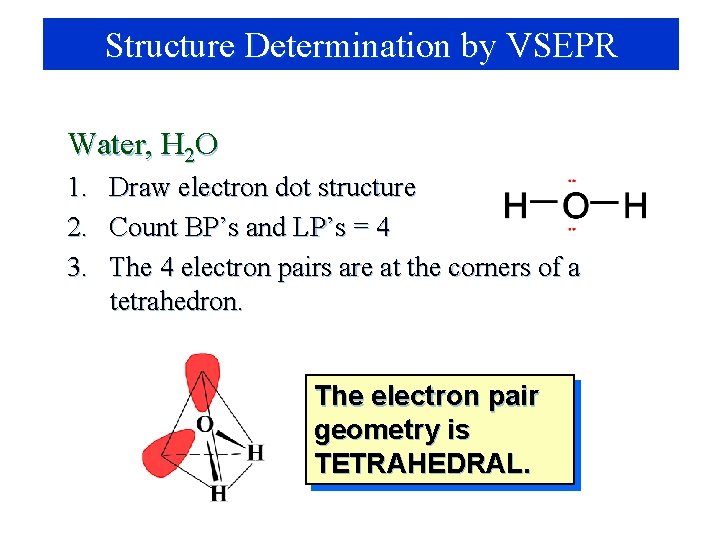
Structure Determination by VSEPR Water, H 2 O 1. 2. 3. Draw electron dot structure Count BP’s and LP’s = 4 The 4 electron pairs are at the corners of a tetrahedron. The electron pair geometry is TETRAHEDRAL.

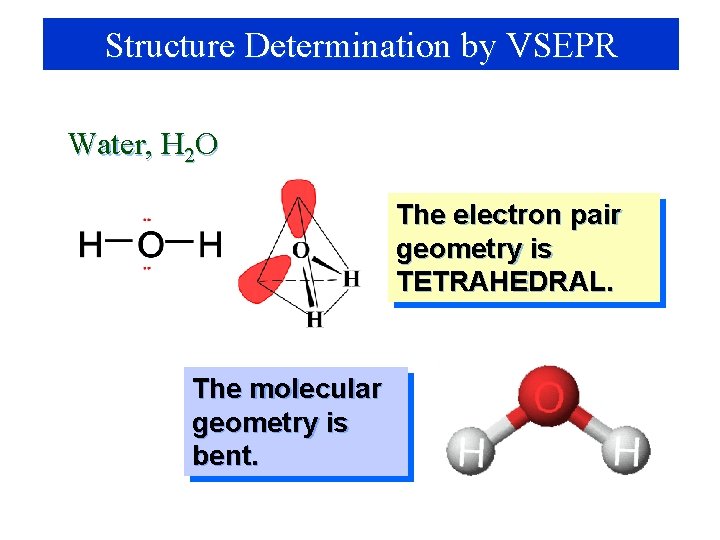
Structure Determination by VSEPR Water, H 2 O The electron pair geometry is TETRAHEDRAL. The molecular geometry is bent.

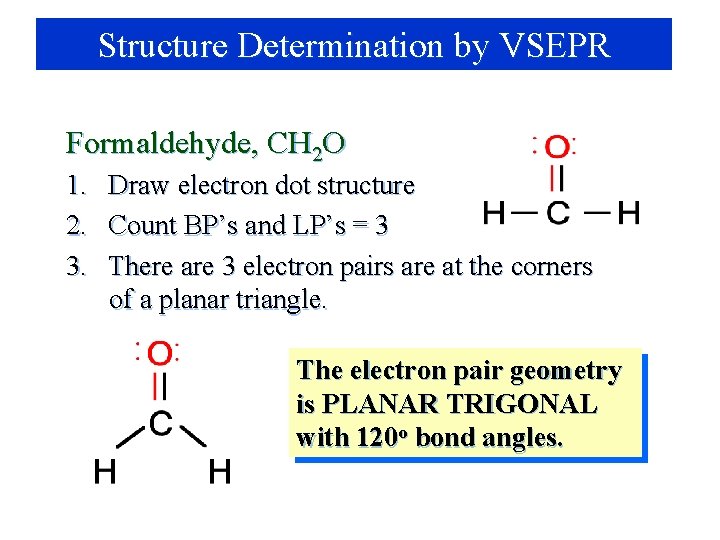
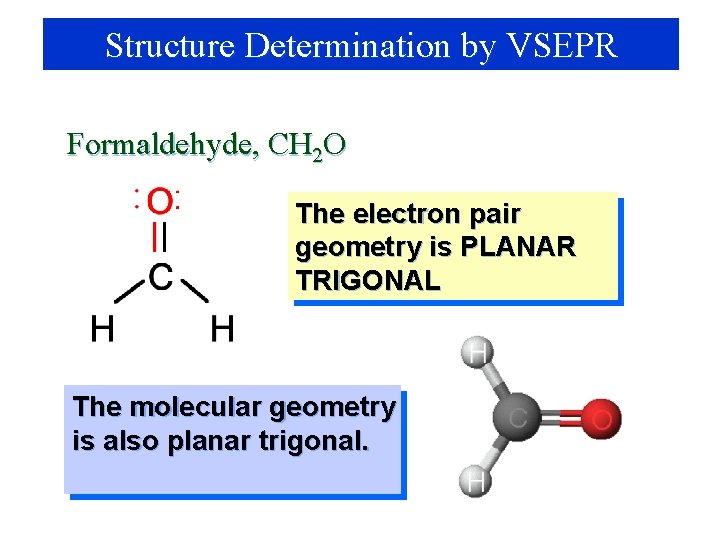
Structure Determination by VSEPR Formaldehyde, CH 2 O 1. Draw electron dot structure 2. Count BP’s and LP’s = 3 3. There are 3 electron pairs are at the corners of a planar triangle. The electron pair geometry is PLANAR TRIGONAL with 120 o bond angles.

Structure Determination by VSEPR Formaldehyde, CH 2 O The electron pair geometry is PLANAR TRIGONAL The molecular geometry is also planar trigonal.

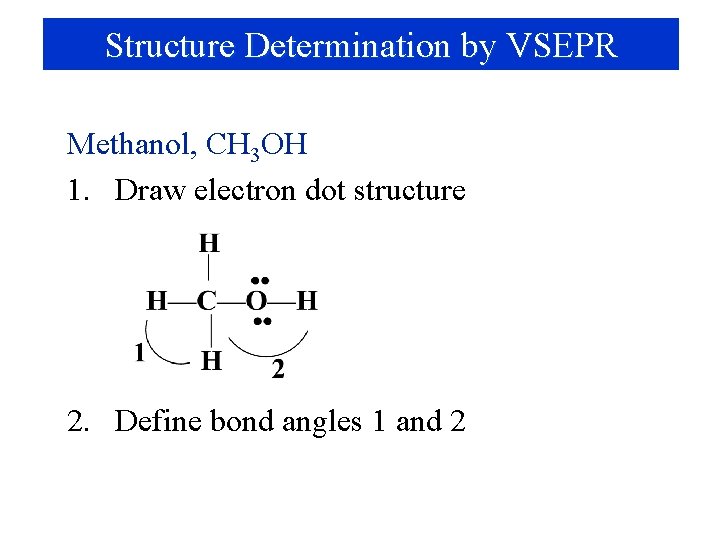
Structure Determination by VSEPR Methanol, CH 3 OH 1. Draw electron dot structure 2. Define bond angles 1 and 2

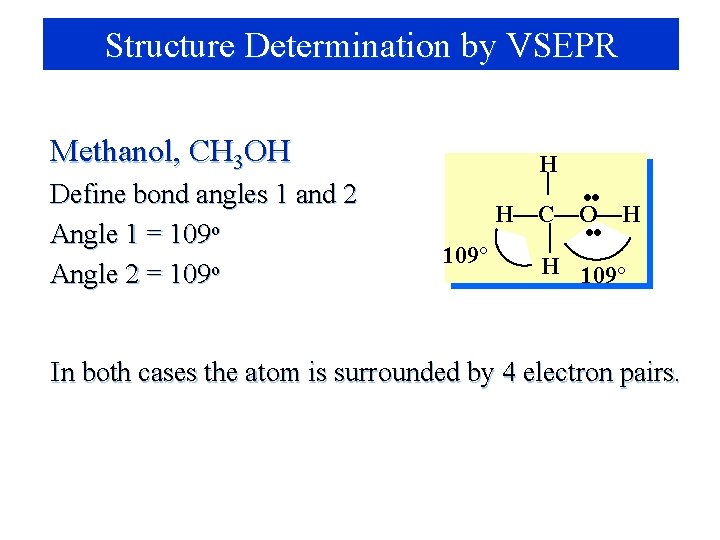
Structure Determination by VSEPR Methanol, CH 3 OH Define bond angles 1 and 2 Angle 1 = 109 o Angle 2 = 109 o H • • H—C—O—H • • 109° H 109° In both cases the atom is surrounded by 4 electron pairs.

Structure Determination by VSEPR Acetonitrile, CH 3 CN Draw the electron dot structure

Structure Determination by VSEPR Acetonitrile, CH 3 CN Define bond angles 1 and 2 H 109 o 109° H 180° N • • H—C—C Angle 1 = Angle 2 = 180 o One C is surrounded by 4 electron “lumps” and the other by 2 “lumps”

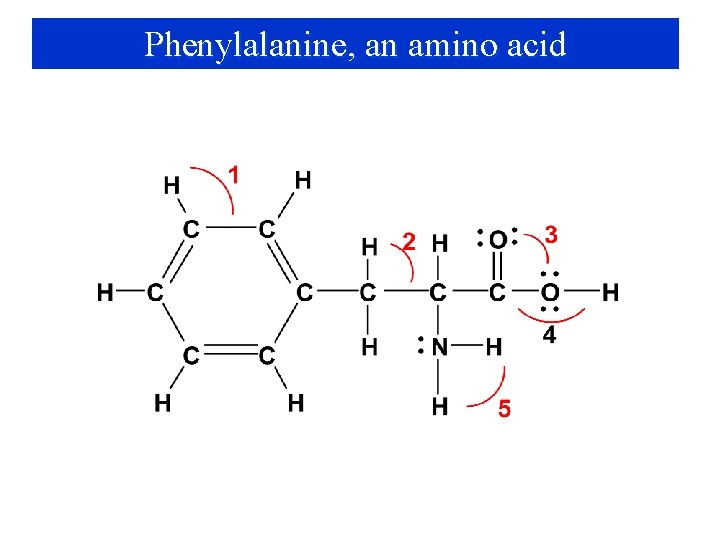
Phenylalanine, an amino acid

Phenylalanine

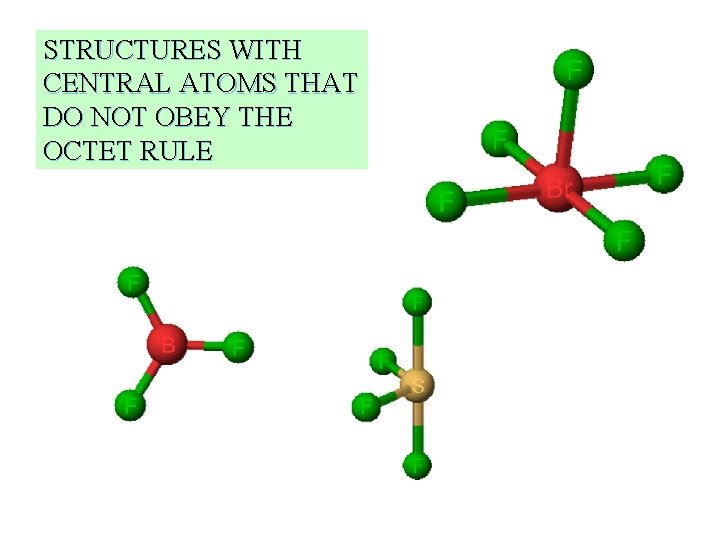
STRUCTURES WITH CENTRAL ATOMS THAT DO NOT OBEY THE OCTET RULE

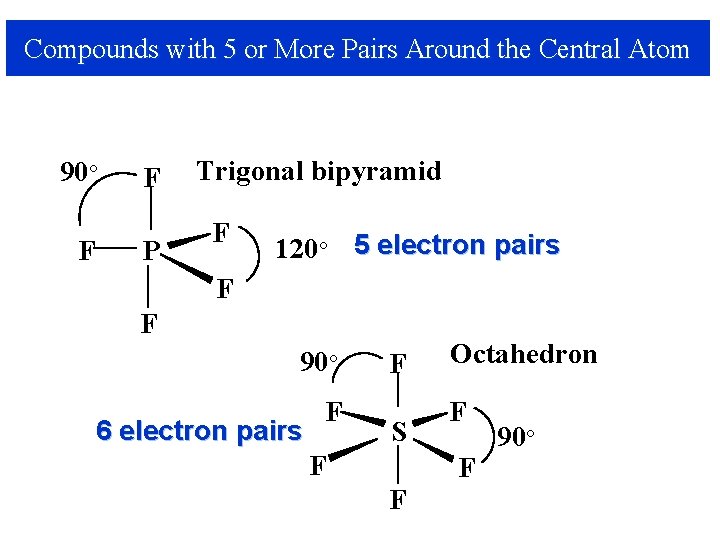
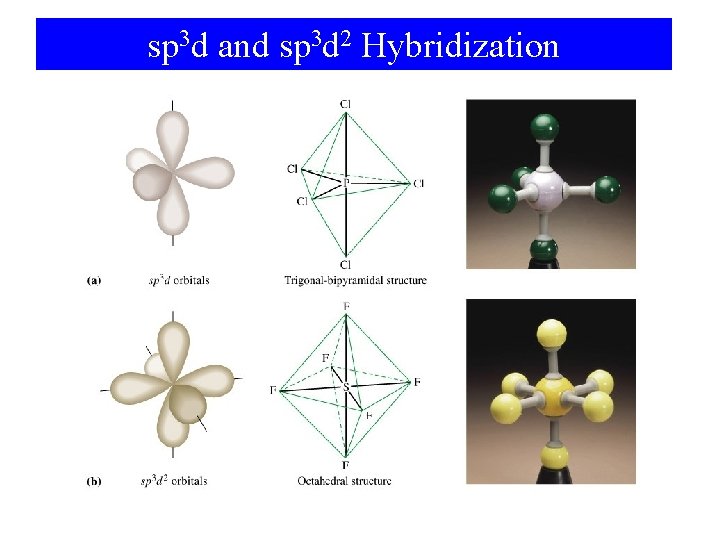
Compounds with 5 or More Pairs Around the Central Atom 90° F F P Trigonal bipyramid F 120° 5 electron pairs F F 90° 6 electron pairs F F S F Octahedron F F F 90°

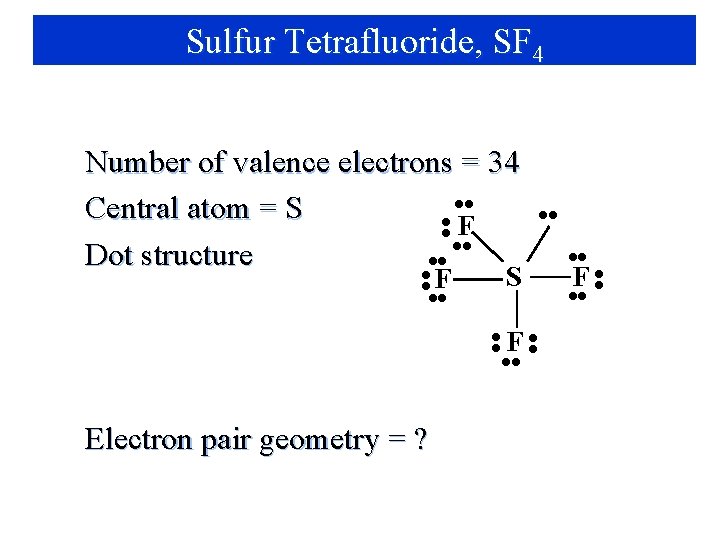
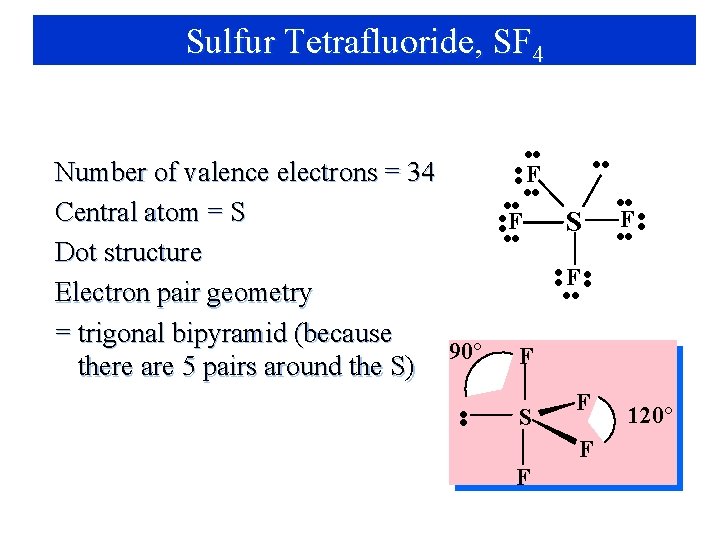
Sulfur Tetrafluoride, SF 4 Number of valence electrons = 34 • • Central atom = S • • F • • Dot structure • • F • • S • • F • • Electron pair geometry = ? F • •

Sulfur Tetrafluoride, SF 4 Number of valence electrons = 34 Central atom = S Dot structure Electron pair geometry = trigonal bipyramid (because 90° there are 5 pairs around the S) • • • • F • • S • • F • • F S F F F 120°

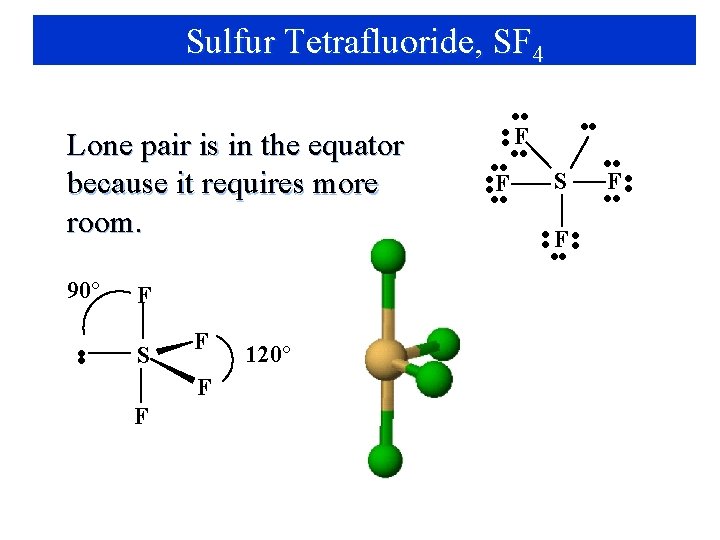
Sulfur Tetrafluoride, SF 4 Lone pair is in the equator because it requires more room. 90° F • • S F F F 120° • • F • • S • • F • •

Hybridization of Atomic Orbitals

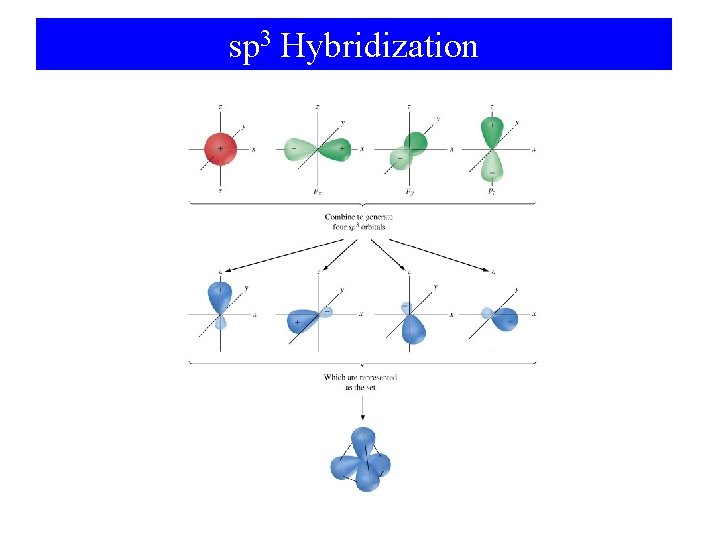
sp 3 Hybridization

Bonding in Methane

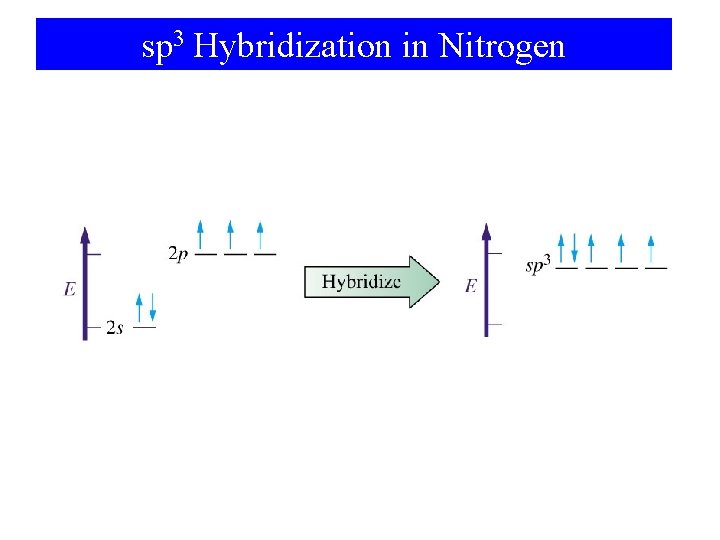
sp 3 Hybridization in Nitrogen

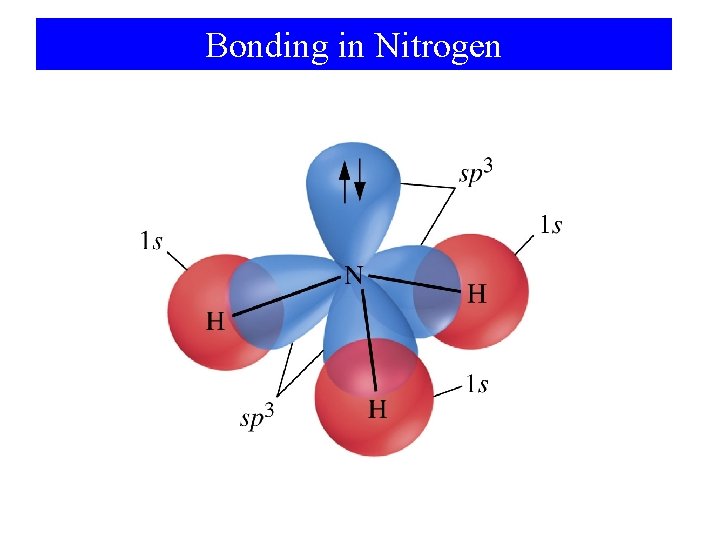
Bonding in Nitrogen

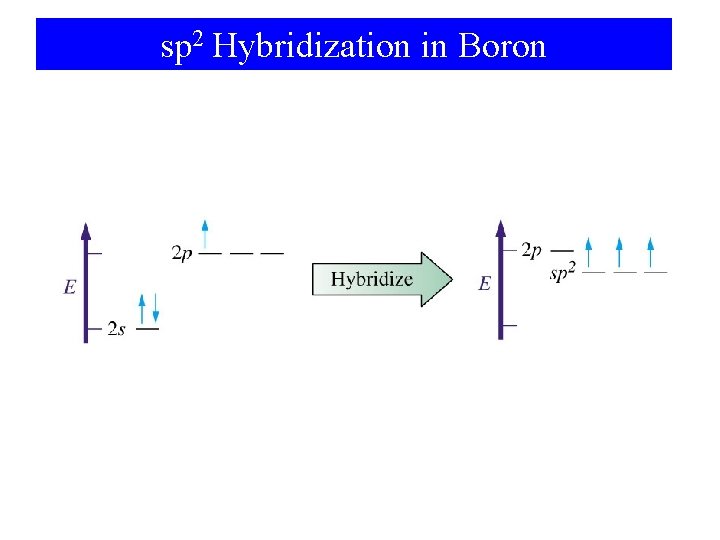
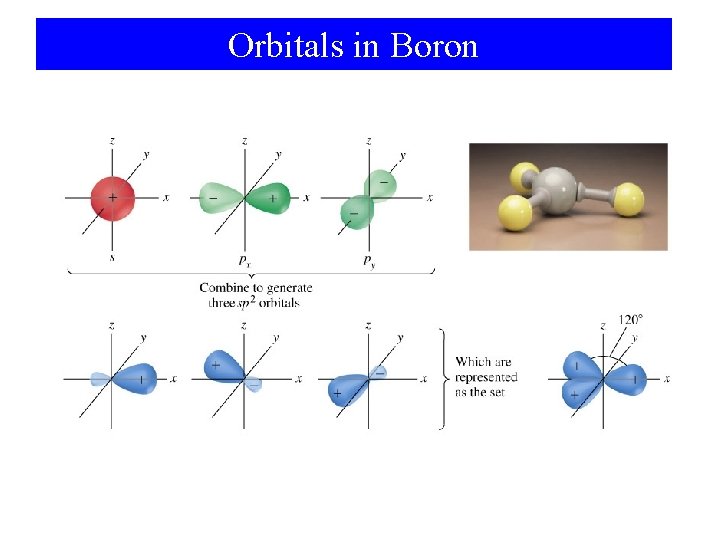
sp 2 Hybridization in Boron

Orbitals in Boron

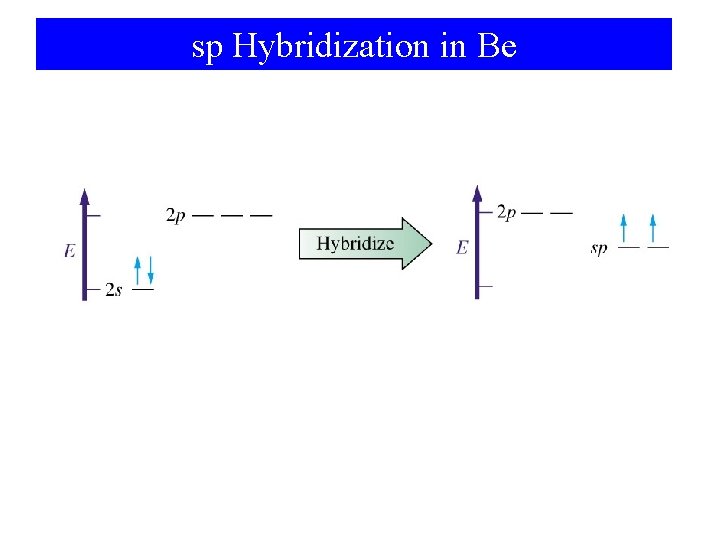
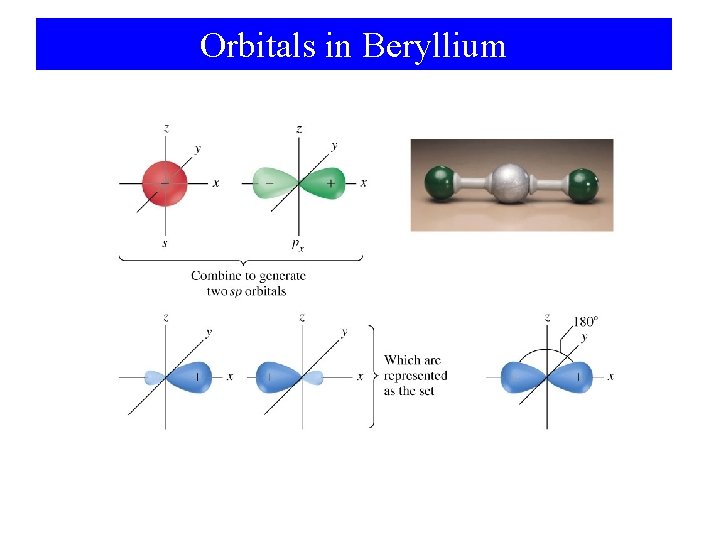
sp Hybridization in Be

Orbitals in Beryllium

sp 3 d and sp 3 d 2 Hybridization

Hybrid Orbitals and VSEPR • Write a plausible Lewis structure. • Use VSEPR to predict electron geometry. • Select the appropriate hybridization.

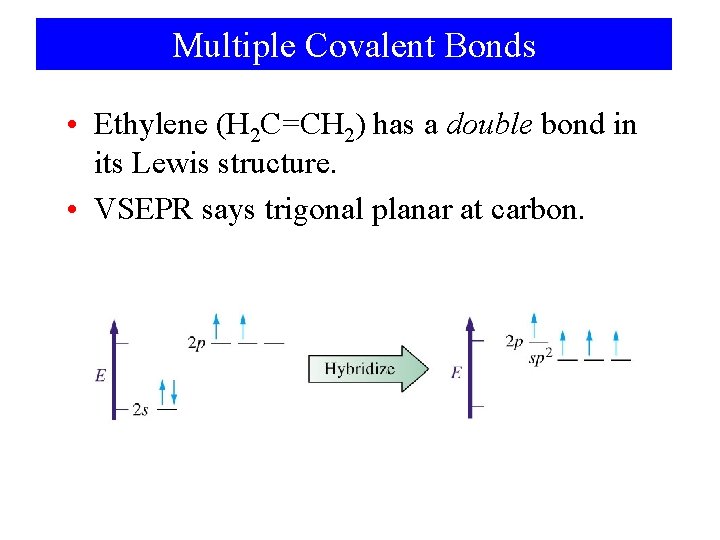
Multiple Covalent Bonds • Ethylene (H 2 C=CH 2) has a double bond in its Lewis structure. • VSEPR says trigonal planar at carbon.

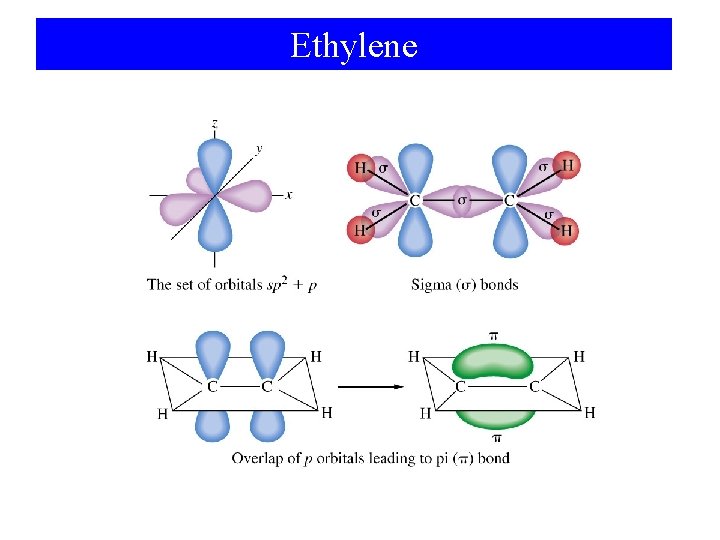
Ethylene

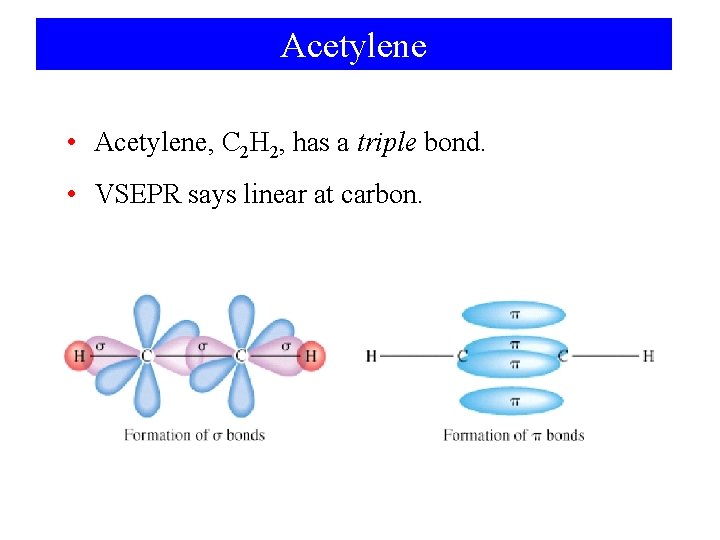
Acetylene • Acetylene, C 2 H 2, has a triple bond. • VSEPR says linear at carbon.

Applying VSEPR Theory • Draw a plausible Lewis structure. • Determine the number of e- groups and identify them as bond or lone pairs. • Establish the e- group geometry. • Determine the molecular geometry. • Multiple bonds count as one group of electrons. • More than one central atom can be handled individually.

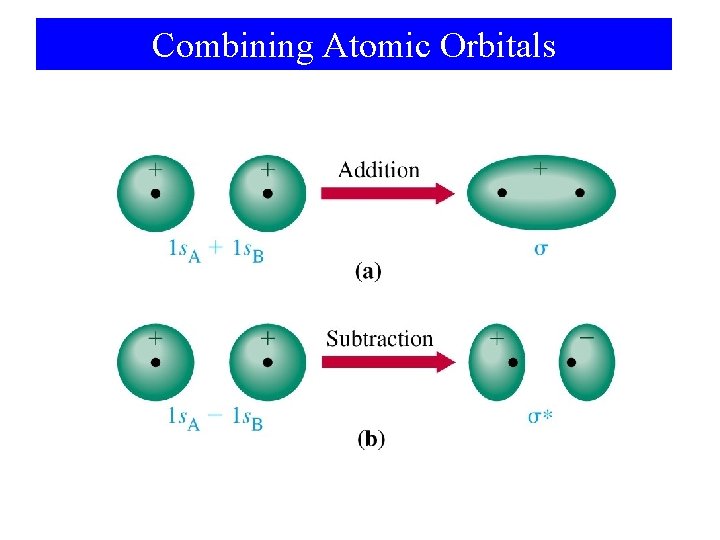
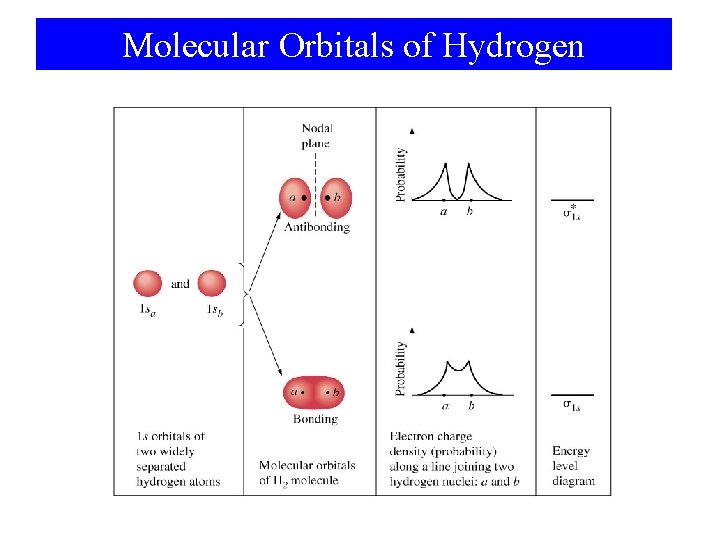
Molecular Orbital Theory • Atomic orbitals are isolated on atoms. • Molecular orbitals span two or more atoms. • LCAO: – Linear Combination of Atomic Orbitals. Ψ 1 = φ1 + φ2 Ψ 2 = φ1 - φ2

Combining Atomic Orbitals

Molecular Orbitals of Hydrogen

Basic Ideas Concerning MOs • Number of MOs = Number of AOs. • Bonding and antibonding MOs formed from AOs. • e- fill the lowest energy MO first. • Pauli exclusion principle is followed. • Hund’s rule is followed

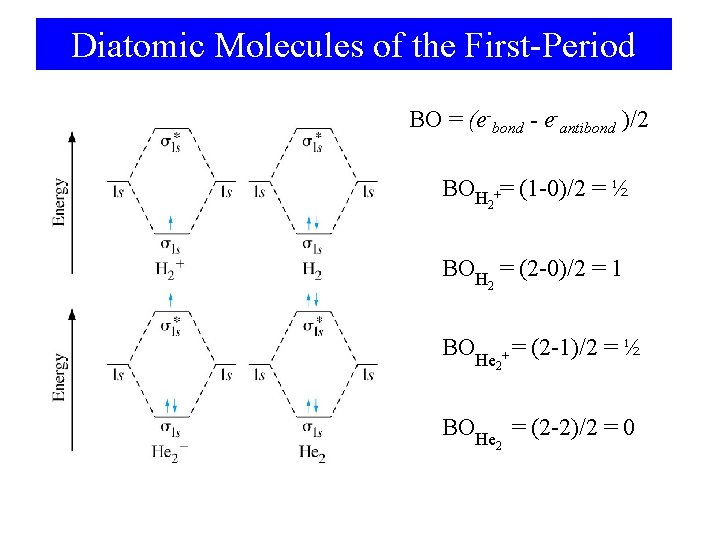
Bond Order • Stable species have more electrons in bonding orbitals than antibonding. # e- in bonding MOs - # e- in antibonding MOs Bond Order = 2

Diatomic Molecules of the First-Period BO = (e-bond - e-antibond )/2 BOH += (1 -0)/2 = ½ 2 BOH = (2 -0)/2 = 1 2 BOHe + = (2 -1)/2 = ½ 2 BOHe = (2 -2)/2 = 0 2

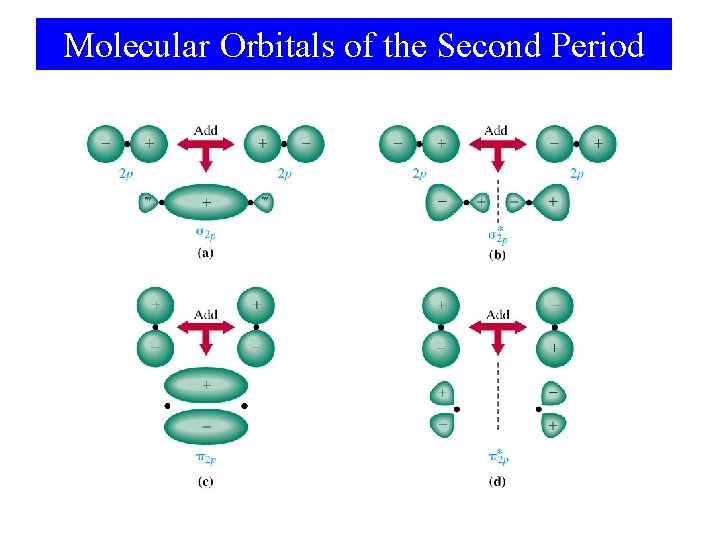
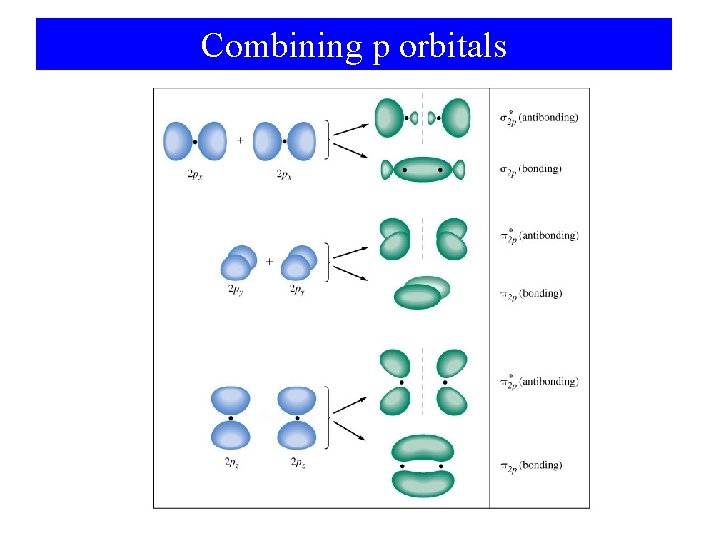
Molecular Orbitals of the Second Period • First period use only 1 s orbitals. • Second period have 2 s and 2 p orbitals available. • p orbital overlap: – End-on overlap is best – sigma bond (σ). – Side-on overlap is good – pi bond (π).

Molecular Orbitals of the Second Period

Combining p orbitals

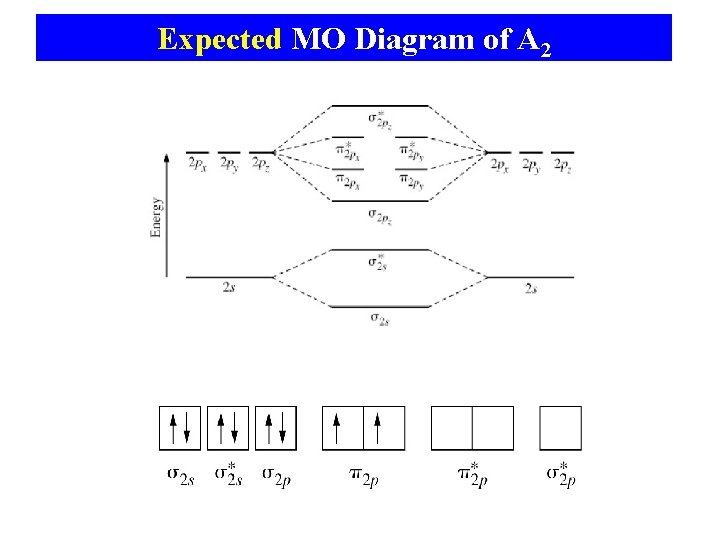
Expected MO Diagram of A 2

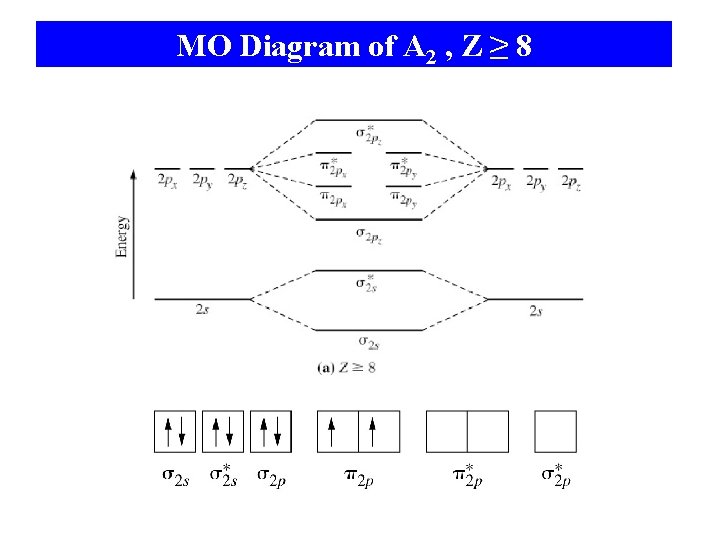
MO Diagram of A 2 , Z ≥ 8

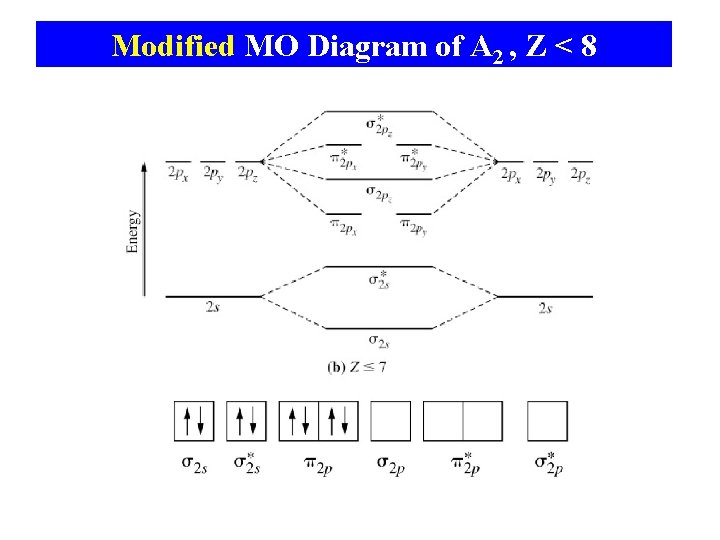
Modified MO Diagram of A 2 , Z < 8

MO Diagrams of 2 nd Period Diatomics


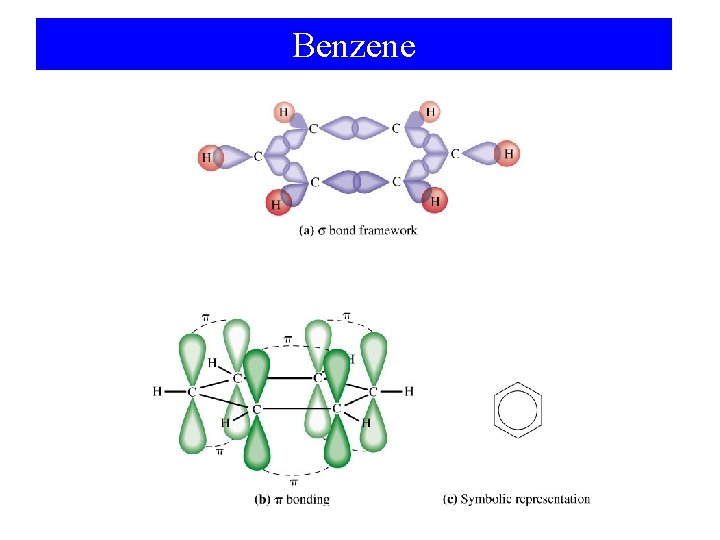
Benzene

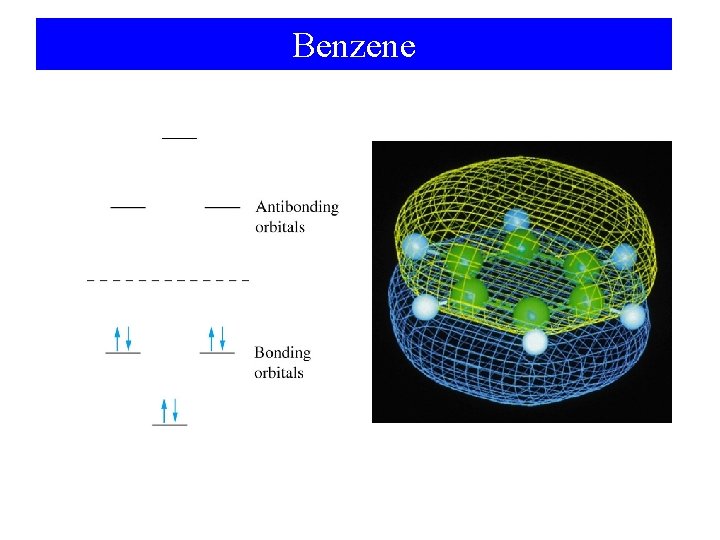
Benzene

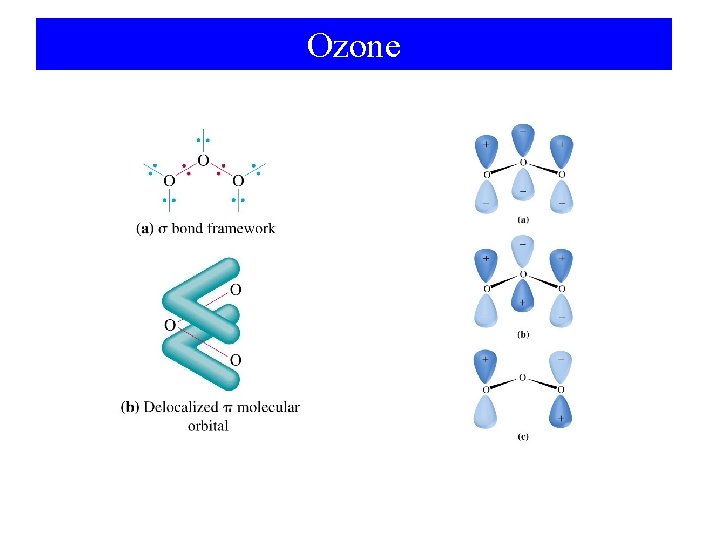
Ozone

Chapter 9 Questions 7, 16, 22, 25, 33 34, 35, 43