General Chemistry M R NaimiJamal Faculty of Chemistry




























- Slides: 65

General Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology


Intermolecular Forces and Some Properties of Liquids • Cohesive Forces – Intermolecular forces between like molecules. • Adhesive Forces – Intermolecular forces between unlike molecules. • Surface Tension – Energy or work required to increase the surface area of a liquid. • Viscosity – A liquids resistance to flow

Van der Waals Forces • Instantaneous dipoles. – Electrons move in an orbital to cause a polarization. • Induced dipoles. – Electrons move in response to an outside force. • Dispersion or London forces. – Instantaneous “dipole – induced dipole” attraction. – Related to polarizability.

Dipole Interactions

Polar Covalent Bonds: Dipole Moments • Dipole moment - Net molecular polarity, due to difference in summed charges • - magnitude of charge Q at end of molecular dipole times distance r between charges • = Q r, in debyes (D) • 1 D = 3. 34 10 30 coulomb meter

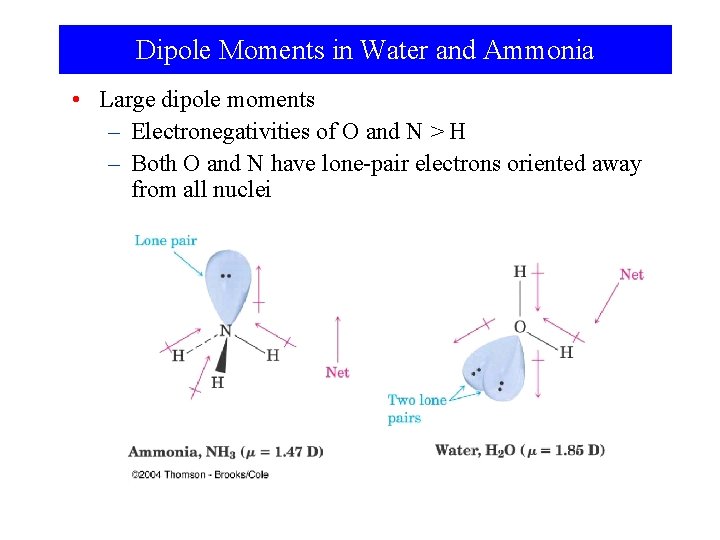
Dipole Moments in Water and Ammonia • Large dipole moments – Electronegativities of O and N > H – Both O and N have lone-pair electrons oriented away from all nuclei

Question Compare the dipole moment of NH 3 with NF 3, which one is more polar?

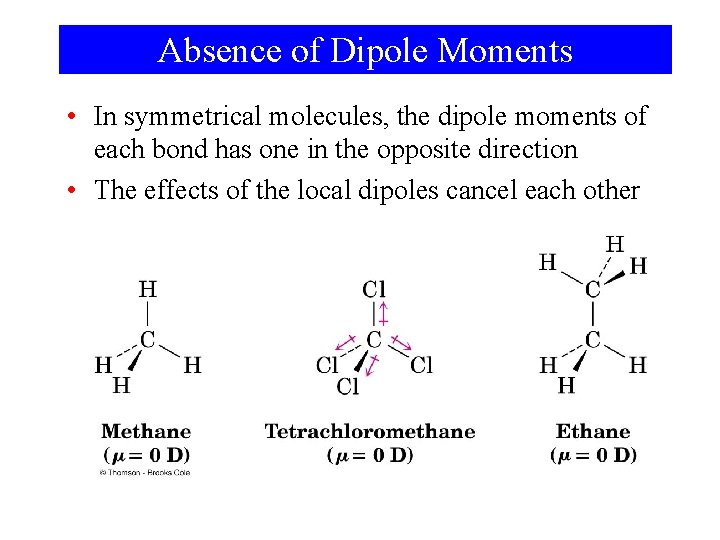
Absence of Dipole Moments • In symmetrical molecules, the dipole moments of each bond has one in the opposite direction • The effects of the local dipoles cancel each other

Phenomenon of Induction

Dispersion or London forces Instantaneous “dipole – induced dipole” attraction, related to polarizability.


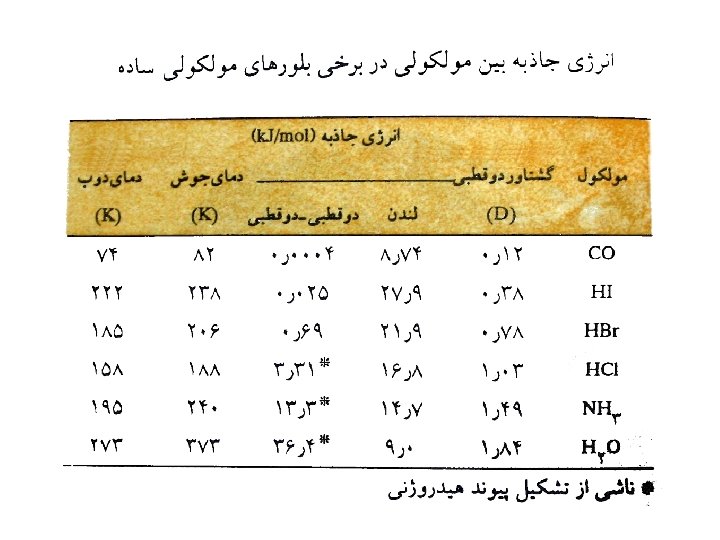
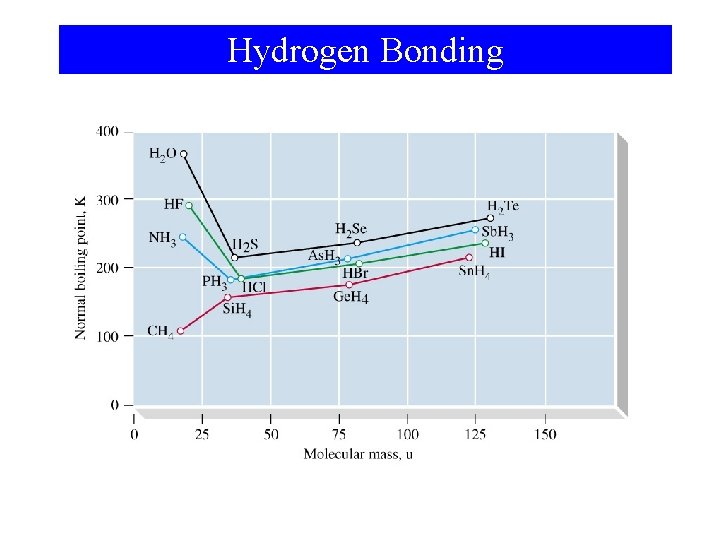
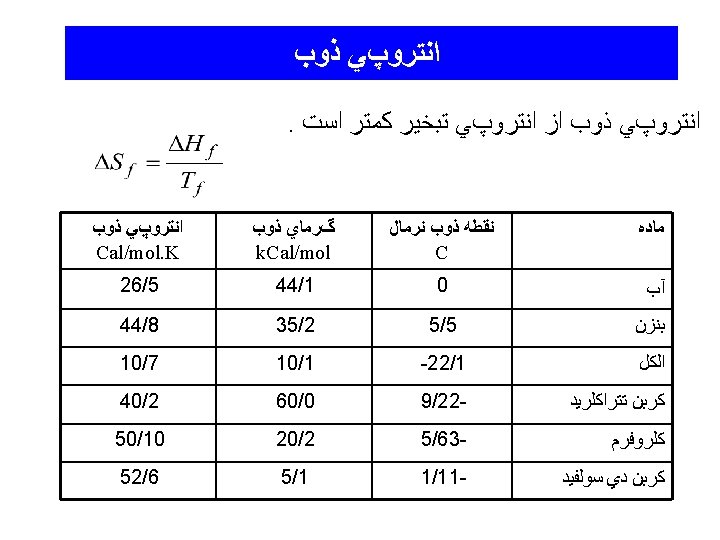
Hydrogen Bonding

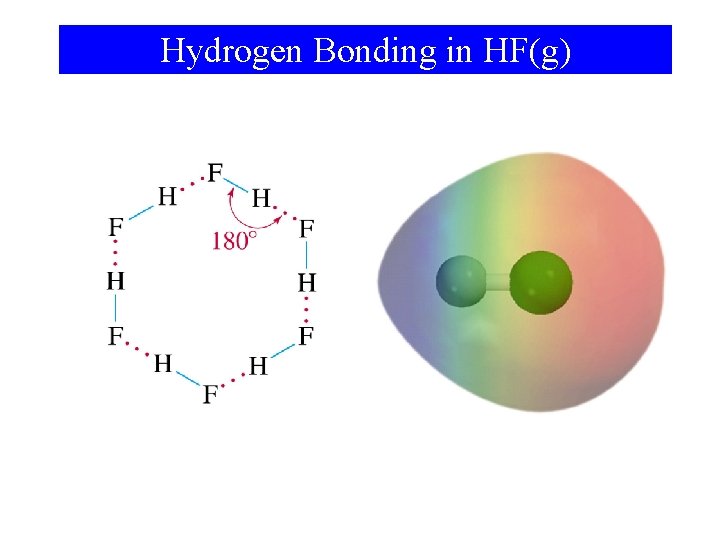
Hydrogen Bonding in HF(g)

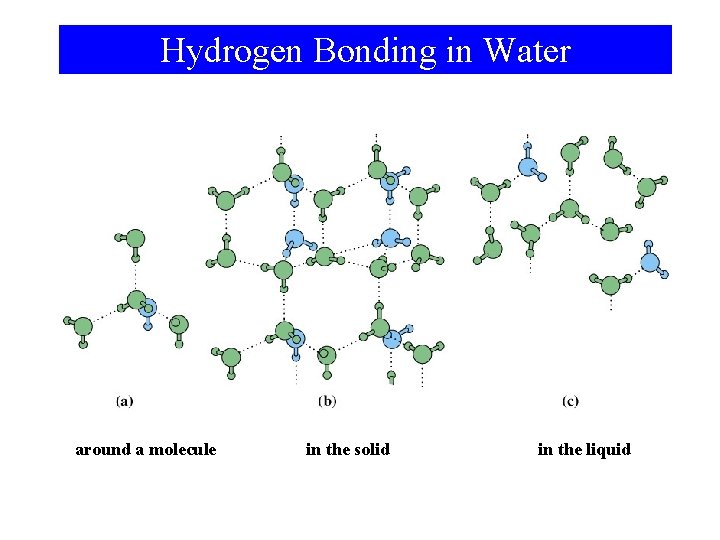
Hydrogen Bonding in Water around a molecule in the solid in the liquid

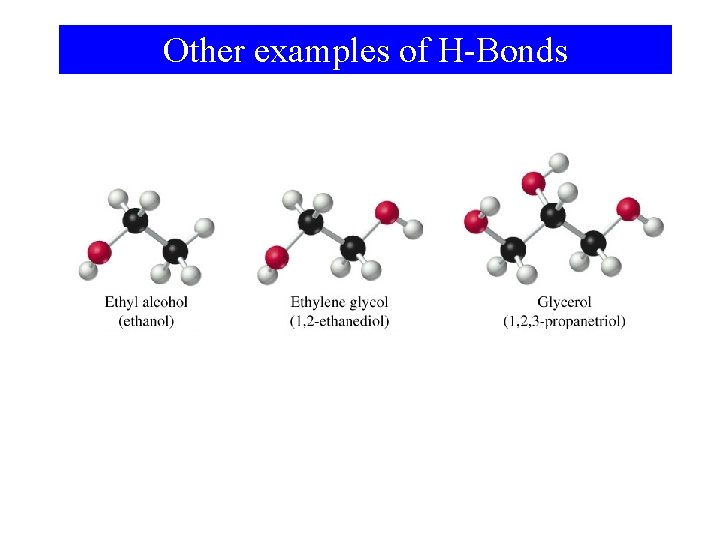
Other examples of H-Bonds

Liquids • Viscosity • Surface Tension • Evaporation


Surface Tension Energy or work required to increase the surface area of a liquid.

Surface Tension

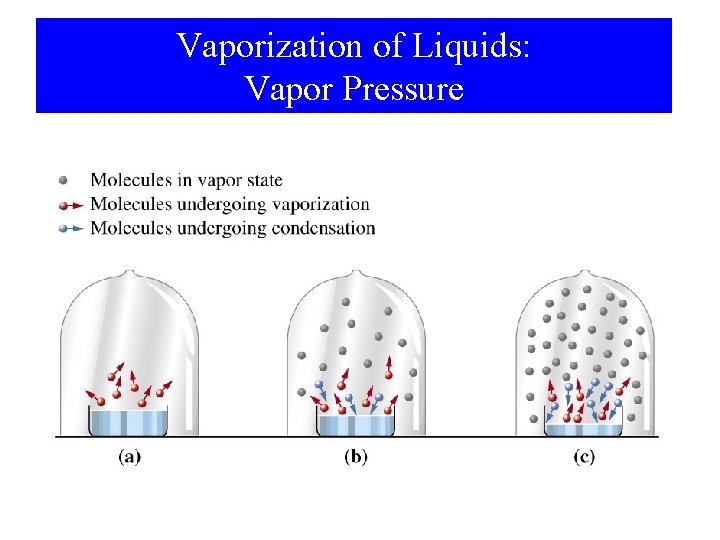
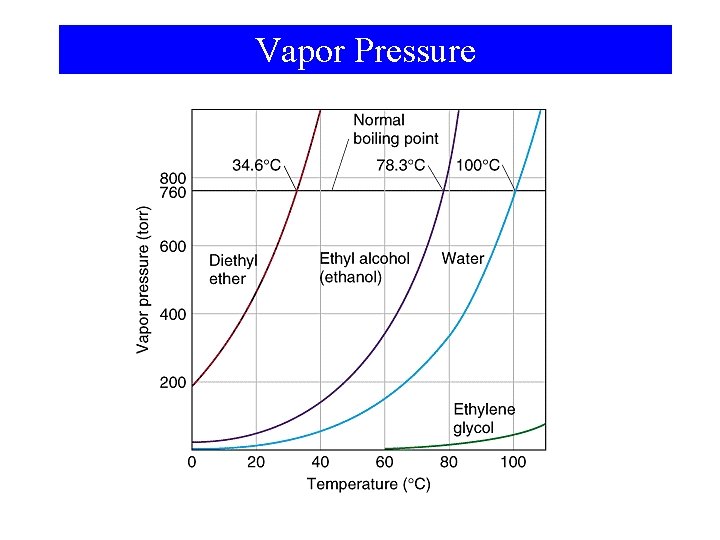
Vaporization of Liquids: Vapor Pressure

Vapor Pressure



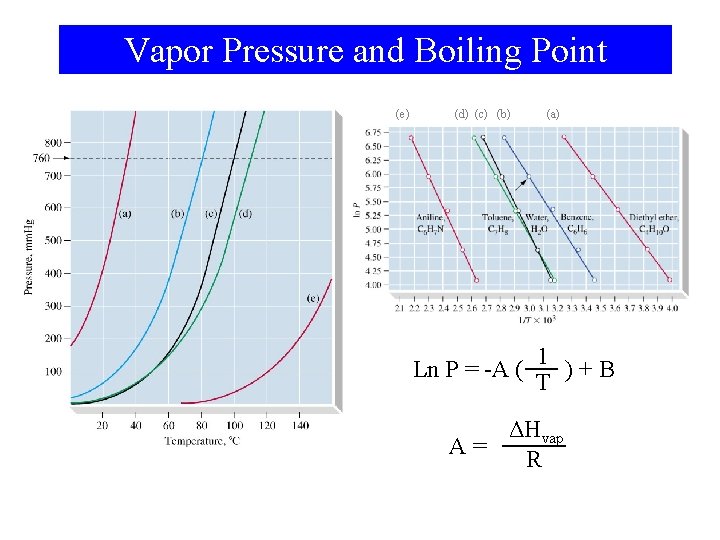
Vapor Pressure and Boiling Point (e) (d) (c) (b) (a) 1 Ln P = -A ( )+B T ΔHvap A= R






Some Properties of Solids Freezing Point Melting Point ΔHfus(H 2 O) = + 6. 01 k. J/mol


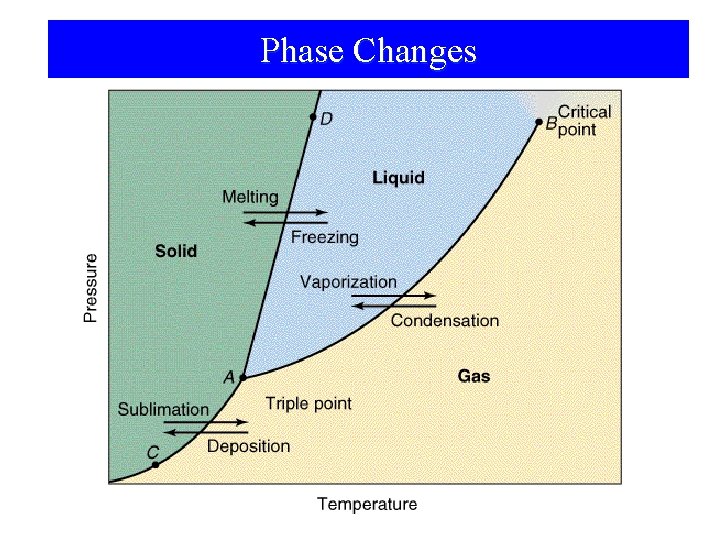
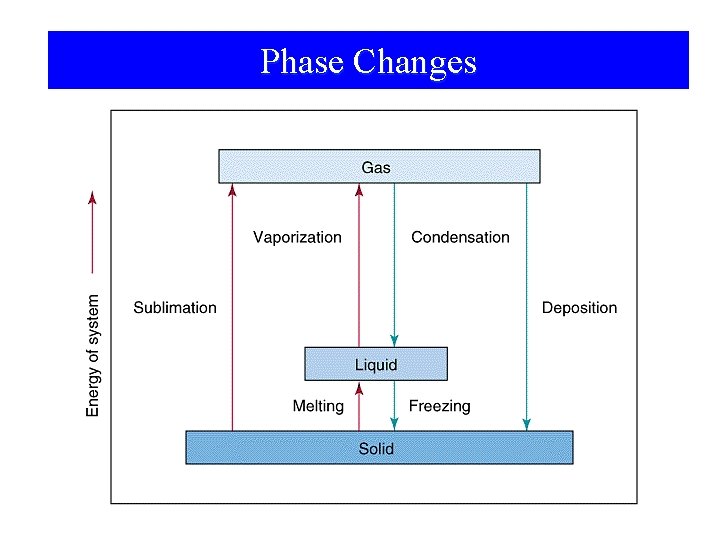
Phase Changes

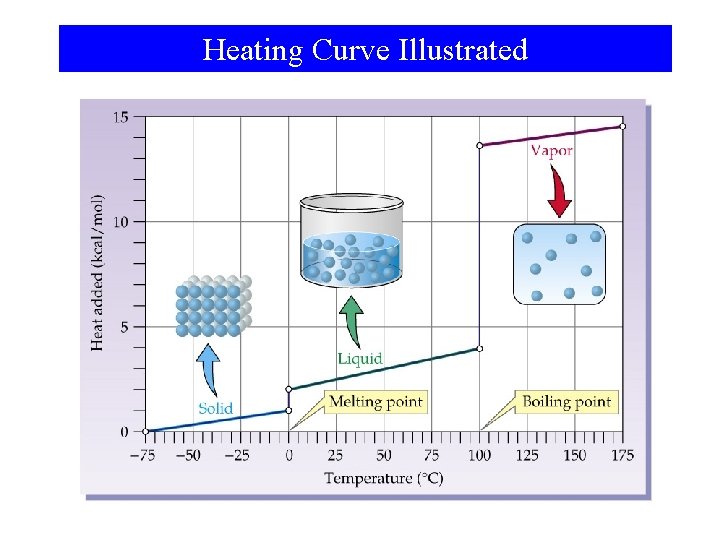
Phase Changes • • • Sublimation: Vaporization: Melting or fusion: Deposition: Condensation: Freezing: solid gas. liquid gas. solid liquid. gas solid. gas liquid solid.

Phase Changes

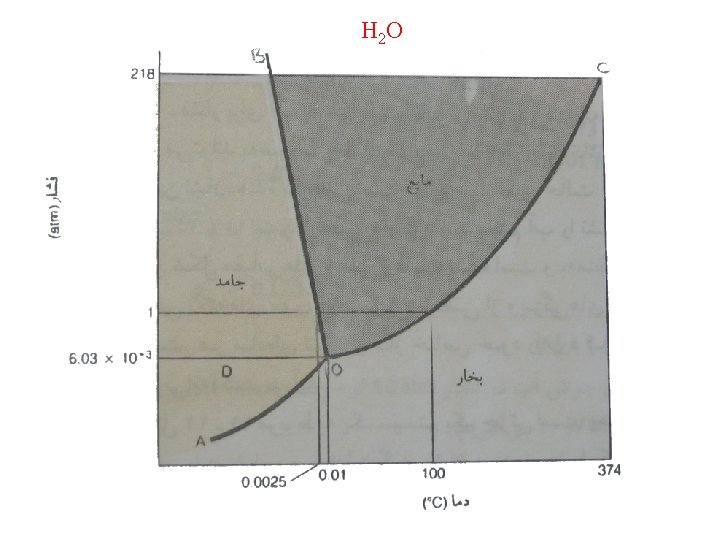
H 2 O

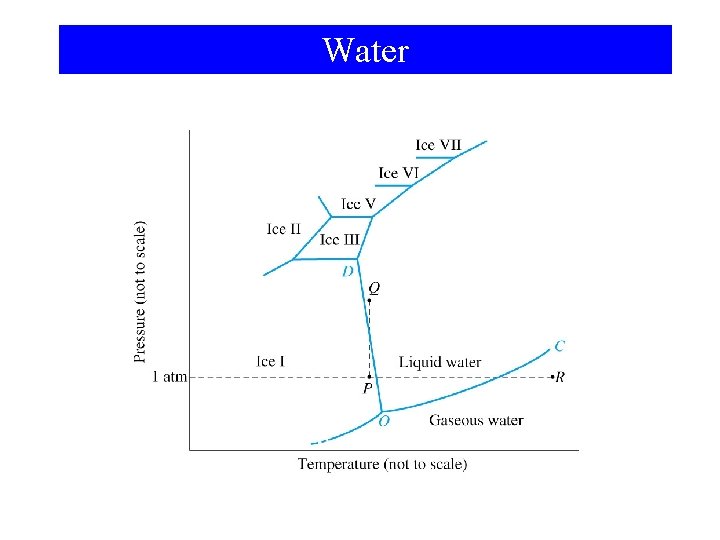
Water

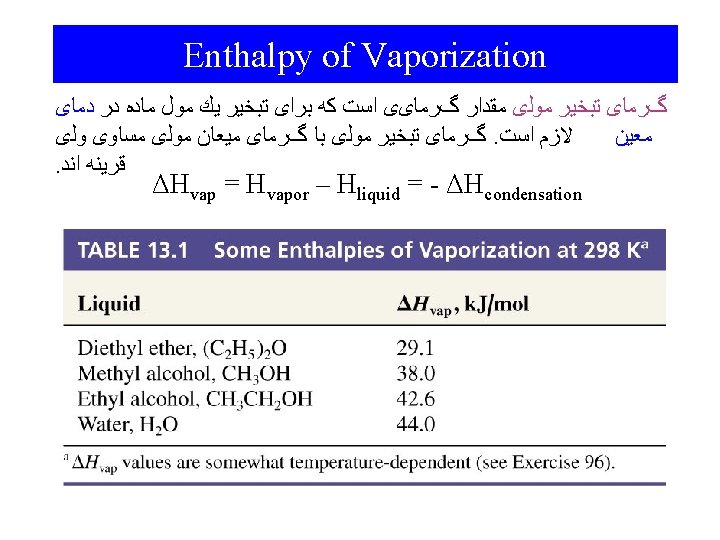
Phase Changes Energy Changes Accompanying Phase Changes – Sublimation: – Vaporization: – Melting or Fusion: – Deposition: – Condensation: – Freezing: Hsub > 0 (endothermic) Hvap > 0 (endothermic) Hfus > 0 (endothermic) Hdep < 0 (exothermic) Hcon < 0 (exothermic) Hfre < 0 (exothermic)

Heating Curve Illustrated

Phase changes

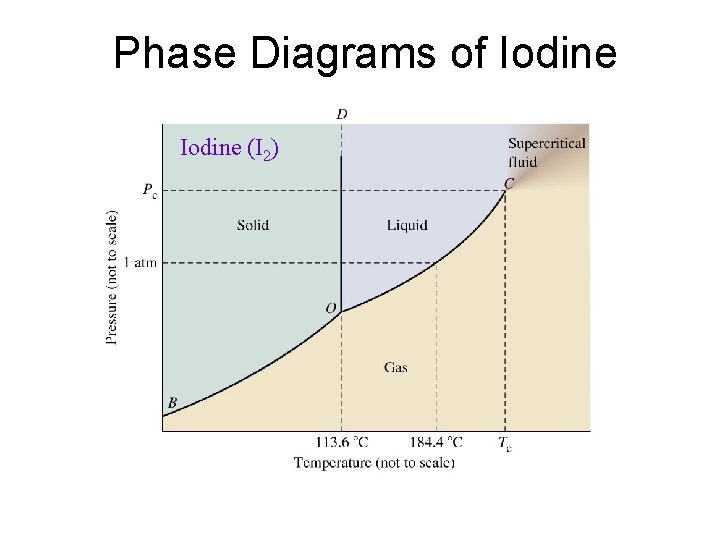
Phase Diagrams of Iodine (I 2)

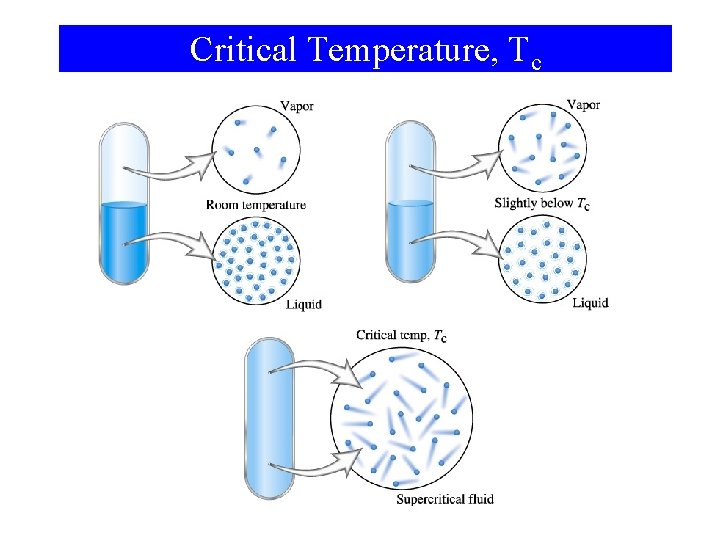
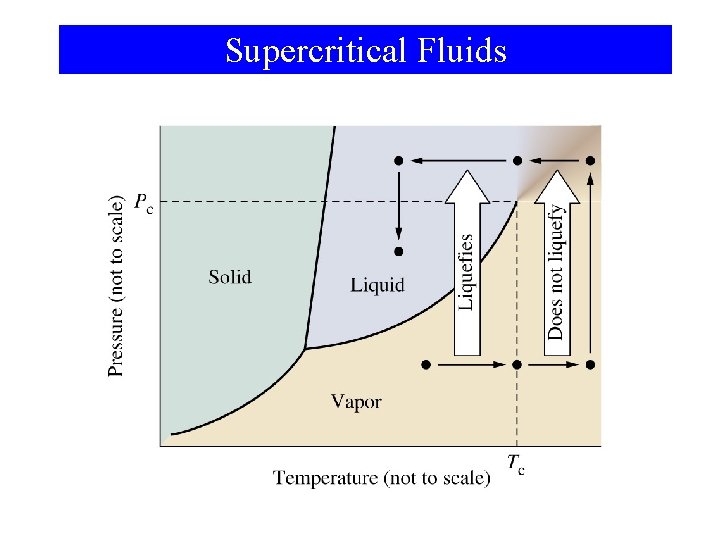
Phase Changes Critical Temperature and Pressure • Gases liquefied by increasing pressure at some temperature. • Critical temperature (Tc): The temperature, above it liquefaction of a gas using pressure is not more possible. • Critical pressure: the minimum pressure required for liquefaction at Tc.

Critical Temperature, Tc

Supercritical Fluids

The Critical Point

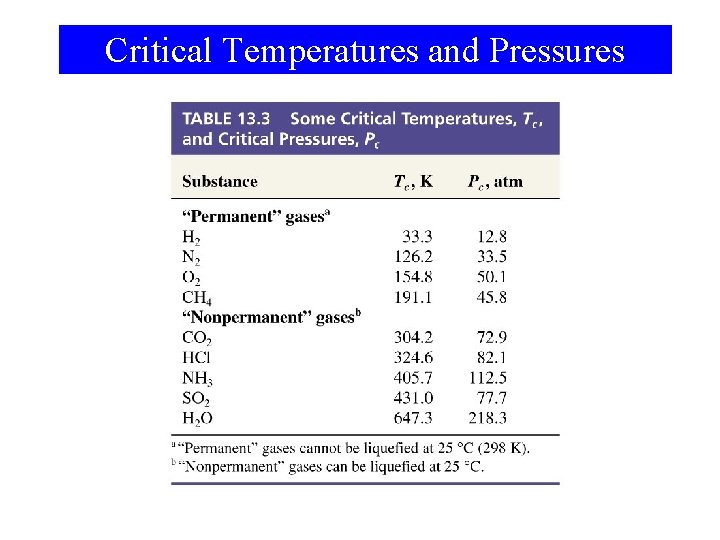
Critical Temperatures and Pressures

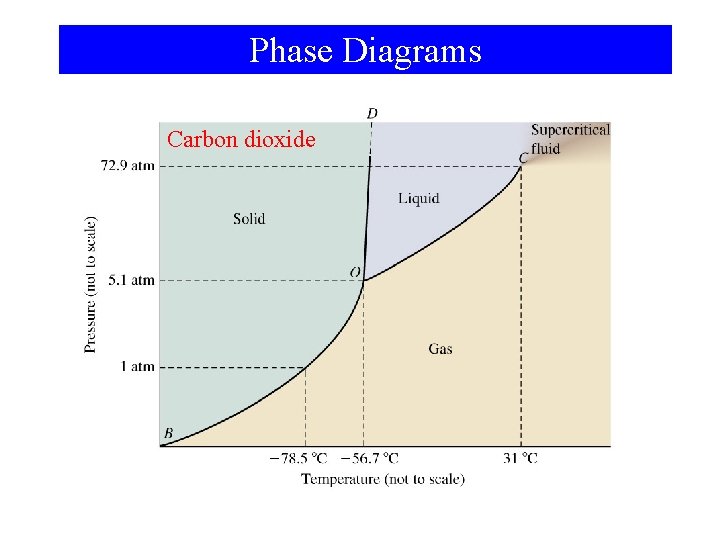
Phase Diagrams Carbon dioxide

Transition to Supercritical CO 2

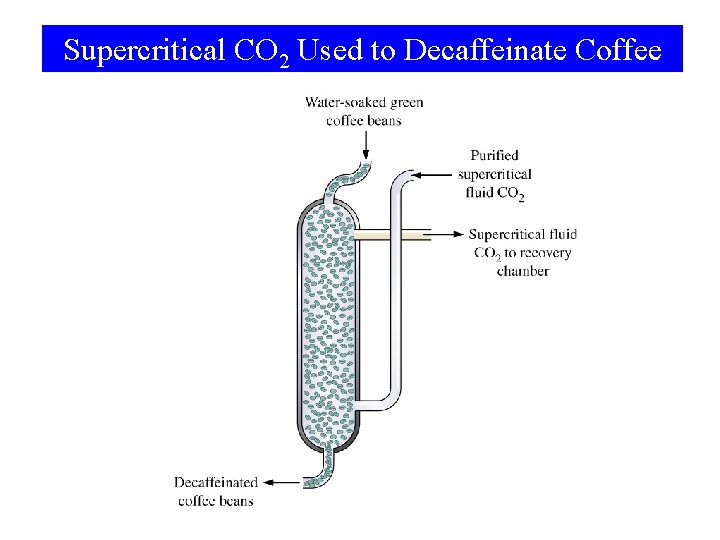
Supercritical CO 2 Used to Decaffeinate Coffee

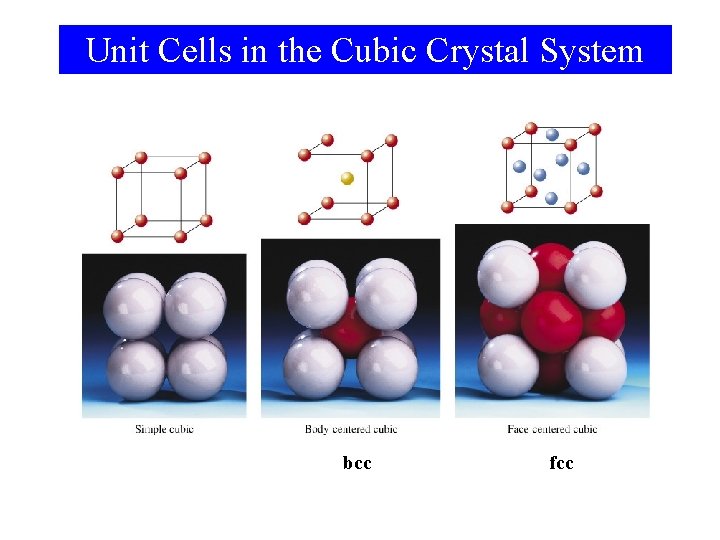
Crystal Structures


Unit Cells in the Cubic Crystal System bcc fcc

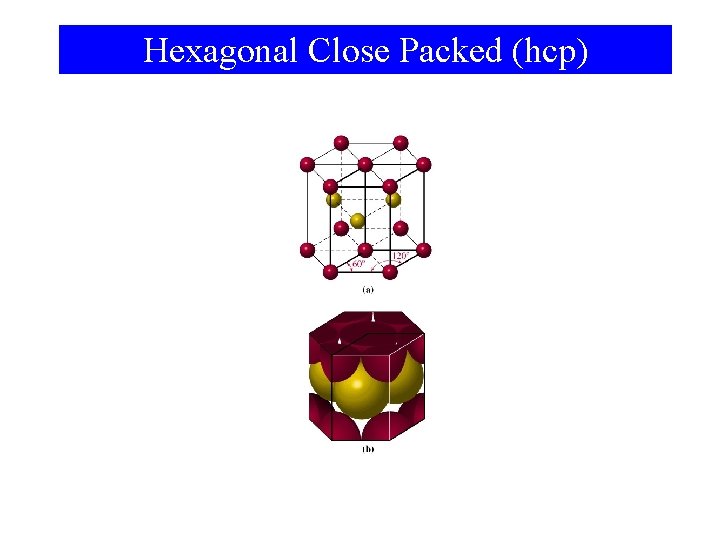
Hexagonal Close Packed (hcp)

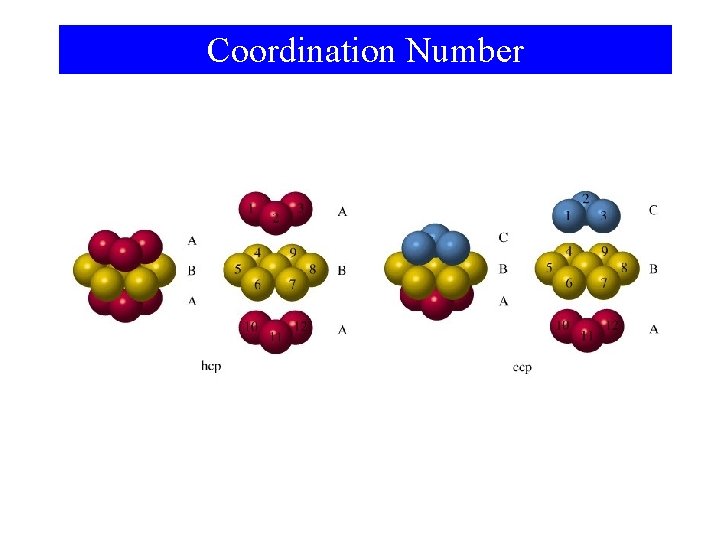
Coordination Number

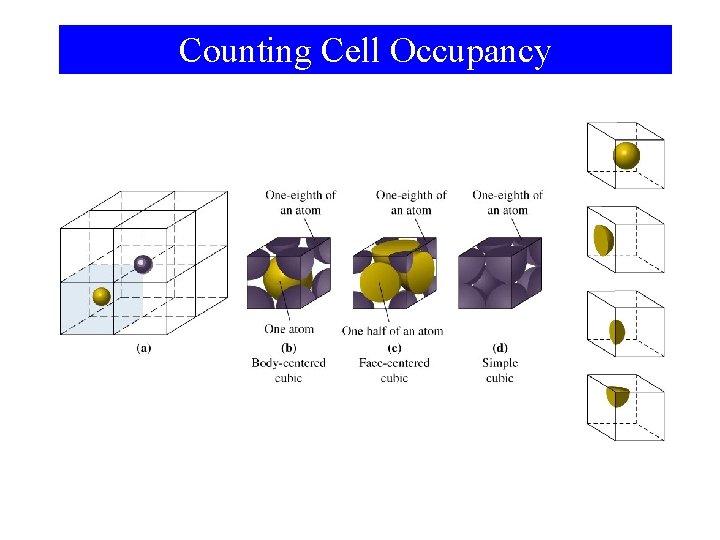
Counting Cell Occupancy

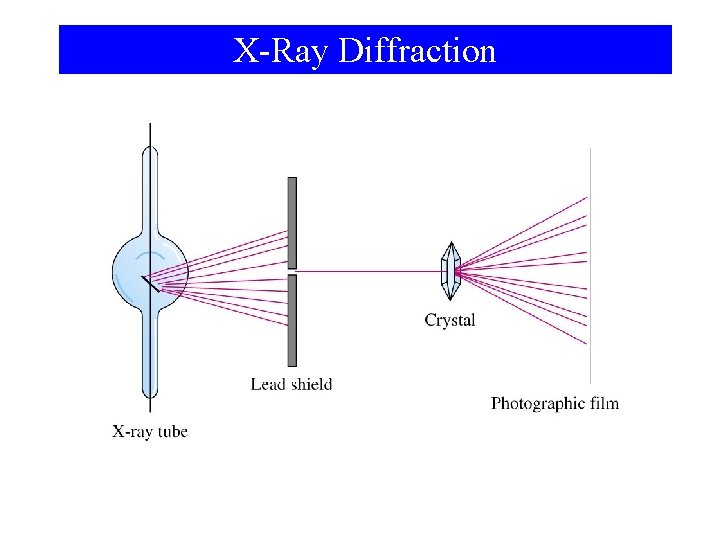
X-Ray Diffraction

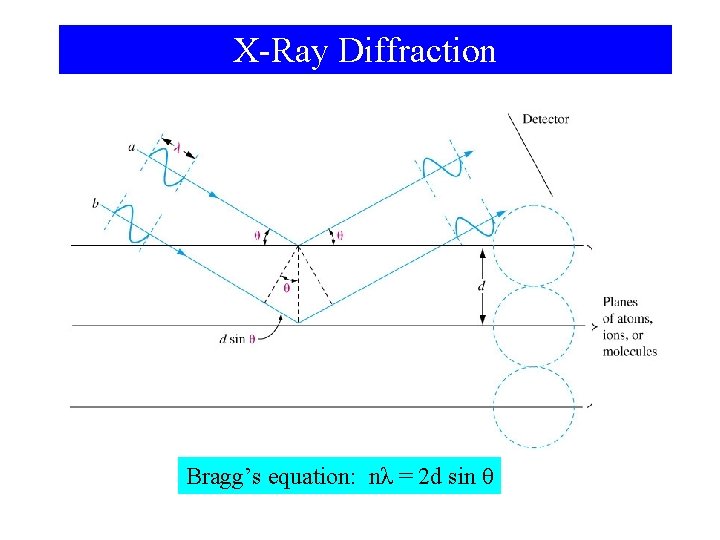
X-Ray Diffraction Bragg’s equation: nλ = 2 d sin θ

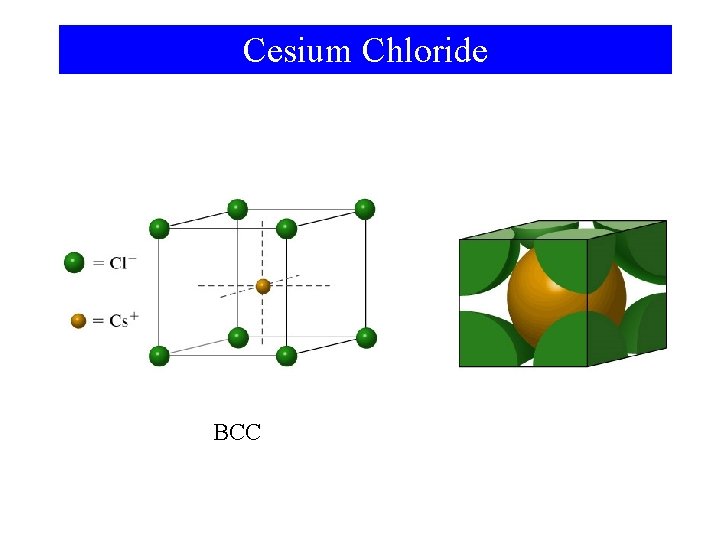
Cesium Chloride BCC

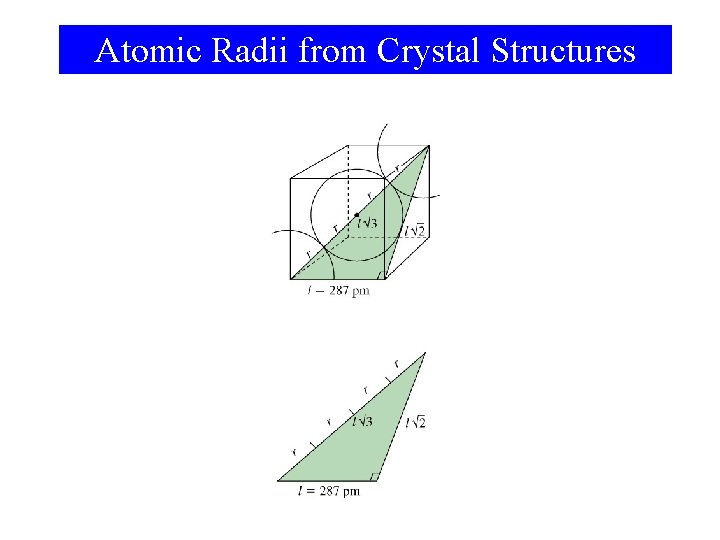
Atomic Radii from Crystal Structures

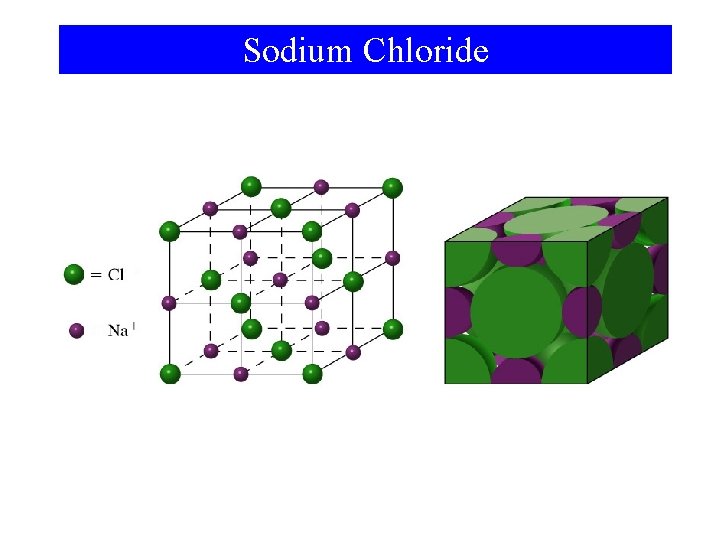
Sodium Chloride

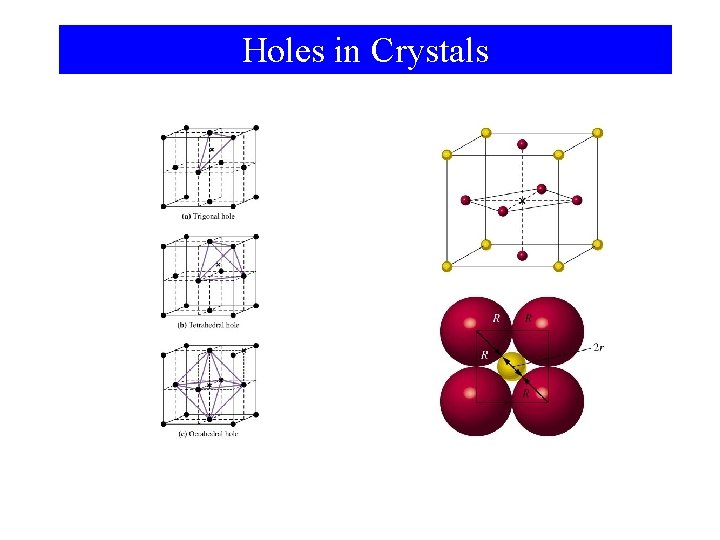
Holes in Crystals

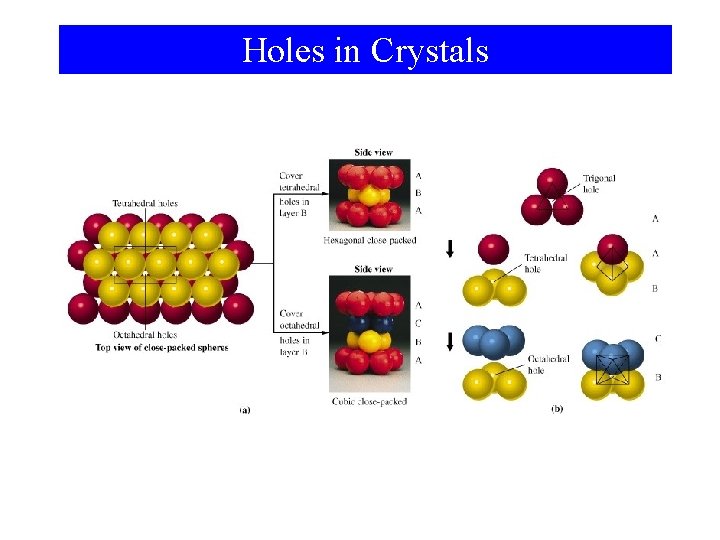
Holes in Crystals

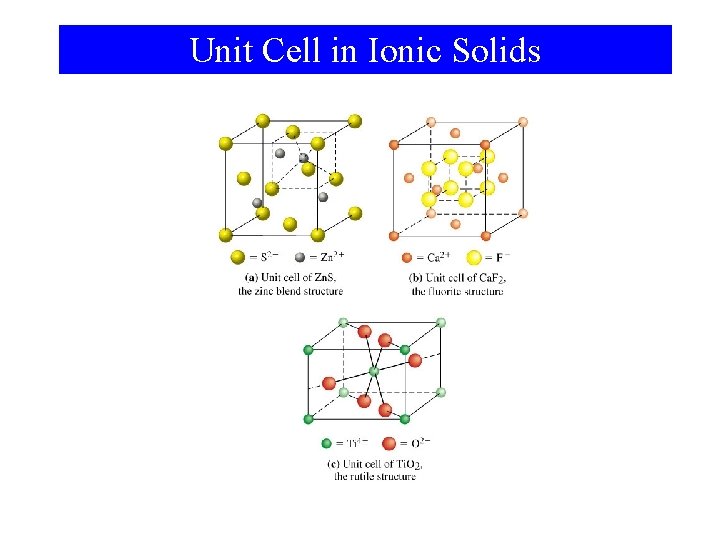
Unit Cell in Ionic Solids

Energy Changes in the Formation of Ionic Crystals

Chapter 11 Questions 9, 14, 16, 20, 23, 28, 33, 34, 46, 53, 58, 64, 70, 74