Electrons in Atoms Chapter 5 Chemistry 11 Early





















![Exceptions to predicted configurations • Chromium- [Ar] 4 s 13 d 5 • Copper Exceptions to predicted configurations • Chromium- [Ar] 4 s 13 d 5 • Copper](https://slidetodoc.com/presentation_image_h/0bd266f4b4d85e63144d2de199ff40c6/image-34.jpg)

















- Slides: 56

Electrons in Atoms Chapter 5

Chemistry 11 Early Models of the Atom

Ancient Greeks were the first to come up with the idea of atoms. Democritus suggested that all matter was made of tiny indivisible particles called atoms. (Greek “atoma”) Democritus

In the early 1800’s, John Dalton came up with the ATOMIC THEORY.

His main points were: Dalton’s Atomic Theory 1) All matter is made of atoms. Atoms are indivisible and indestructible. 2) All atoms of a given element are identical in mass and properties 3) Compounds are formed by a combination of two or more different kinds of atoms. 4) A chemical reaction is a rearrangement of atoms.

crookes tube J. J. Thomson's Experiments Using Crooke’s tubes and other equipment, J. J. Thomson discovered the electron and measured its e/m (charge to mass) ratio. Later, “e” was found and the mass of an electron was found to be 9. 10938188 × 10 -28 grams (much lighter than H)

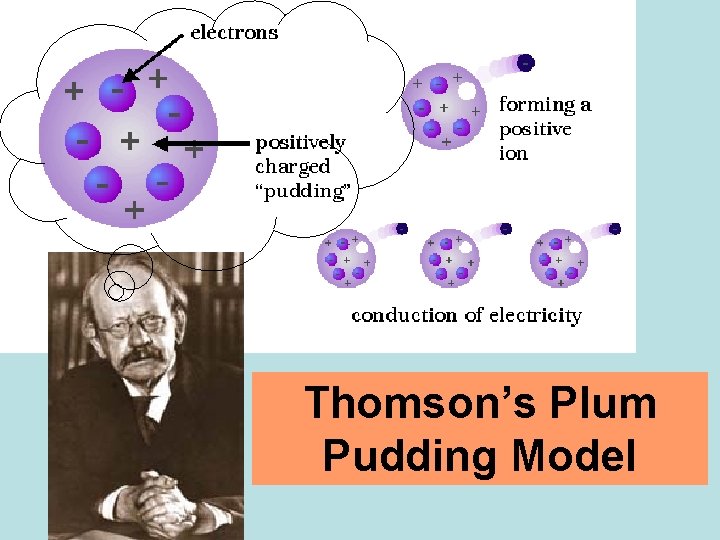
Thomson’s Plum Pudding Model

Ernest Rutherford

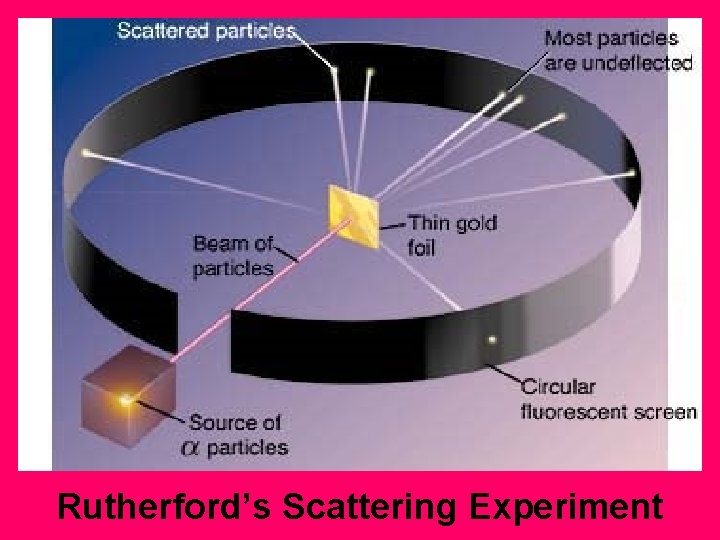
Rutherford’s Scattering Experiment

Rutherford's Experiment

Rutherford could not explain why the electron didn’t fall into the nucleus and destroy the atom.

Neils Bohr

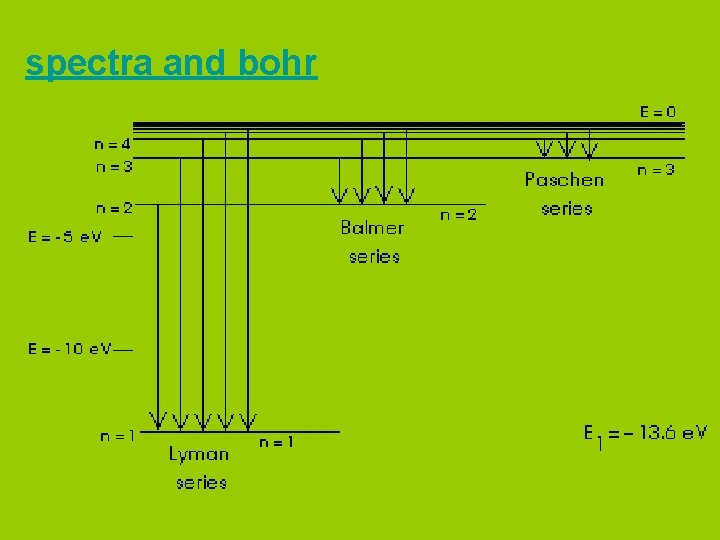
spectra and bohr

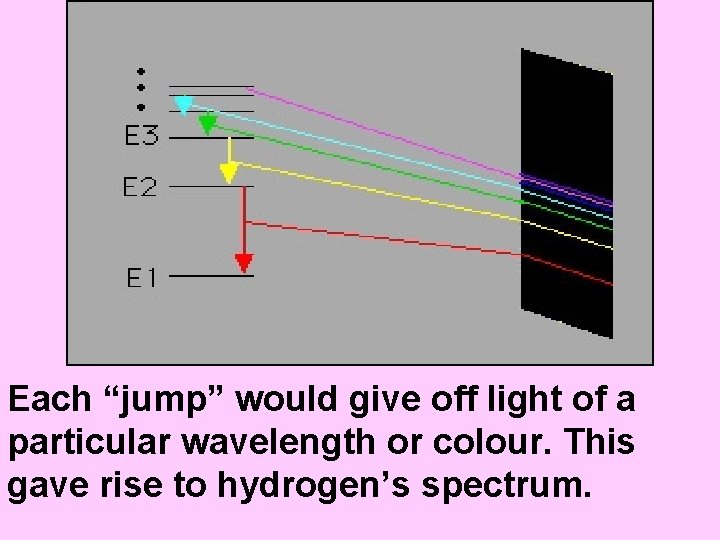
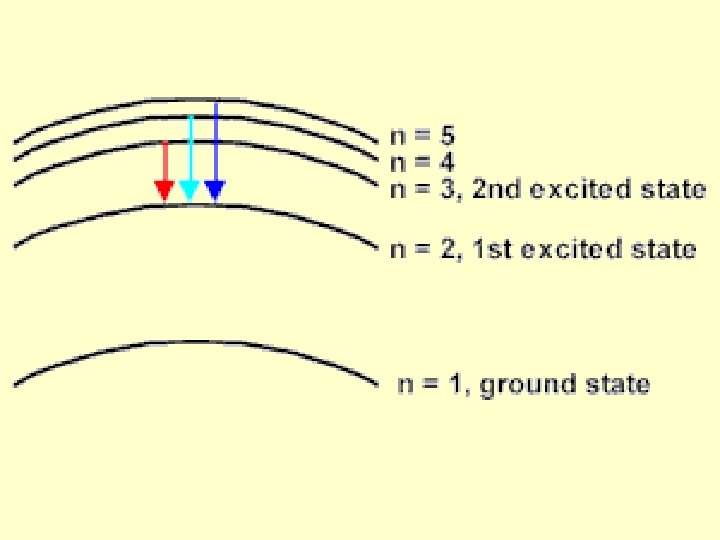
Bohr pictured the hydrogen atom as having discrete energy “levels” which the electron could “inhabit”. In it’s ground state, the electron would be in the lowest level (n=1) When the atom was “excited” the electron could “jump” to a higher level. When the electron came back down, it released energy in the form of light.

Each “jump” would give off light of a particular wavelength or colour. This gave rise to hydrogen’s spectrum.

According to Bohr, each energy “level” corresponded to a different “orbit” of an electron around the atom. (Like planets around the sun. )


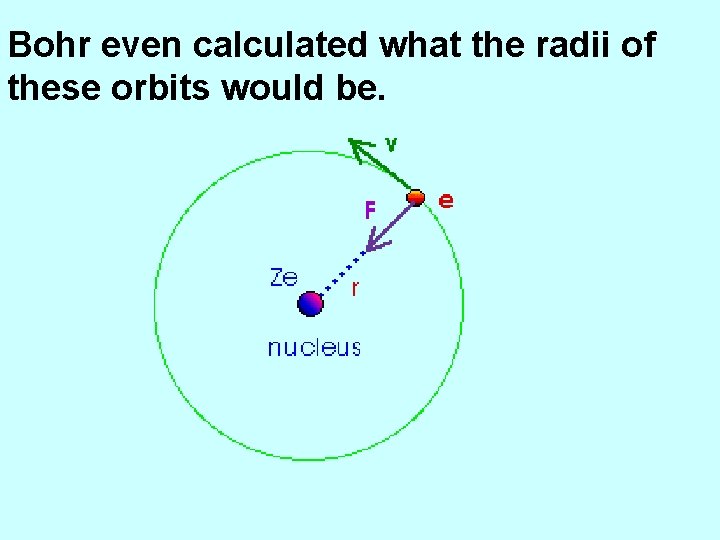
Bohr even calculated what the radii of these orbits would be.

In the 1920’s things changed! Although Bohr’s idea of energy levels was still accepted, his idea of planetary orbits for electrons was rejected! REJECT ! !

The Quantum Mechanical Model (QMM) • 1926 - Austrian physicist Erwin Schrodinger used the results of Rutherford and Bohr to devise and solve a mathematical equation describing the behavior of the electron in a hydrogen atom

• Unlike the Bohr model, the quantum mechanical model does not involve an exact path the electron takes around the nucleus • The quantum mechanical model determines the allowed energies an electron can have and how likely (probability) it is to find the electron in various locations around the nucleus • The cloud is more dense where the probability of finding an electron is high

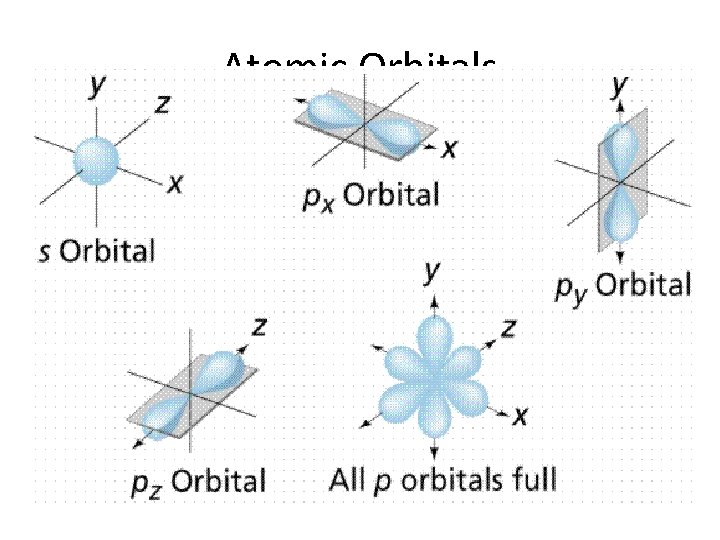
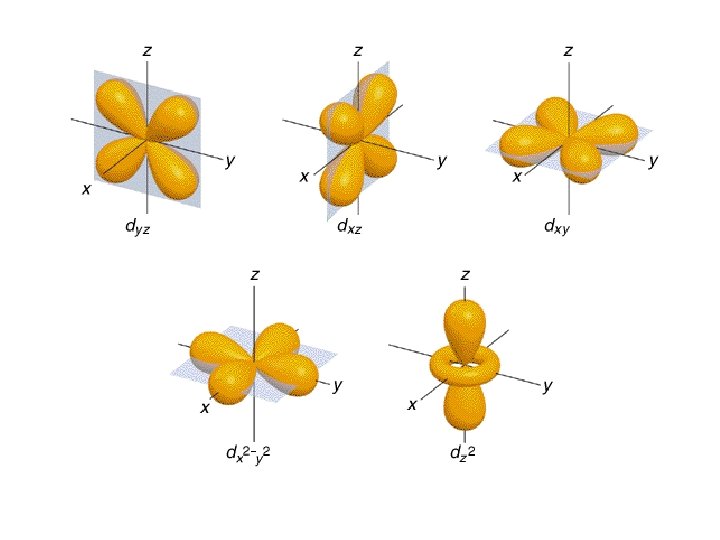
• Atomic Orbital- a 3 D region around the nucleus describing the electron’s probable location

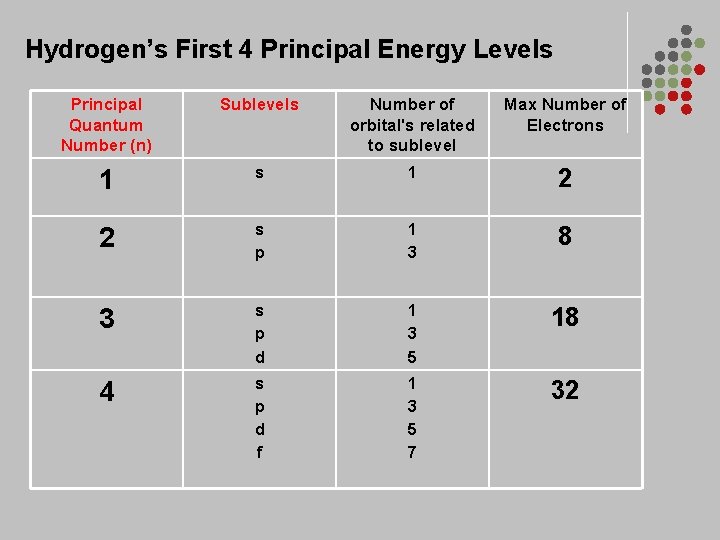
Atomic Orbitals • Energy Levels- are labeled by • Principal Quantum Number (n)- 1, 2, 3, 4… • Within each there are • Energy Sublevels- the energy levels contained with the principal energy level

Atomic Orbitals


Hydrogen’s First 4 Principal Energy Levels Principal Quantum Number (n) Sublevels Number of orbital's related to sublevel Max Number of Electrons 1 s 1 2 2 s p 1 3 8 3 s p d 1 3 5 18 4 s p d f 1 3 5 7 32

Practice read pg 127 -132 Qs 132 on page 1 -7

Electron Arrangement in Atoms • Electrons and the nucleus interact to make the most stable arrangement possible. • Electron Configurations- the ways in which electrons are arranged in various orbitals around the nuclei of atoms

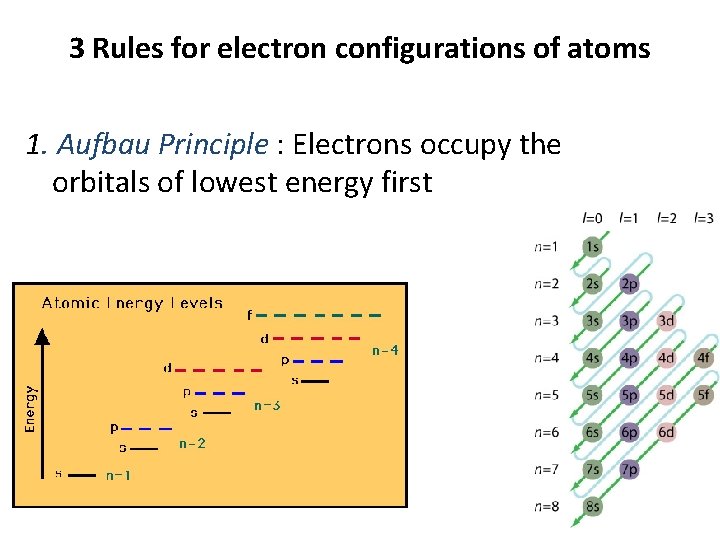
3 Rules for electron configurations of atoms 1. Aufbau Principle : Electrons occupy the orbitals of lowest energy first

2. Pauli Exclusion Principle: an electron orbital may describe at most two electrons – To occupy the same orbital, two electrons must have opposite spins (↓or ↑)

3. Hund’s Rule- electrons occupy orbitals of the same energy in a way that makes the number of electrons with the same spin direction as large as possible.


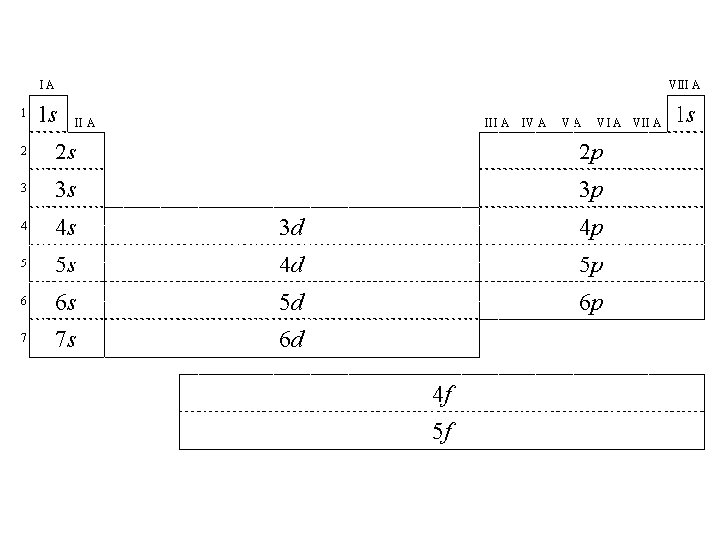
Noble Gas Configuration • Method of representing electron configurations of noble gases using bracketed symbols. – Neon= [Ne] • Also used to shorten electron configurations – Sodium: #11 - instead of 1 s 22 p 63 s 1 can be shortened to [Ne] 3 s 1
![Exceptions to predicted configurations Chromium Ar 4 s 13 d 5 Copper Exceptions to predicted configurations • Chromium- [Ar] 4 s 13 d 5 • Copper](https://slidetodoc.com/presentation_image_h/0bd266f4b4d85e63144d2de199ff40c6/image-34.jpg)
Exceptions to predicted configurations • Chromium- [Ar] 4 s 13 d 5 • Copper - [Ar] 4 s 13 d 10 • Illustrates the increased stability of half-filled and filled sets of s and d orbital's

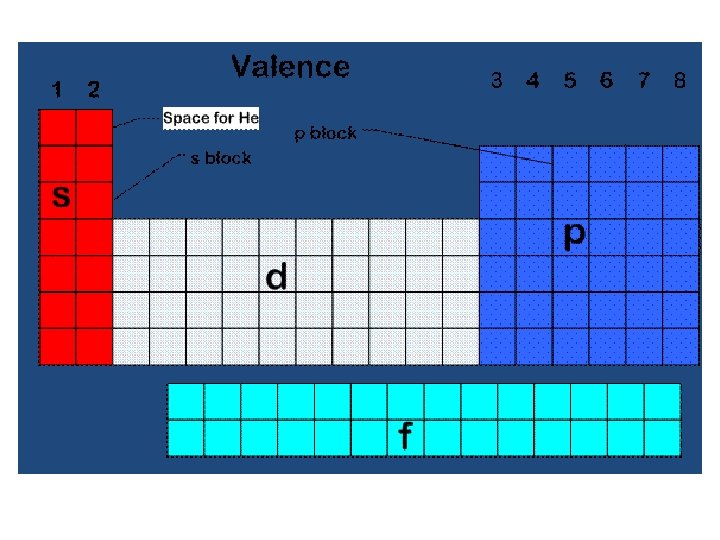
Valence Electrons (V. E. ) • Electrons in the atom’s outermost orbital's • Determine the chemical properties of an element • V. E. are used in forming chemical bonds


Electron Dot Structures • Consists of the element’s symbol and inner-level electrons surrounded by dots representing the atom’s valence electrons • V. E. are placed one at a time on the four sides of the symbol and then paired up until all are used

• Examples


• Chemists found Rutherford’s nuclear model to be lacking because it did not begin to account for the differences in chemical behavior among various elements • Early 1900’s- scientists observed that certain elements emit visible light when heated in a flame chemical behavior

Wave Nature of Light • Electromagnetic Radiation- form of energy that exhibits wavelike behavior as it travels through space

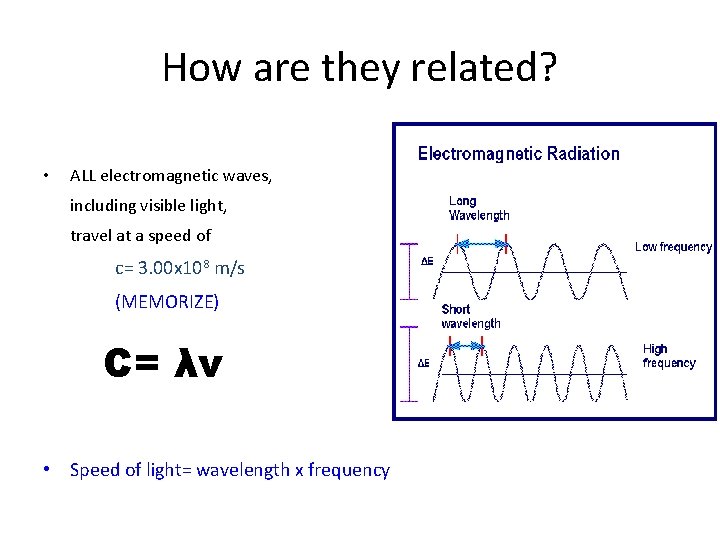
Vocabulary to know. . • Wavelength- shortest distance between equivalent points on a continuous wave – Symbol- λ (lambda) – Unit- meters, centimeters, or nanometers (1 nm= 1 x 10 -9 m) • Frequency- the number of waves that pass a give point per second – Symbol- ν (nu) – Unit- Hertz (SI Unit)= (1/s)= (s-1) • Amplitude- the wave’s height from the origin to a crest, or from the origin to a trough

How are they related? • ALL electromagnetic waves, including visible light, travel at a speed of c= 3. 00 x 108 m/s (MEMORIZE) C= λν • Speed of light= wavelength x frequency

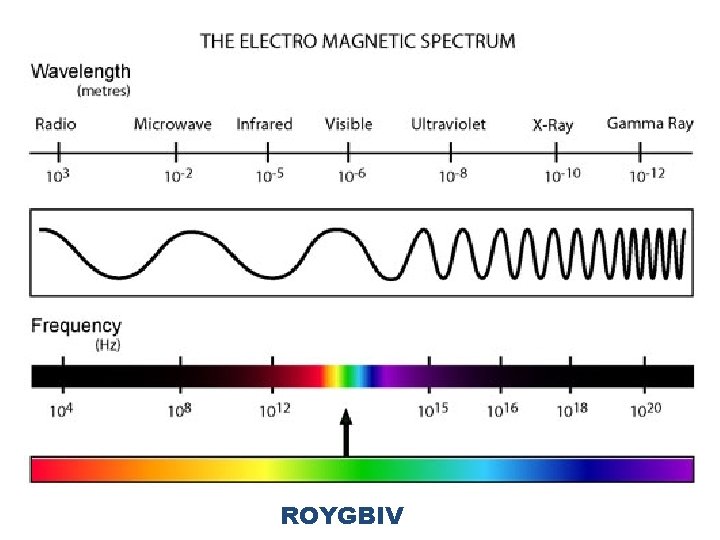
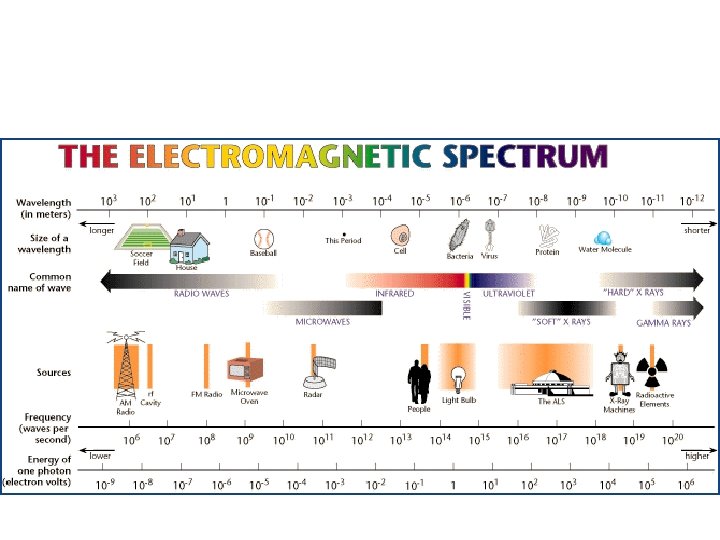
Electromagnetic Spectrum • Aka EM Spectrum • Encompasses all forms of electromagnetic radiation – The only differences in the types of radiation being their wavelengths and frequencies

ROYGBIV


Calculations • Microwaves are used to transmit information. What is the wavelength of a microwave having a frequency of 3. 44 x 109 Hz? – Know: C= λν • C= 3. 00 x 108 m/s • ν = 3. 44 x 109 Hz • λ = ? ? ?

λ = c/ ν λ= 8. 72 x 10 -2 m DON’T FORGET YOUR SIG FIG RULES!!!

Particle Nature of Light • Quantum Concept – Explained why colors of heated matter correspond to different frequencies and wavelengths – Max Plank- “matter can gain or lose only in small, specific amounts called quanta” • Quantum- the minimum amount of energy that can be gained or lost by an atom

• Energy of a quantum is related to the frequency of the emitted radiation by the equation Equantum= hv – E= energy – h = Plank’s Constant (6. 626 x 10 -34 J) – v= frequency – Joule (J)= SI unit for energy

Photoelectric Effect • Electrons, called photoelectrons, are emitted from a metal’s surface when light of a certain frequency shines on the surface – Photon- a particle of EM radiation with no mass that carries a quantum of energy Ephoton= hv

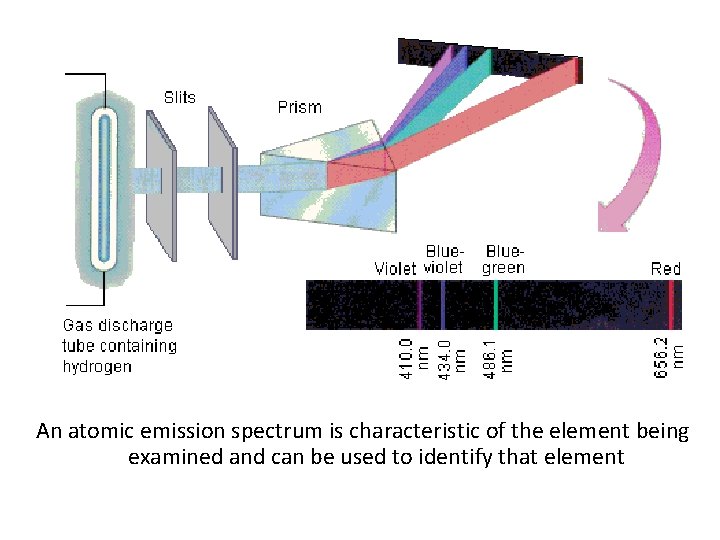
Atomic Emission Spectra • Set of frequencies of the electromagnetic waves emitted by atoms of the element – Example- The light of neon sign is produced by passing electricity through a tube filled with neon gas. Neon atoms release energy by emitting light.

An atomic emission spectrum is characteristic of the element being examined and can be used to identify that element

Section 5. 2 Bohr Model of the Atom • Proposed that the hydrogen atom has only certain allowable energy states • Ground State- lowest allowable energy state of an atom

• Bohr’s model worked well to explain Hydrogen - however it did not explain other elements • Substantial evidence indicates that electrons do not move around the nucleus in circular orbits

• The de Broglie equation predicts that all moving particles have wave like characteristics λ= h/mv • The Heisenburg uncertainty principal - states that it is fundamentally impossible to know precisely both the velocity and position of a particle at the same time