Electrons in Atoms Electron configuration Electrons z The













![Notation z. Shorthand Configuration S 16 e [Ne] Noble Gas 2 4 3 s Notation z. Shorthand Configuration S 16 e [Ne] Noble Gas 2 4 3 s](https://slidetodoc.com/presentation_image_h/343047a63bb05c1d31d4cb59ecbe5940/image-19.jpg)

- Slides: 22

Electrons in Atoms Electron configuration

Electrons z. The arrangement of electrons in an atom will determine the chemical and physical properties of that atom (or ion). z. Because of this it is important to know where those electrons are (or are most likely to be found)

Where do we find electrons? Electrons in atoms are arranged as LEVELS (n) SUBLEVELS (l) ORBITALS (ml) Think of these as the electrons address.

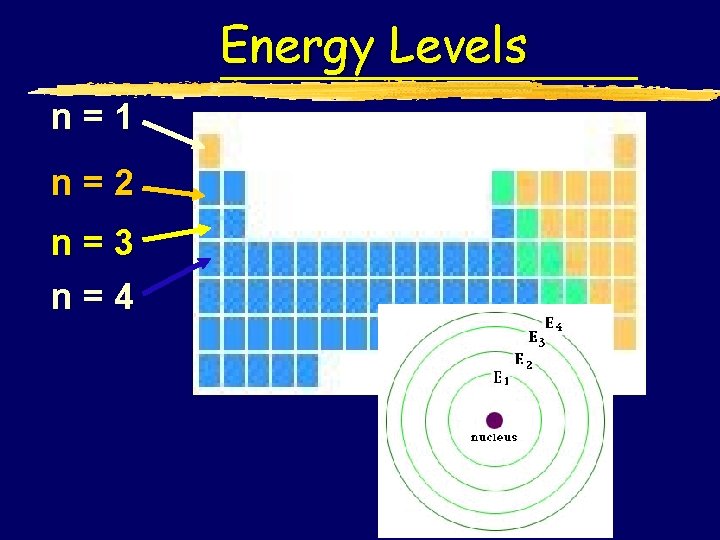
Energy Level (n) This tells you how far the electron is from the nucleus, the higher the energy level the further from the nucleus z. Currently n can be 1 thru 7, because there are 7 periods on the periodic table

Energy Levels n=1 n=2 n=3 n=4

Sublevels zattempt to describe where the electrons are likely to be found ys, p, d, f ORBITALS z. Found within the sublevels Each orbital can hold a maximum of 2 electrons

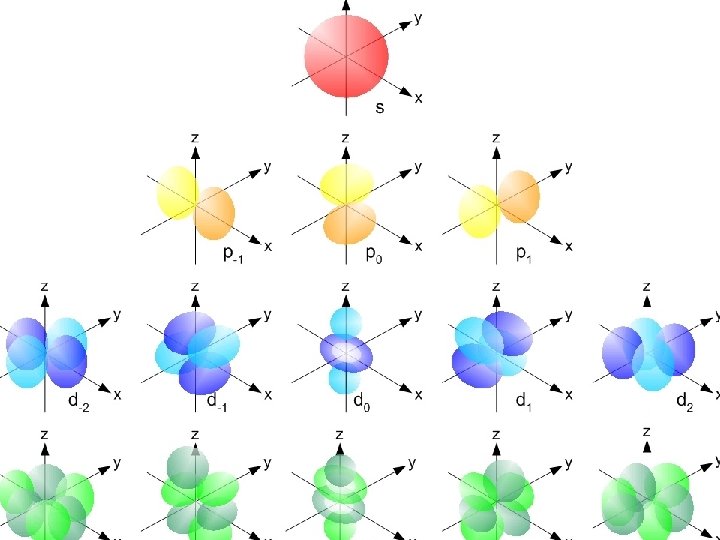
Types of Orbitals z. The most probable area to find these electrons takes on a shape z. So far, we have 4 shapes. They are named s, p, d, and f. z. Each orbital has different “flavors” ys= 1 flavor yp = 3 flavors yd = 5 flavors yf = 7 flavors x. Each “flavor” can hold a maximum of 2 electrons



How many electrons can be in a sublevel? Remember: A maximum of two electrons can be placed in an orbital. s orbitals p orbitals d orbitals f orbitals Number of orbitals 1 3 5 7 Number of electrons 2 6 10 14

Electron possible per energy level 2 n 2 = the number of electrons possible in an energy level z. Ex: 3 rd energy level, n=3 2(3)2 = 18

Electron Configurations 4 2 p Energy Level Sublevel Number of electrons in the sublevel 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2 4 f 14… etc.

General Rules z. Pauli Exclusion Principle y. Each orbital can hold TWO electrons with opposite spins.

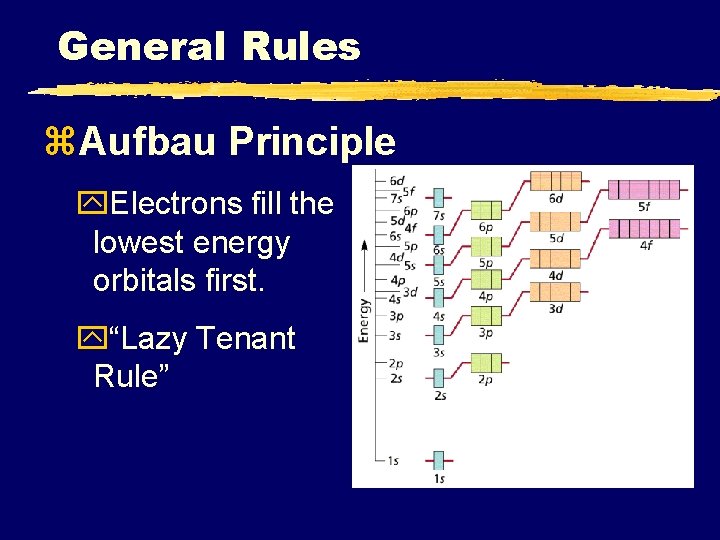
General Rules z. Aufbau Principle y. Electrons fill the lowest energy orbitals first. y“Lazy Tenant Rule”

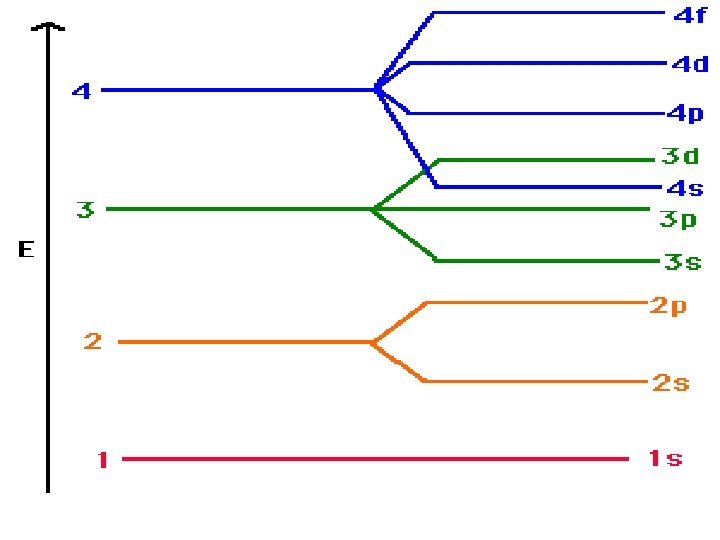
Diagonal Rule Steps: 1 s 2 s 3 s 1. Write the energy levels top to bottom. 2. Write the orbitals in s, p, d, f order. Write the same number of orbitals as the energy level. 3. Draw diagonal lines from the top right to the bottom left. 4. To get the correct order, 2 p 3 p 3 d follow the arrows! 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 5 g? 6 s 6 p 6 d 6 f 6 g? 6 h? 7 s 7 p 7 d 7 f 7 g? 7 h? By this point, we are past the current periodic table so we can stop. 7 i?

General Rules z. Hund’s Rule y. Within a sublevel, place one e- per orbital before pairing them. y“Empty Bus Seat Rule” WRONG RIGHT

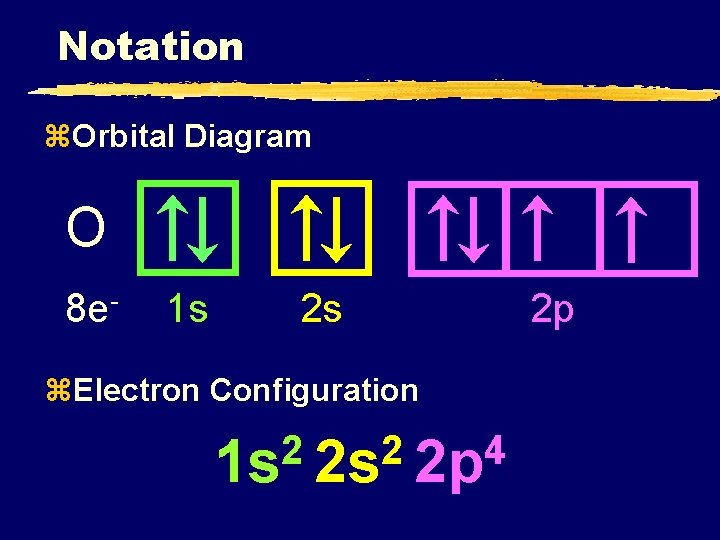
Notation z. Orbital Diagram O 8 e- 1 s 2 s z. Electron Configuration 2 2 4 1 s 2 s 2 p 2 p

Notation z. Longhand Configuration S 16 e 6 2 2 2 1 s 2 s 2 p 3 s Core Electrons 4 3 p Valence Electrons
![Notation z Shorthand Configuration S 16 e Ne Noble Gas 2 4 3 s Notation z. Shorthand Configuration S 16 e [Ne] Noble Gas 2 4 3 s](https://slidetodoc.com/presentation_image_h/343047a63bb05c1d31d4cb59ecbe5940/image-19.jpg)
Notation z. Shorthand Configuration S 16 e [Ne] Noble Gas 2 4 3 s 3 p Valence Electrons

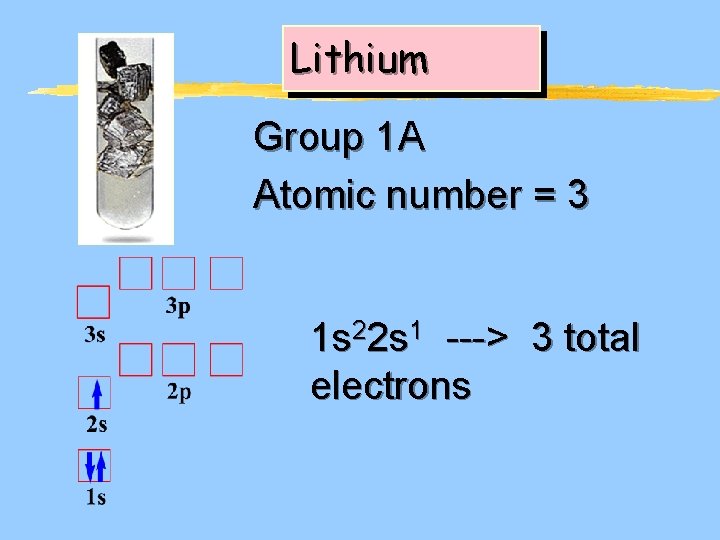
Lithium Group 1 A Atomic number = 3 1 s 22 s 1 ---> 3 total electrons

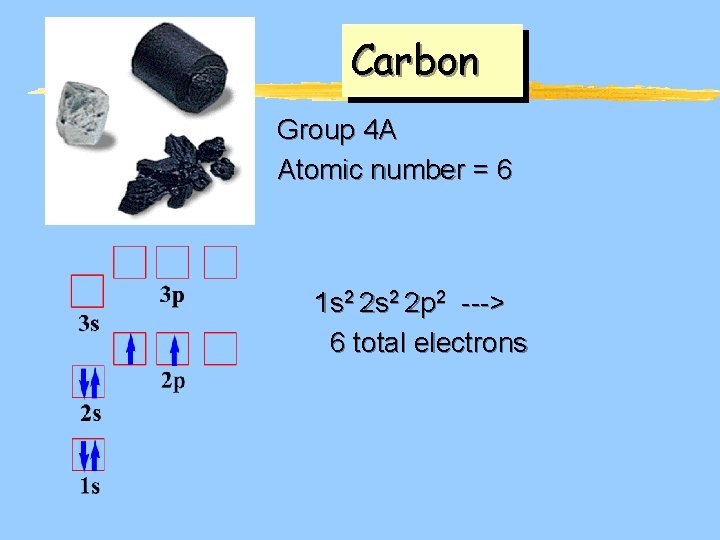
Carbon Group 4 A Atomic number = 6 1 s 2 2 p 2 ---> 6 total electrons

http: //www. youtube. com/watch? v=Vb 6 k. Axw. SWg. U