Electron Configuration The arrangement of electrons in an









- Slides: 23

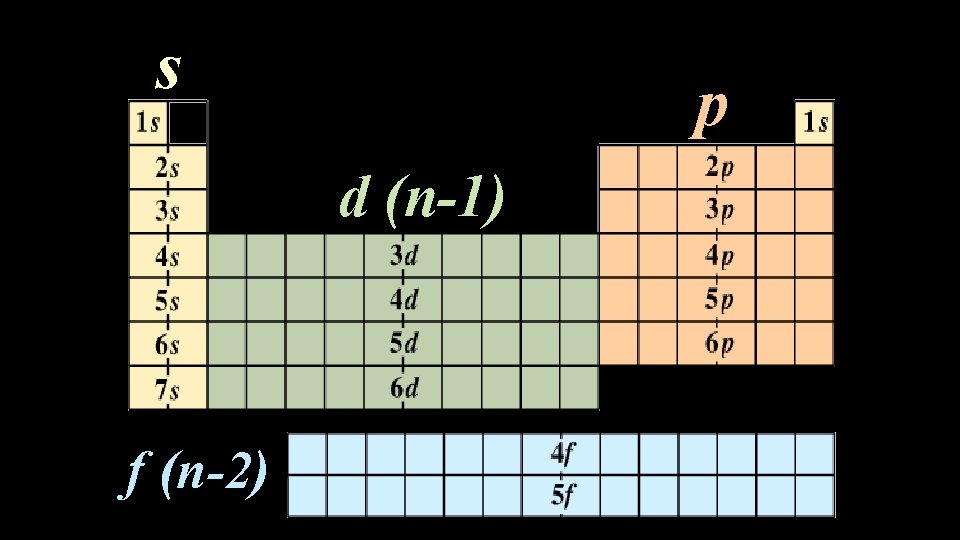
Electron Configuration: • The arrangement of electrons in an atom s 1 2 3 4 5 6 7 f (n-2) p d (n-1) 6 7 © 1998 by Harcourt Brace & Company

Electron Configuration: • Quantum model states that electrons are found in Orbitals: • a 3 -D space around the nucleus (“electron cloud”) • Electron Orbitals: s p d f

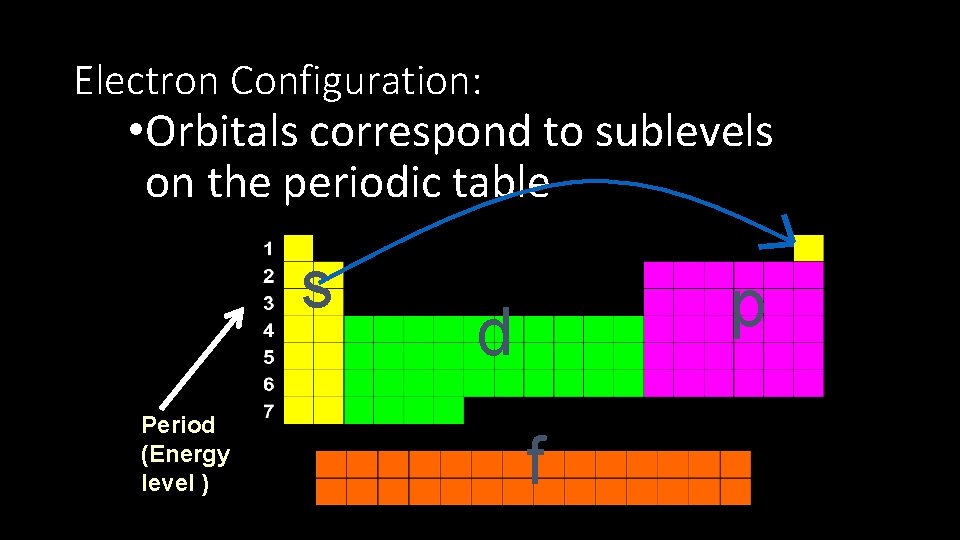
Electron Configuration: • Orbitals correspond to sublevels on the periodic table s Period (Energy level ) p d f

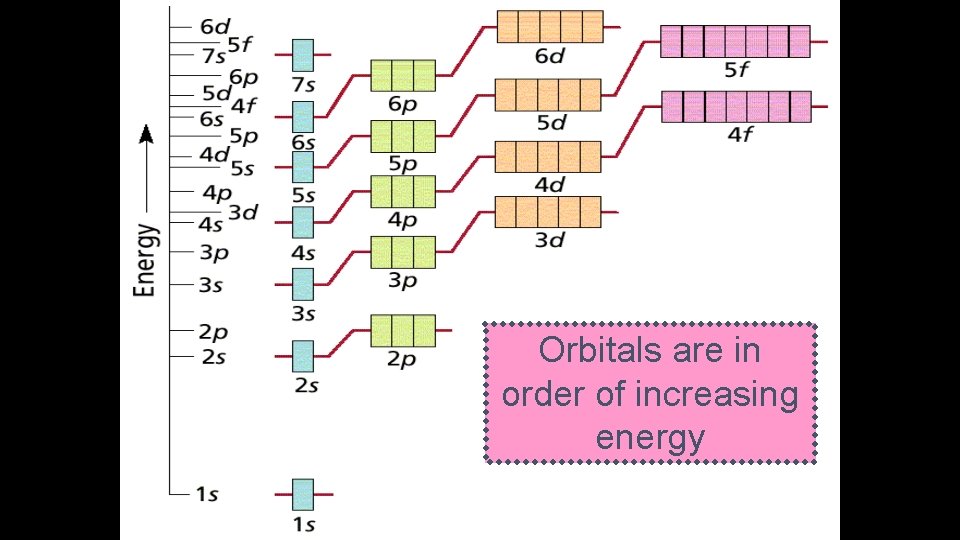
Electron Configuration Rules: 1. Aufbau Principle § Electrons occupy the lowest energy orbitals first - “Lazy Tenant Rule” § s p d f

Orbitals are in order of increasing energy

Electron Configuration Rules: 2. Pauli Exclusion Principle: § Each orbital can hold TWO electrons with opposite spins. (Orbital diagram notation)

Electron Configuration Rules: 3. Hund’s Rule § Within a sublevel, place one e per orbital before pairing them. § “Empty Bus Seat Rule” WRONG RIGHT

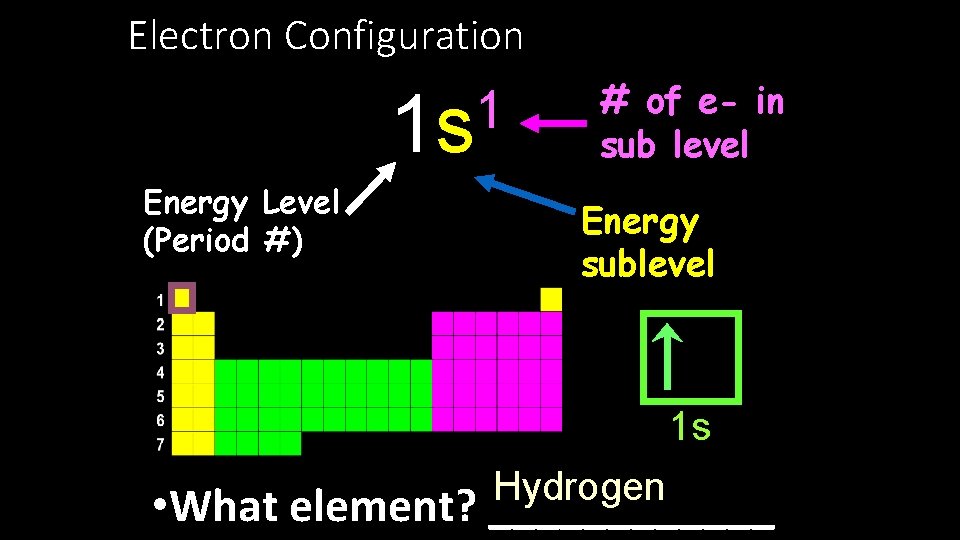
Electron Configuration 1 1 s Energy Level (Period #) # of e- in sub level Energy sublevel 1 s Hydrogen • What element? ______

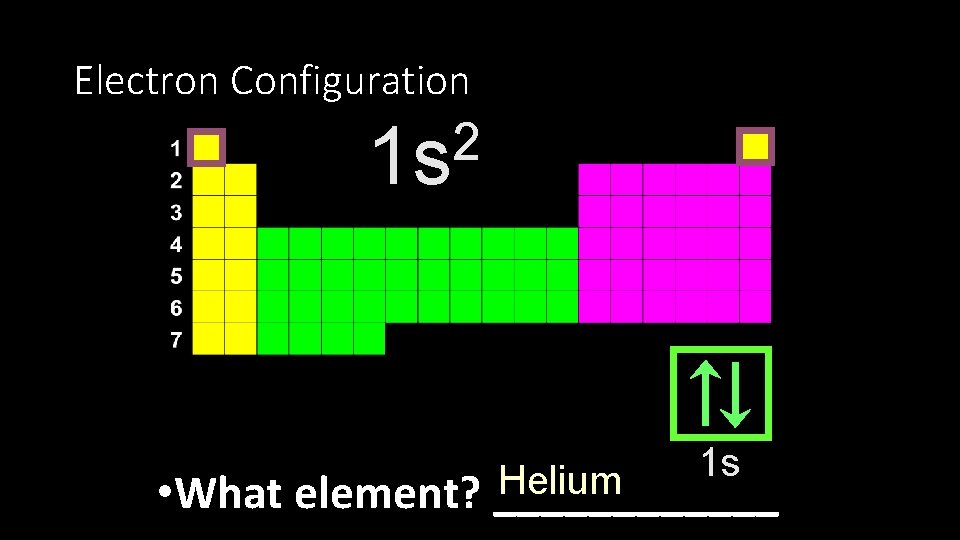
Electron Configuration 2 1 s 1 s Helium • What element? ______

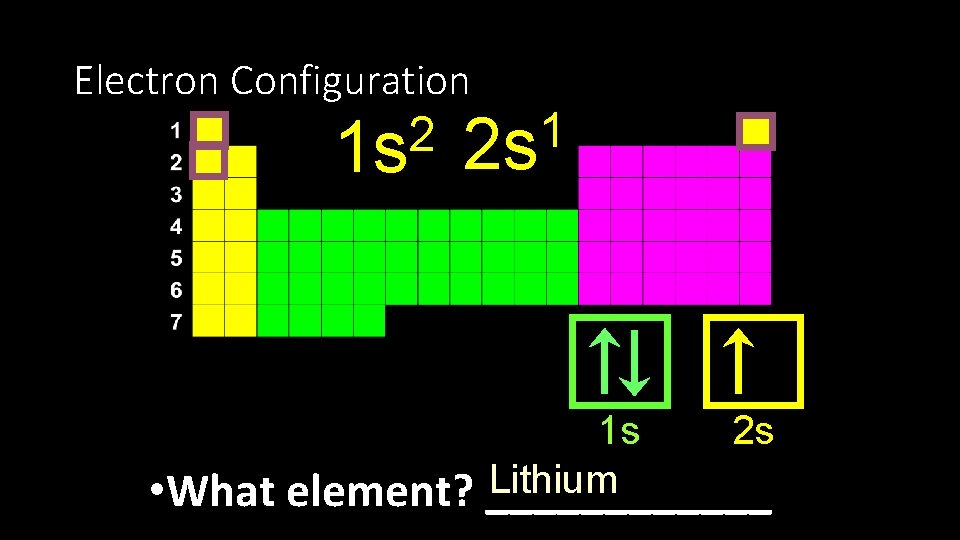
Electron Configuration 2 1 s 1 2 s 2 s 1 s Lithium • What element? ______

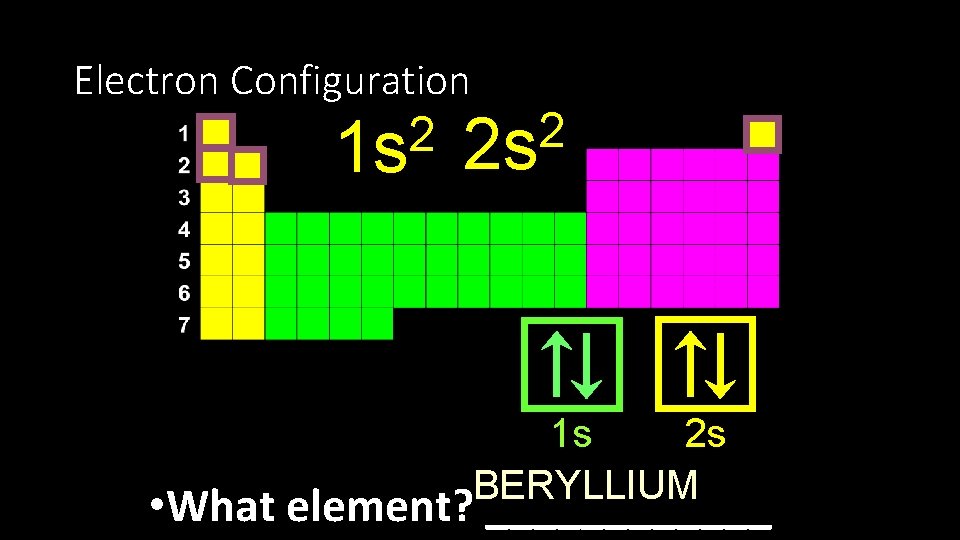
Electron Configuration 2 1 s 2 2 s 2 s 1 s BERYLLIUM • What element? ______

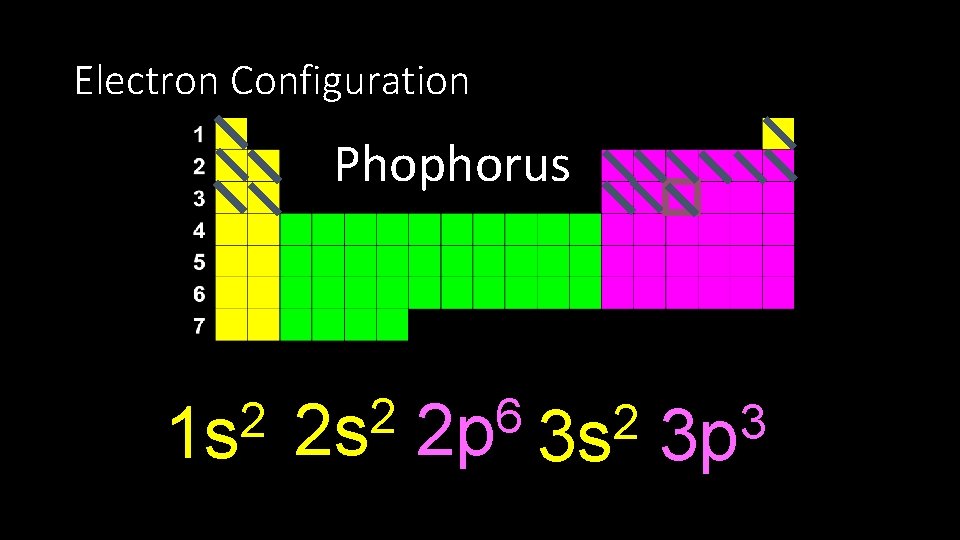
Electron Configuration Phophorus 2 1 s 2 2 s 6 2 p 2 3 s 3 3 p

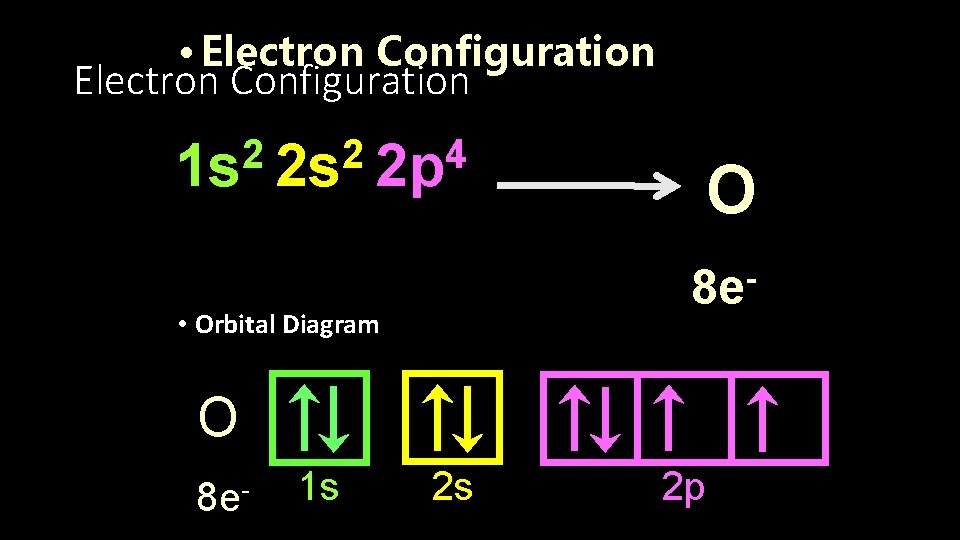
• Electron Configuration 2 2 4 1 s 2 s 2 p O 8 e- • Orbital Diagram O 8 e- 1 s 2 s 2 p

s p d (n-1) f (n-2) © 1998 by Harcourt Brace & Company

Core vs. Valence Electrons Core electrons: Valence Electrons: electrons found on the outerinner energy most energy levels 2 1 s 2 2 s 6 2 p 2 3 s 3 3 p

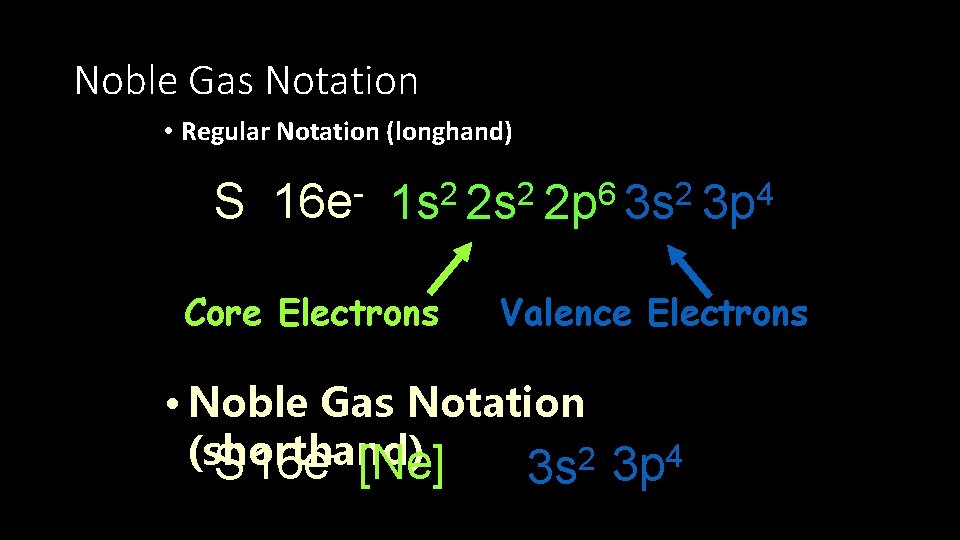
Core vs. Valence Electrons S 16 e 1 s 2 2 p 6 3 s 2 3 p 4 Core Electrons Mg 12 e- Valence Electrons 6 2 2 2 1 s 2 s 2 p 3 s

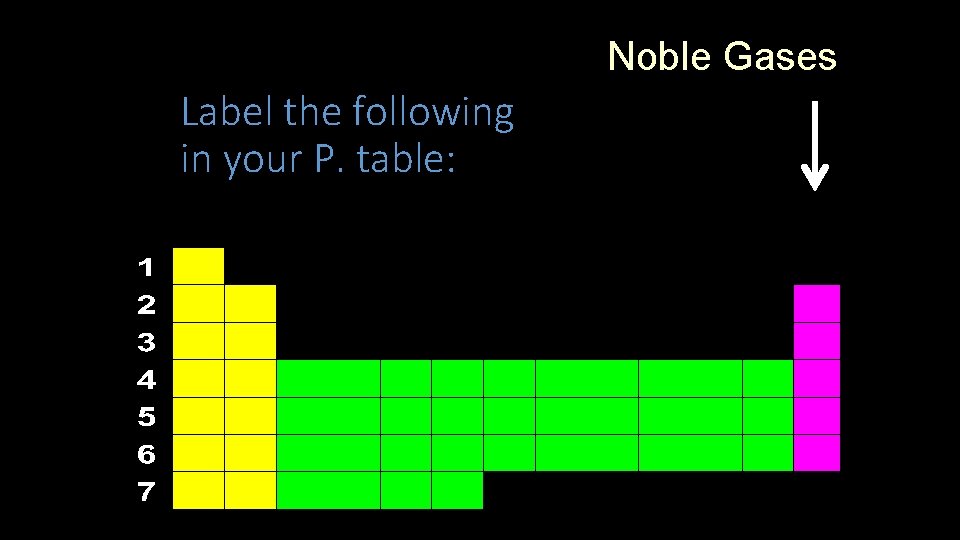
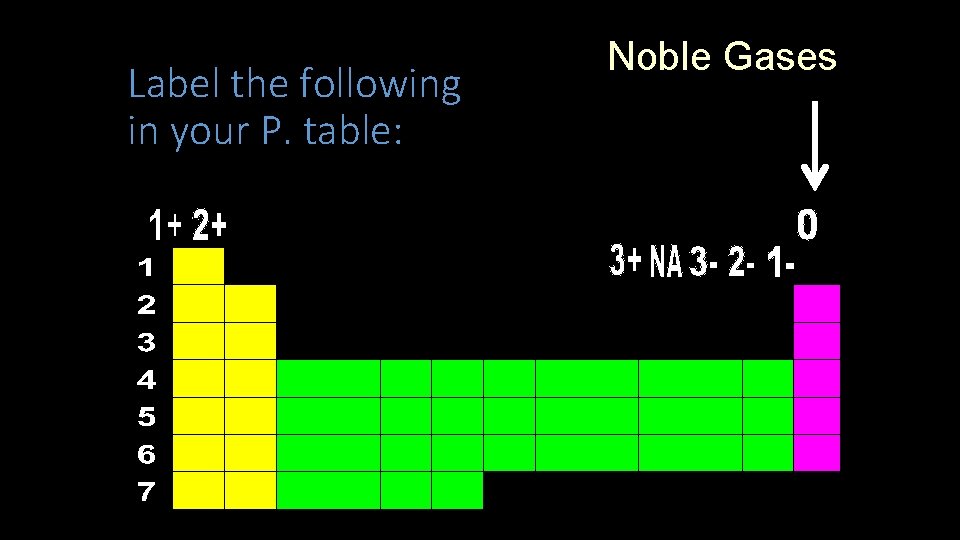
Noble Gases Label the following in your P. table:

Noble Gas Notation • Noble Gas Notation 1. 2. Write the noble gas of previous row in brackets (Core e-) Fill in the rest of the configuration (Valence e-). Ex: Ca 20 e [Ar] 4 s 2 Cl 17 e [Ne]3 s 23 p 5

Noble Gas Notation • Regular Notation (longhand) S 16 e 1 s 2 2 p 6 3 s 2 3 p 4 Core Electrons Valence Electrons • Noble Gas Notation (shorthand) 4 2 S 16 e- [Ne] 3 s 3 p

Ion Notation • Ion- atom with a charge because of a gain or lose valence electrons to become more stable. • Noble Gases are already stable • Other atoms will try to be like the Noble Gases

Label the following in your P. table: Noble Gases

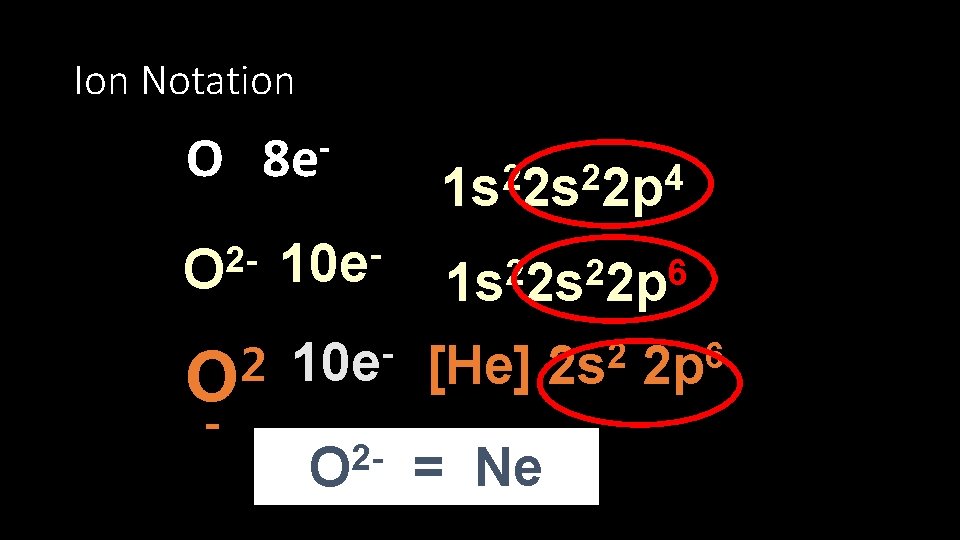
Ion Notation O 8 e 2 O 2 O - 10 e 2 2 4 1 s 2 s 2 p 2 2 6 1 s 2 s 2 p 10 e [He] 2 O = Ne 2 2 s 6 2 p

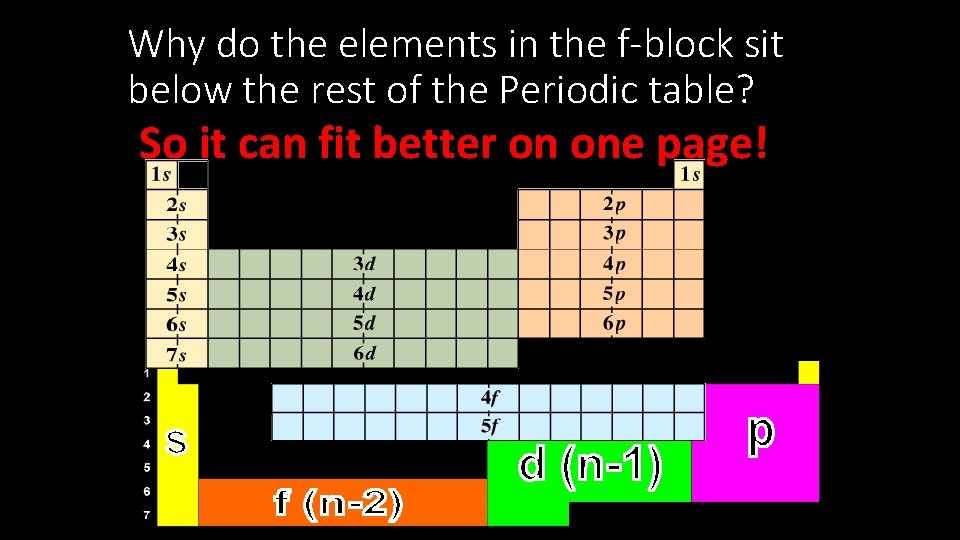
Why do the elements in the f-block sit below the rest of the Periodic table? So it can fit better on one page!