Electron Configurations Objectives List the total number of

Electron Configurations

Objectives List the total number of electrons needed to fully occupy each main energy level n State the Aufbau principle, the Pauli exclusion principle, and Hund’s rule n Describe the electron configurations for the atoms of any element using orbital notation, electron-configuration notation, and when appropriate, noble-gas notation n

Electron Configuration n Arrangement of electrons in an atom n Atoms occupy the lowest energy arrangement—ground-state electron configuration

Rules n 1. Aufbau principle: principle electrons enter orbitals of lowest energy levels first n Orbitals in same sublevel (s, p, d, f) are of equal energy n s sublevel has lowest energy n Sublevels in different main levels can overlap n 1 s is the lowest energy orbital
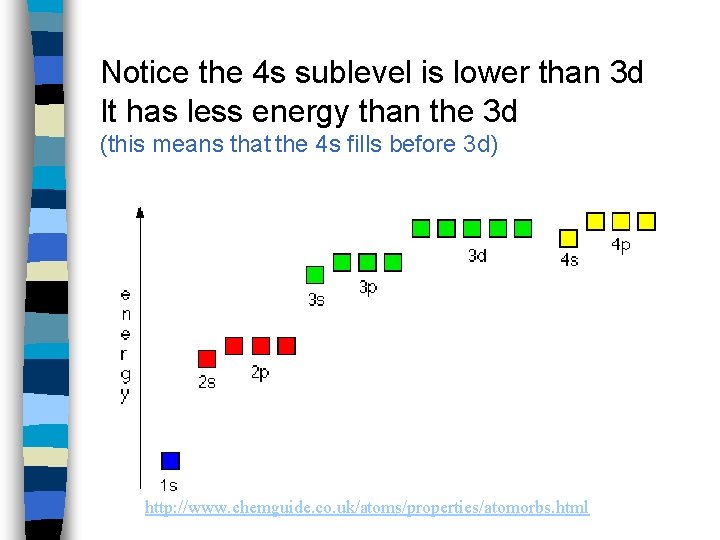
Notice the 4 s sublevel is lower than 3 d It has less energy than the 3 d (this means that the 4 s fills before 3 d) http: //www. chemguide. co. uk/atoms/properties/atomorbs. html

Text p. 105 Figure 4 -16 “The order of the atomic orbitals can be somewhat difficult to remember. Fortunately, there is a mnemonic device that can help in this regard. The slide sequence below details this device. ” http: //www. iun. edu/%7 Ecpanhd/C 101 webnotes/modern-atomic-theory/mnemonicdev. html

Rules n 2. Pauli exclusion principle: principle quantum numbers of atomic orbitals may describe only one electron n Two electrons in the same orbital must have opposite spins n Spins are clockwise & counterclockwise 1 s orbital

Rules n 3. Hund’s Rule: Rule when electrons occupy orbitals of equal energy, one electron enters each orbital until all the orbitals contain one electron with parallel spins n Sublevels add one electron to each orbital first n Second electrons added to the orbitals are then paired with an opposite spin n a p orbitals filling below b c
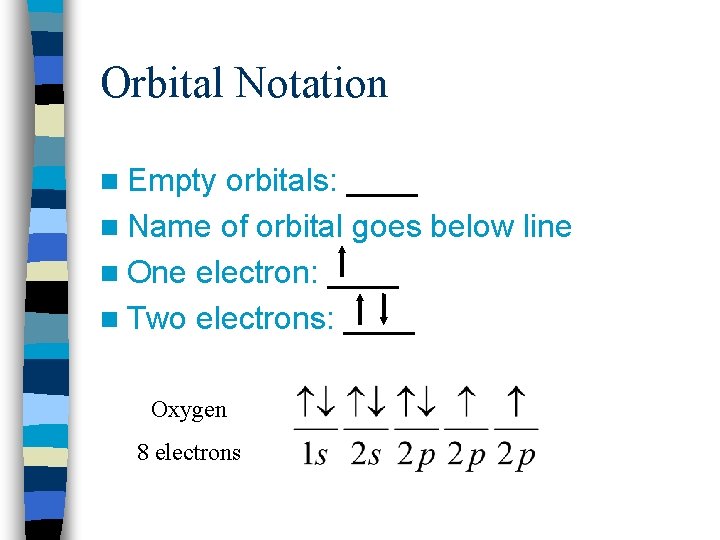
Orbital Notation n Empty orbitals: ____ n Name of orbital goes below line n One electron: ____ n Two electrons: ____ Oxygen 8 electrons

Electron-Configuration Notation n Eliminate lines and arrows n Number of electrons in a sublevel is the superscript Oxygen 8 electrons 1 s 2 2 p 4 http: //glencoe. mcgrawhill. com/olcweb/cgi/pluginpop. cgi? it=swf: : 800: : 600: : /sites/dl/free/007874637 x /514701/chem_ch 05_t 05_4. swf: : Electron%20 Configurations%20 and%20 Orbit al%20 Diagrams%20 for%20 Elements%201– 10

Practice n Write the orbital notation for Al. the electron-configuration notation for Mn.

Practice n The electron configuration of nitrogen is 1 s 2 2 p 3. How many electrons are present in nitrogen? What is the atomic number of nitrogen? Write the orbital notation for nitrogen.

Practice n The electron configuration of fluorine is 1 s 2 2 p 5. What is the atomic number of fluorine? How many of its p orbitals are filled? How many unpaired electrons does a fluorine atom contain?
- Slides: 13