Electron Arrangement Chemistry 6E Electron Arrangement Lesson Objectives

Electron Arrangement Chemistry 6(E)

Electron Arrangement Lesson Objectives • Express the arrangement of electrons in atoms using – Electron configurations – Lewis valence electron dot structures

Atomic Orbitals • Heisenberg uncertainty principle – it’s impossible to know the exact velocity and position of a particle at the same time • Atomic orbitals – areas where electrons are likely to be found – Four types of orbitals • s, p, d, and f s-orbital shape p-orbital shape d-orbital shapes – Quantum numbers – sets of numbers that describe the properties of atomic orbitals and the electrons in them

Principle Energy Levels • Principle quantum number – (n) represents an atomic orbital’s size and principle energy level • Electrons that are further away from the nucleus have more energy • Electrons in larger orbitals are more likely to be further from the nucleus n=2 n=1 nucleus Larger orbitals = more E

Quantum Numbers – Sublevels are labeled using their principle energy level and orbital type • Higher principle energy levels are composed of more sublevels Ex) Principle energy level 1 contains one n sublevel Orbital type 1 s { • Principle energy levels can be divided into sublevels Sublevel Ex) Principle energy level 2 contains two 2 s sublevels 2 p
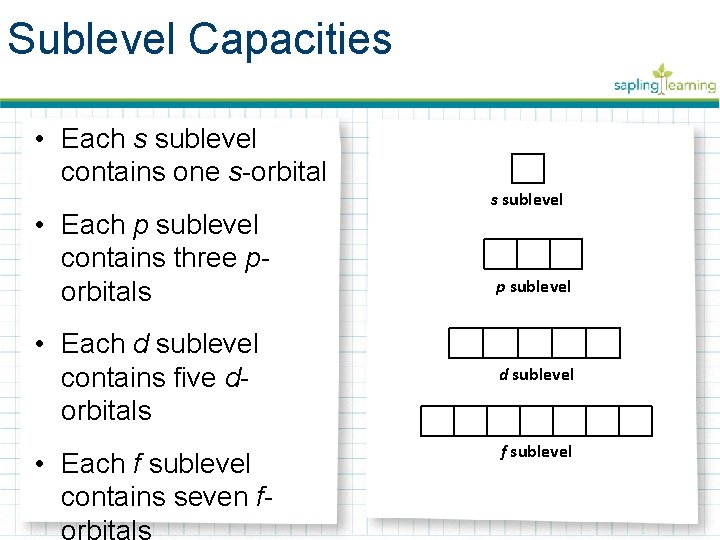
Sublevel Capacities • Each s sublevel contains one s-orbital • Each p sublevel contains three porbitals • Each d sublevel contains five dorbitals • Each f sublevel contains seven forbitals s sublevel p sublevel d sublevel f sublevel
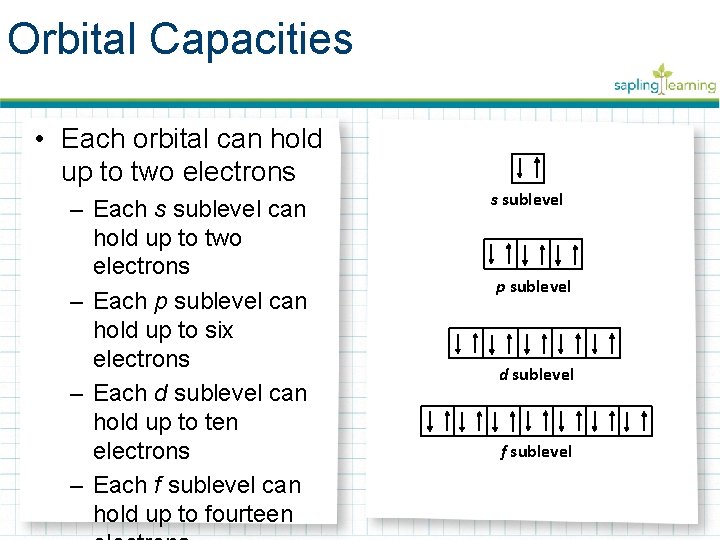
Orbital Capacities • Each orbital can hold up to two electrons – Each s sublevel can hold up to two electrons – Each p sublevel can hold up to six electrons – Each d sublevel can hold up to ten electrons – Each f sublevel can hold up to fourteen s sublevel p sublevel d sublevel f sublevel

Ground-State Electron Configurations • Electron configuration – describes an atom’s electron arrangement • Systems with lower energy are more stable • Ground-state electron configuration – arrangement of electrons that gives an atom the least possible energy – Three rules govern how electrons are arranged in ground -state electron configurations: • Aufbau principle • Pauli exclusion principle • Hund’s rule

Aufbau Principle • Electrons achieve their ground-state when they occupy the closest available orbital to the nucleus • Aufbau Principle – electrons fill available orbitals with the least energy first • Aufbau diagrams show the order of orbitals from least to greatest energy
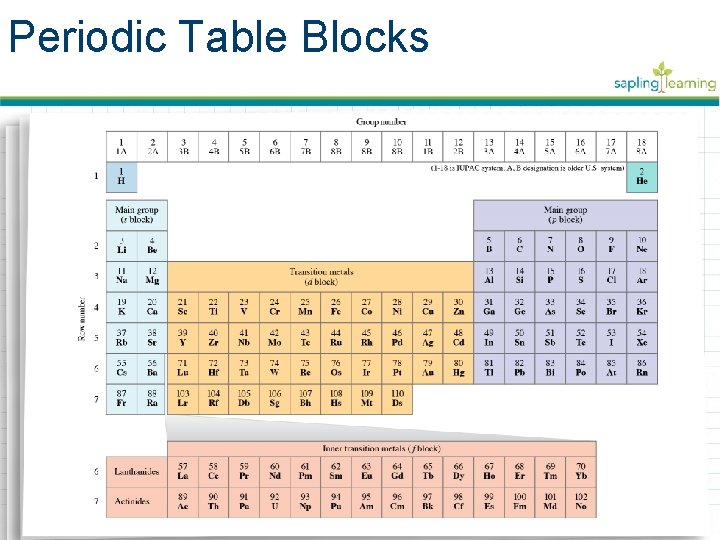
Periodic Table Blocks
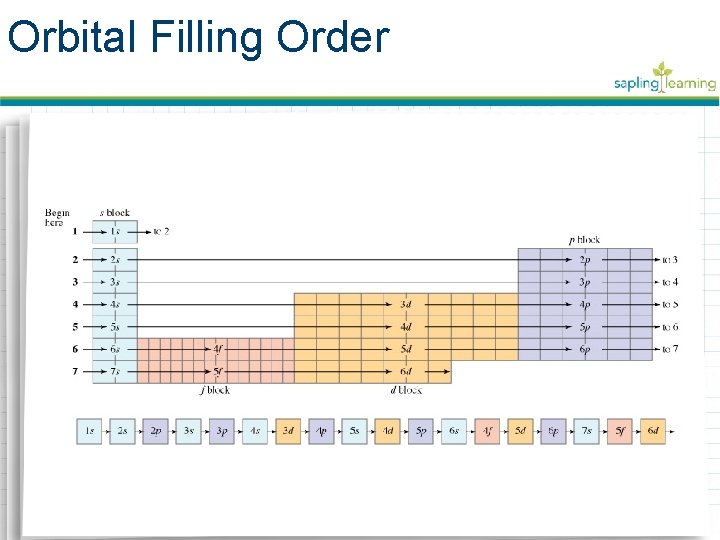
Orbital Filling Order

Pauli Exclusion Principle • Pauli exclusion principle – each orbital can hold a maximum of two electrons – When in the same orbital electrons must have opposite spin – Direction of the electron’s spin is represented the direction of the arrow

Hund’s Rule 2 p Energy • Negative charges of electrons repel each other • Hund’s rule – when filling equal energy orbitals, electrons fill each orbital singly before filling orbitals with another electron in them 2 s 1 s

FSGPT How would you describe the order that the electrons fill the orbitals in?
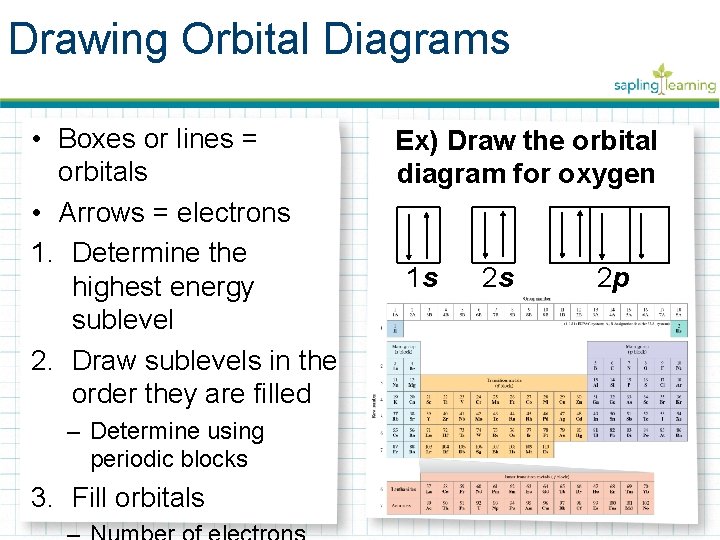
Drawing Orbital Diagrams • Boxes or lines = orbitals • Arrows = electrons 1. Determine the highest energy sublevel 2. Draw sublevels in the order they are filled – Determine using periodic blocks 3. Fill orbitals Ex) Draw the orbital diagram for oxygen 1 s 2 s 2 p

Electron Configuration Notation 1. Determine the highest energy sublevel of the atom 2. Write the sublevels in the order they are filled – Determine using periodic blocks 3. Write the number of electrons in each sublevel as a superscript Ex) Write the electron configuration of iron 2 2 6 2 6 1 s 2 s 2 p 3 s 3 p 4 s 3 d
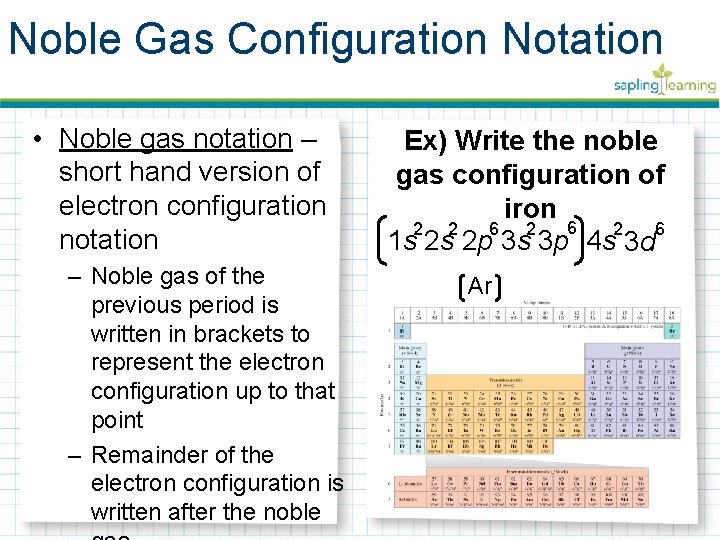
Noble Gas Configuration Notation • Noble gas notation – short hand version of electron configuration notation – Noble gas of the previous period is written in brackets to represent the electron configuration up to that point – Remainder of the electron configuration is written after the noble Ex) Write the noble gas configuration of iron 6 6 2 2 6 1 s 2 s 2 p 3 s 3 p 4 s 3 d Ar

Valence Electrons • • Valence Electrons Valence electrons – electrons found in the highest occupied energy level • Outermost electrons of the electron cloud • Establish the chemical characteristics of elements • Only electrons represented in Lewis electron dot structures • Symbolized by dots
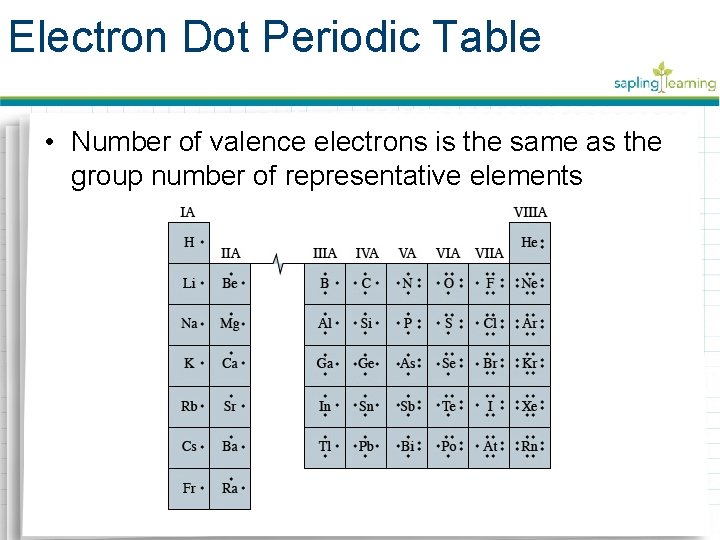
Electron Dot Periodic Table • Number of valence electrons is the same as the group number of representative elements

Writing Lewis Electron Dot Structures • Number of valence electrons is the same as the group number of representative elements Ex) Group 5 A elements have 5 valence electrons – N is in group 5 A N

Writing Electron Dot Structures 1. Identify the number of valence electrons using the periodic table 2. Place the corresponding number of electron dots around the symbol – First assign one dot per side – If there are still more Ex) Dot Structure for Phosphorus P

FSGPT • What is the relationship between valence electrons and where the elements are located on the periodic table?

Valence Electrons and Ion Formation • Ions form when electrons are lost or gained • Ionic charges are based on the number of electrons lost or gained • Losing or gaining electrons changes the ratio of positive particles (p+) to negative particles (e-) and causes an overall charge to form Ex) Na lost 1 e- to become Na+ 10 e 11 p+ + +1 charge F gained 1 e- to become F 10 e 9 p+ + -1 charge

Valence Electrons and Ion Formation • Generally, charge can be be determined by an ion’s group number • Metals lose valence electrons to form cations • Nonmetals gain valence electrons to form anions 1+ 2+ 3+ 3 - 2 - 1 -

Electron Dot Structures for Ions • Excess or deficit of electrons will be represented by – Number of electrons around the symbol – Charge outside the dot structure Ex) Neutral calcium becomes a calcium ion 2+ Ca Ex) Neutral sulfur becomes a sulfide ion S 2 -

Electron Arrangement Lesson Objectives • Express the arrangement of electrons in atoms using – Electron configurations – Lewis valence electron dot structures
- Slides: 26