Electron Configurations and the Periodic Table The electron


Electron Configurations and the Periodic Table • The electron configuration of an atom’s highest occupied energy level generally governs the atom’s chemical properties (the highest occupied level of the noble gases contain stable octets – outer s and p orbitals are completely filled with 8 electrons. • The exception is helium (2 electrons in highest occupied energy level). • Review valence electrons.
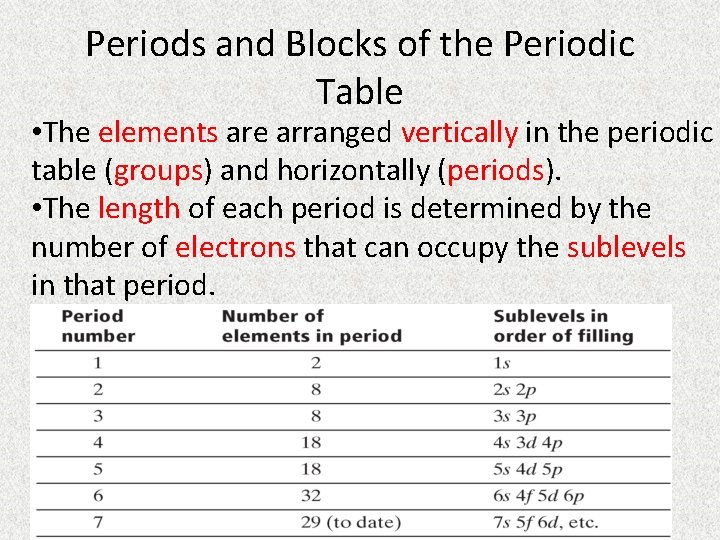
Periods and Blocks of the Periodic Table • The elements are arranged vertically in the periodic table (groups) and horizontally (periods). • The length of each period is determined by the number of electrons that can occupy the sublevels in that period.

• The period of an element can be determined from its electron configuration. • Example: Arsenic, As, has the electron configuration [Ar]3 d 104 s 24 p 3. • The highest occupied energy level is n=4 so As is in the 4 th period. Electron configuration is [Kr]4 d 55 s 1. Period 5!!
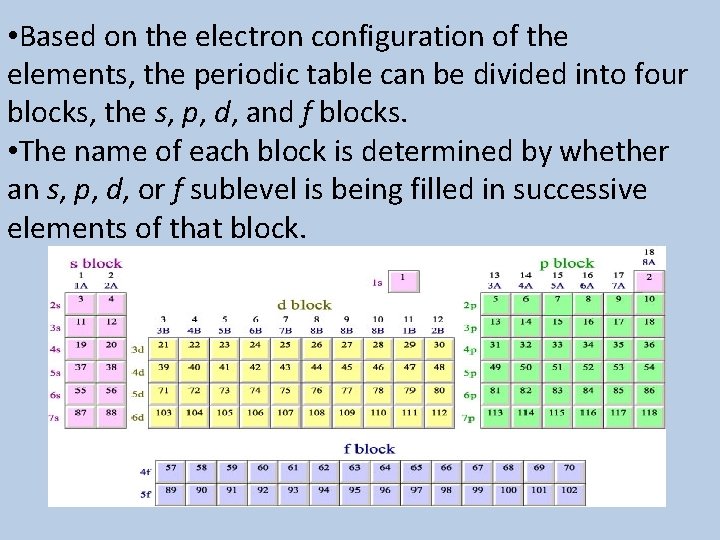
• Based on the electron configuration of the elements, the periodic table can be divided into four blocks, the s, p, d, and f blocks. • The name of each block is determined by whether an s, p, d, or f sublevel is being filled in successive elements of that block.

The s-Block Elements: Groups 1 and 2 • Elements in the same group share similar chemical properties. • Elements in groups 1 (alkali metals) and 2 (alkalineearth metals) are chemically reactive. • The outermost energy level in an atom of each Group 1 element contains a single s electron. This electron is lost with ease which helps to make these metals extremely reactive. • The outermost energy level in an atom of each Group 2 element contains 2 s electrons. This makes them slightly less reactive.

• Group 1 elements have a silvery appearance and are soft enough to cut with a knife. • They combine vigorously with most nonmetals. • They are extremely reactive with air or moisture and must be stored in kerosene.

• Group 2 metals are harder, denser, and stronger than group 1 metals. • They are also less reactive. • Although they are less reactive than alkali metals, they are too reactive to be found in nature as free elements. Calcium Barium

Hydrogen and Helium • Special cases in the classification of elements. • Hydrogen is located above Group 1 but has unique properties. • Helium has configuration like Group 2 but is in Group 18 due to chemical stability.

The d-Block Elements: Groups 3 -12 • The d-block elements (transition metals) have metallic properties. • They are good conductors of heat and have a high luster. Copper Mercury Tungsten

• These elements are less reactive than the elements in Groups 1 and 2. • Some are so unreactive that they do not easily form compounds, existing in nature as free elements. • Palladium, platinum, and gold are among the least reactive of all the elements. Palladium Platinum Gold

The p-Block Elements: Groups 13 -18 • The properties of these elements vary greatly because the p-block consists of nonmetals, and metalloids. • Group 17 (halogens) are the most reactive nonmetals. • They react vigorously with most metals to form salts. Chlorine Iodine Bromine

• The metalloids have properties of metals and nonmetals. • They are the most brittle solids. Silicon

• The metals are harder and denser than the metals in Groups 1 and 2, but softer and less dense than transition metals. • With the exception of bismuth, these metals are sufficiently reactive to be found in nature only in the form of compounds. • Once obtained as free metals, however, they are stable in the presence of air. Bismuth Aluminum

The f-Block Elements: Lanthanides and Actinides • Located between Groups 3 and 4 in the 6 th and 7 th periods. • Lanthanides are shiny metals similar in reactivity to the Group 2 metals. Cerium Europium

• Actinides are radioactive. • The first four have been found naturally on Earth and the remaining actinides were made in a laboratory. Uranium Thorium
- Slides: 16