Unit 5 Electron Configuration Pauli Exclusion Principle No























![Example X X X - Germanium X X X X X [Ar]4 s Example X X X - Germanium X X X X X [Ar]4 s](https://slidetodoc.com/presentation_image_h2/2164d3aa788131e9c14d8d8b62c3f479/image-39.jpg)


![Copper Expect: [Ar] 4 s 2 3 d 9 Actual: [Ar] 4 s Copper Expect: [Ar] 4 s 2 3 d 9 Actual: [Ar] 4 s](https://slidetodoc.com/presentation_image_h2/2164d3aa788131e9c14d8d8b62c3f479/image-43.jpg)

![Write the Lewis Oxygen (O) [He] Iron structures for: 2 s 2 2 Write the Lewis Oxygen (O) [He] Iron structures for: 2 s 2 2](https://slidetodoc.com/presentation_image_h2/2164d3aa788131e9c14d8d8b62c3f479/image-48.jpg)
- Slides: 48

Unit. 5 Electron Configuration

Pauli Exclusion Principle No two electrons have the same quantum #’s Maximum electrons in any orbital is two ( )

How are The two electrons arranged in an atom natures of electromagnetic radiation: Particles vs. Waves How to use the periodic table to list the configuration or orbital diagram What quantum numbers are and how they are related to electron configuration. How the periodic table is arranged The basic periodic trends

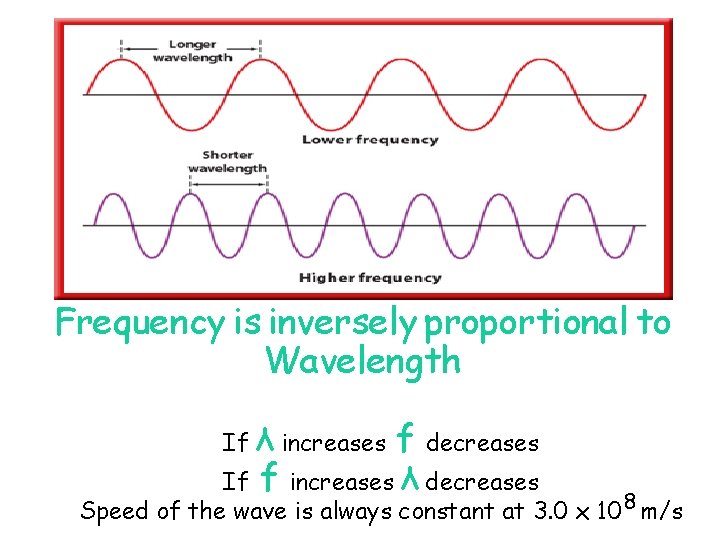
Wave Nature of Electromagnetic Radiation Property of Waves Frequency ▪ No. of waves per second Wave Length ▪ Distance between corresponding points in a wave Amplitude ▪ Size of the wave peak


Frequency is inversely proportional to Wavelength If λ increases f decreases If f increases λ decreases Speed of the wave is always constant at 3. 0 x 108 m/s

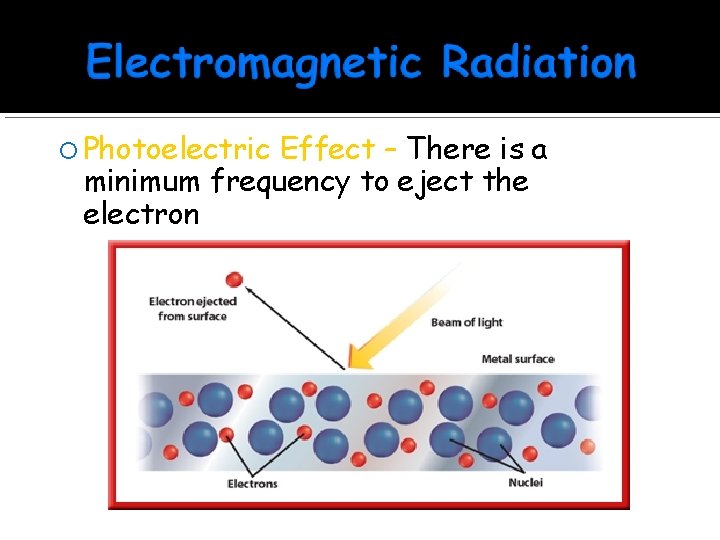
Particle of Light Nature Wave nature could not explain all observations (Plank & Einstein) Photoelectric Effect When light strikes a metal electrons are ejected Atomic Line Spectra ▪ When elements are heated, they emit a unique set of frequencies of visible and non-visible light.

Photoelectric Effect – There is a minimum frequency to eject the electron

Photoelectric Only explained by “energy packets” of light called a quantum E=hf Quantum - minimum amount of energy that can be gained or lost by an atom H = Plank’s constant Photons are massless particles of light of a certain quantum of energy Effect As frequency increases energy increases

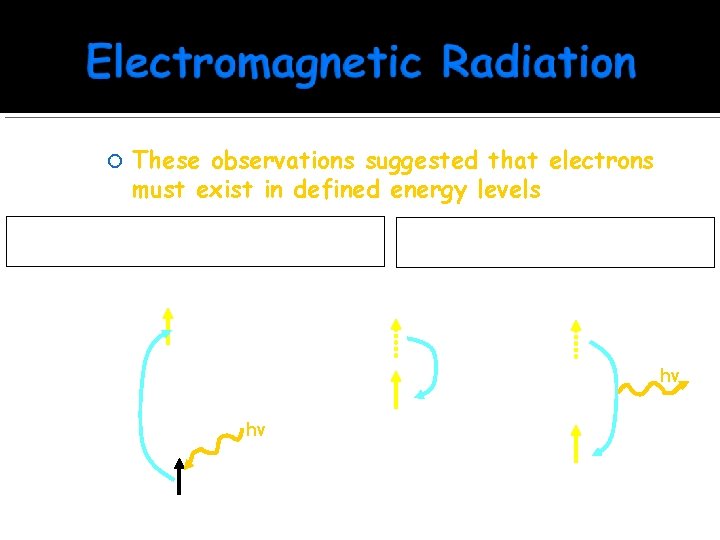
Atomic Line Spectra Electrons in an atom add energy to go to an “excited state”. When they relax back to the ground state, they emit energy in specific energy quanta

These observations suggested that electrons must exist in defined energy levels First, the electron absorbs energy and jumps from the ground state to an excited state 5 ______ 4 ______ 3 ______ 2 ______ 1 ______ hv Next, the excited electron relaxes to a lower excited state or ground state 5 ______ 4 ______ 3 ______ 2 ______ 1 ______ hv

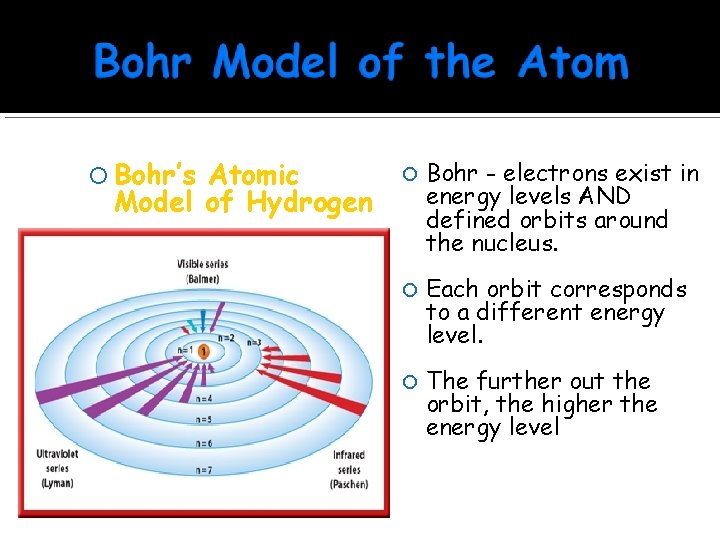
Bohr’s Atomic Model of Hydrogen Bohr - electrons exist in energy levels AND defined orbits around the nucleus. Each orbit corresponds to a different energy level. The further out the orbit, the higher the energy level

Transitions of the visible spectrum There are three main series of transition Paschen Series- IR ▪ Drops into 3 rd Level Balmer Series Visible ▪ Drops into 2 nd Level Lyman Series - UV ▪ Drops into 1 st Level

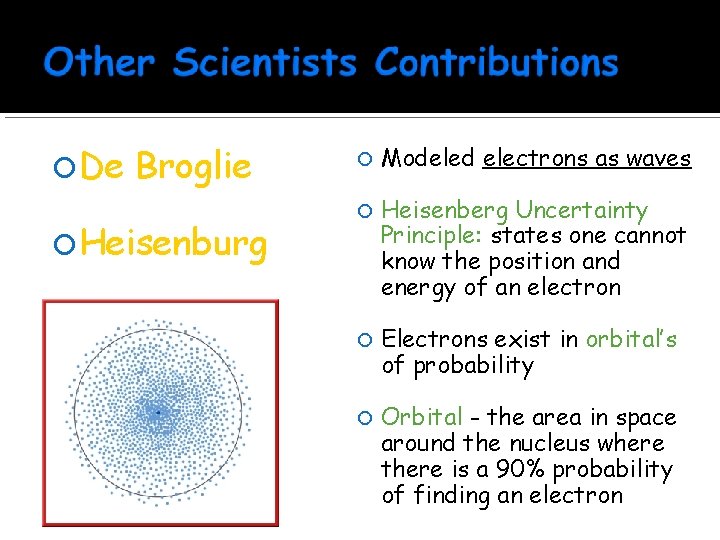
De Broglie Heisenburg Modeled electrons as waves Heisenberg Uncertainty Principle: states one cannot know the position and energy of an electron Electrons exist in orbital’s of probability Orbital - the area in space around the nucleus where there is a 90% probability of finding an electron

Schrödinger Wave Equation - mathematical solution of an electron’s energy in an atom quantum mechanical model of the atom – current model of the atom treating electrons as waves.

Quantum Numbers Wave Equation generates 4 variable solutions n - size l - shape m - orientation s – spin Address of an electron

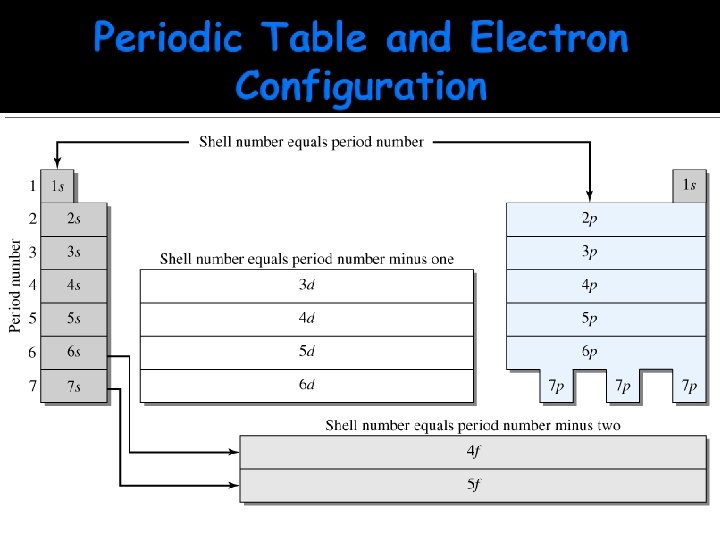
n – Primary Quantum Number Describes the size and energy of the orbital n is any positive # n = 1, 2, 3, 4, …. Found table Like in on the periodic the “state” you live

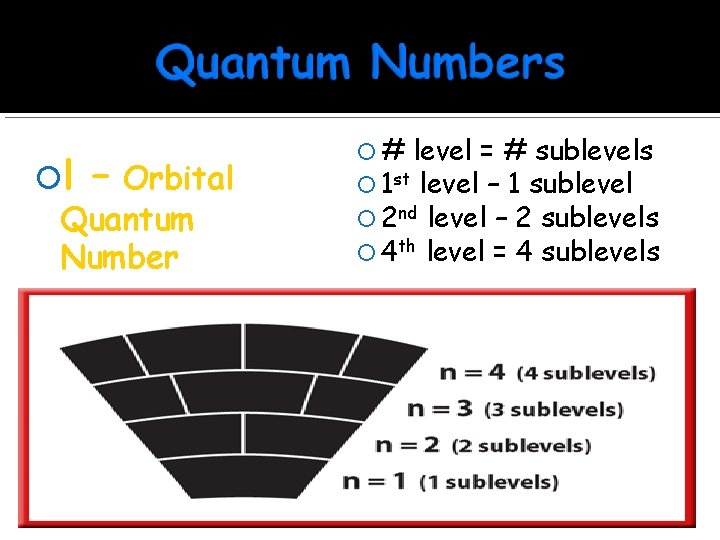
l – Orbital Quantum Number n=3 l = 0, 1, 2 n=2 Sub-level of energy Describes the shape of the orbital l = 0, 1, 2, 3, 4, …. (n-1) l = 0, 1 n=1 l=0 “City” you live in

l – Orbital Quantum Number # level = # sublevels 1 st level – 1 sublevel 2 nd level – 2 sublevels 4 th level = 4 sublevels

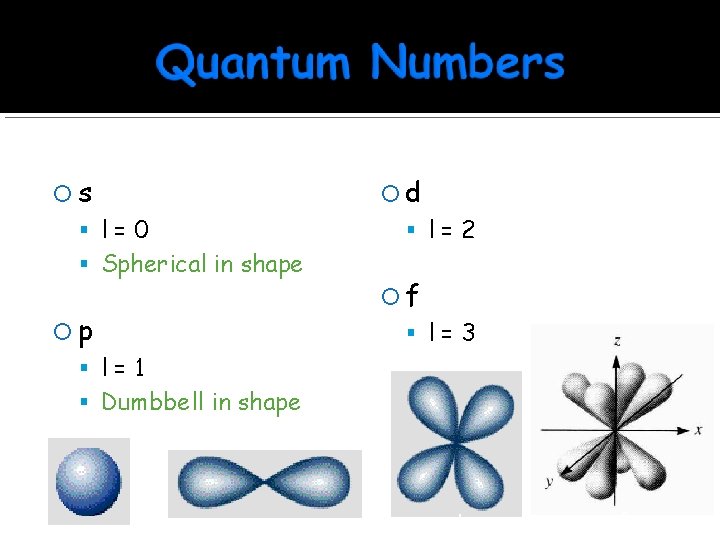
s Sublevels are named for their shape d l=0 l=2 Spherical in shape p f l=3 l=1 Dumbbell in shape s p d f

m – Magnetic Quantum Number Describes the orientation of the orbital in space Also denotes how many orbital's are in each sublevel For each sublevel there are 2 l +1 orbital's m = 0, ± 1, ± 2, ± 3, ±l “Street” you live on

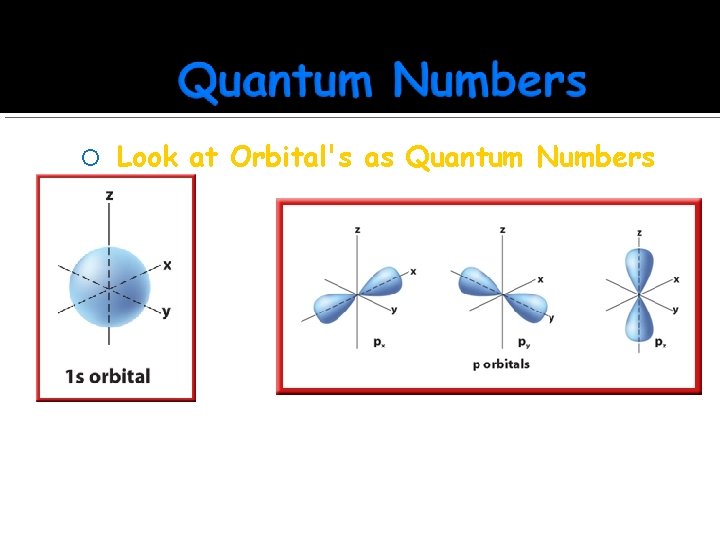
Look at Orbital's as Quantum Numbers l=0 m=0 Can only be one s orbital l = 1 m = -1, 0, +1 For each p sublevel there are 3 possible orientations, so three 3 orbital's

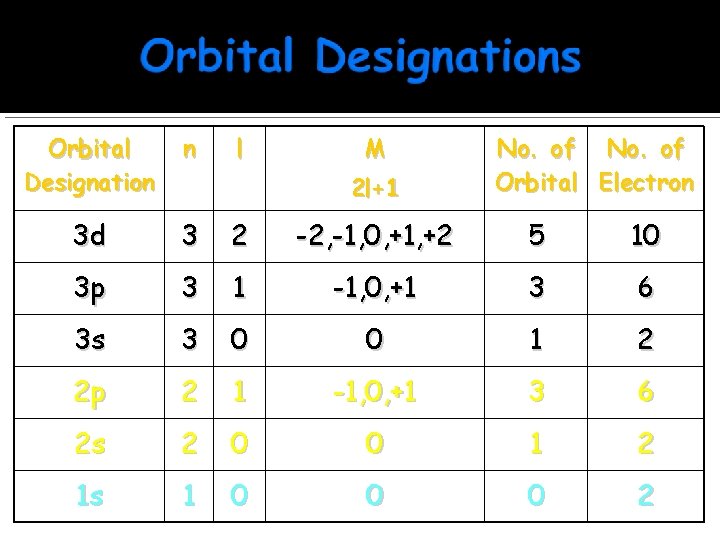
Orbital Designation n l M 3 d 3 2 -2, -1, 0, +1, +2 5 10 3 p 3 1 -1, 0, +1 3 6 3 s 3 0 0 1 2 2 p 2 1 -1, 0, +1 3 6 2 s 2 0 0 1 2 1 s 1 0 0 0 2 2 l+1 No. of Orbital Electron

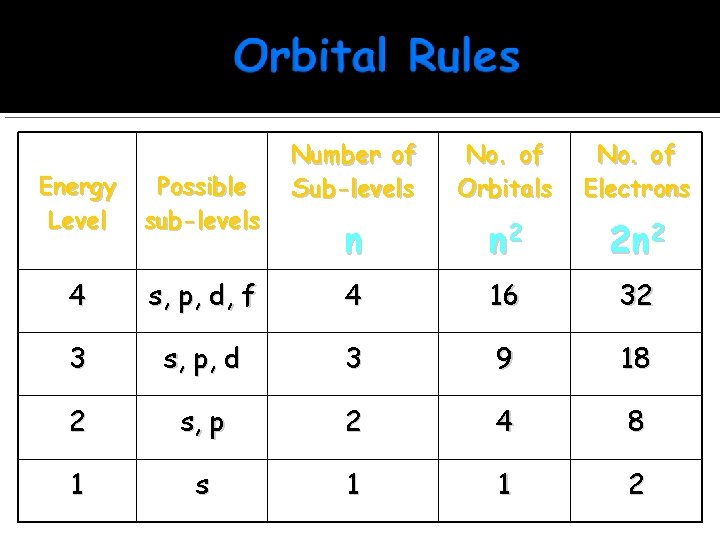
Number of Sub-levels No. of Orbitals No. of Electrons n n 2 2 n 2 Energy Level Possible sub-levels 4 s, p, d, f 4 16 32 3 s, p, d 3 9 18 2 s, p 2 4 8 1 s 1 1 2

How is the Bohr model different from the model of Dalton? Who contributed to the modern model of the atom? How is it different from Bohr’s? Why do atoms give unique atomic line spectra? What Is are ground and excited states? 2 d possible? 4 f ? 2 s ? 6 p? 1 p? How many total orbital's in the 2 nd level? 4 th level.

Quantum…. What…. . ?

Aufbau Principal Lowest energy orbital available fills first “Lazy Rule” Tenant

Pauli Exclusion Principle No two electrons have the same quantum #’s Maximum electrons in any orbital is two ( )

Hund’s Rule RIGHT When filling degenerate orbital's, electrons will fill an empty orbital before pairing up with another electron. Empty room rule WRONG


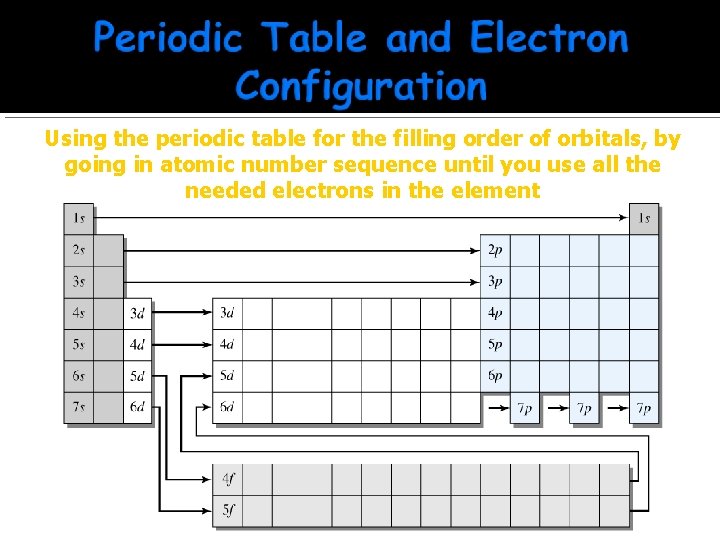
Using the periodic table for the filling order of orbitals, by going in atomic number sequence until you use all the needed electrons in the element

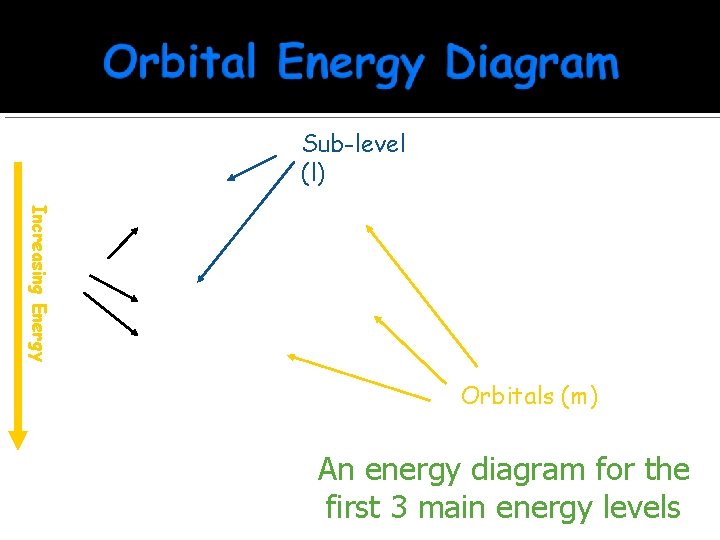
Increasing Energy Level (n) Sub-level (l) 1 s ______ 2 p ______ 3 s ______ 3 p ______ Orbitals (m) An energy diagram for the first 3 main energy levels

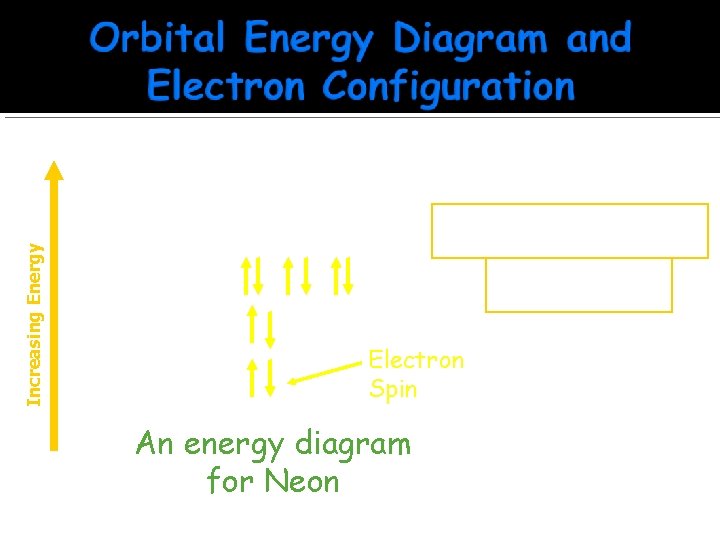
Increasing Energy p 3 s p ______ 1 s 2 2 px 2 2 py 2 2 pz 2 ______ 2 s ______ 1 s ______ Electron Spin An energy diagram for Neon 1 s 2 2 p 6 Electron Configuration Notation

Orbital Notation O (atomic number 8) shows each orbital ____ ____ 1 s 2 s 2 px 2 py 2 pz 3 s 1 s 22 p 4 electron configuration!

Write the orbital S (atomic number 16) notation for S ____ ____ 1 s 2 s 1 s 22 p 63 s 23 p 4 2 p 3 s ____ 3 p How many unpaired electrons does sulfur have? 2 unpaired electrons!

Bellringer What As ? is the electron configuration for A. Use the long form B. Use the noble gas notation Log onto SFHS website – navigate to Staff / Curtis and select the Gizmo link

Valence As Electrons The electrons in the outermost energy level. s and p electrons in last shell (atomic number 33) 5 valence electrons 1 s 22 p 63 s 23 p 64 s 23 d 104 p 3

Longhand Configuration S 16 e- 1 s 2 2 p 6 3 s 2 3 p 4 Core Electrons Valence Electrons Shorthand Configuration S 16 e [Ne] 2 3 s 4 3 p
![Example X X X Germanium X X X X X Ar4 s Example X X X - Germanium X X X X X [Ar]4 s](https://slidetodoc.com/presentation_image_h2/2164d3aa788131e9c14d8d8b62c3f479/image-39.jpg)
Example X X X - Germanium X X X X X [Ar]4 s 2 3 d 10 4 p 2

Let’s Practice P (atomic number 15) Noble Gas Configuration Ca (atomic number 20) 1 s 22 p 63 s 23 p 64 s 2 [Ar] 4 s 2 1 s 22 p 63 s 23 p 3 As (atomic number 33) 1 s 22 p 63 s 23 p 64 s 23 d 104 p 3 [Ne] 3 s 23 p 3 [Ar] 4 s 23 d 104 p 3 W (atomic number 74) 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 66 s 24 f 145 d 4 [Xe] 6 s 24 f 145 d 4

Your Turn N (atomic number 7) 1 s 22 p 3 Na (atomic number 11) 1 s 22 p 63 s 1 Noble Gas Configuration [He] 2 s 22 p 3 [Ne] 3 s 1 Sb (atomic number 51) 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 3 Cr (atomic number 24) 1 s 22 p 63 s 23 p 64 s 23 d 4 [Kr]5 s 24 d 105 p 3 [Ar] 4 s 23 d 4

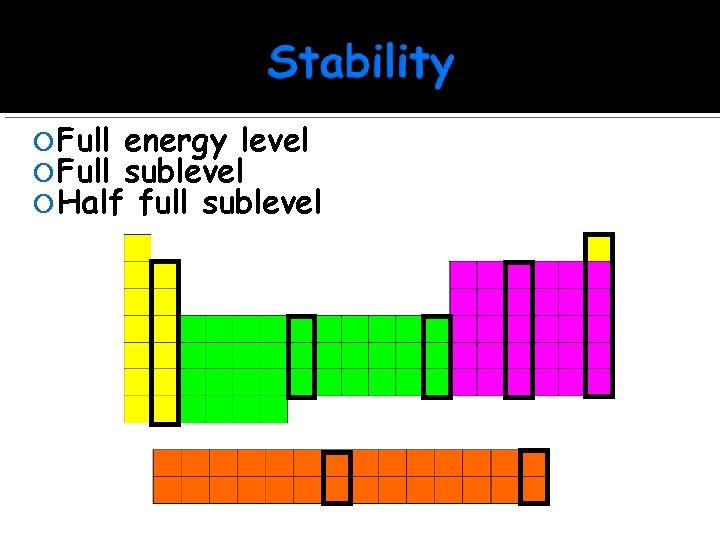
Full energy level Full sublevel Half full sublevel
![Copper Expect Ar 4 s 2 3 d 9 Actual Ar 4 s Copper Expect: [Ar] 4 s 2 3 d 9 Actual: [Ar] 4 s](https://slidetodoc.com/presentation_image_h2/2164d3aa788131e9c14d8d8b62c3f479/image-43.jpg)
Copper Expect: [Ar] 4 s 2 3 d 9 Actual: [Ar] 4 s 1 3 d 10 Silver Expect: [Kr] 5 s 2 4 d 9 Actual: [Kr] 5 s 1 4 d 10 Chromium Molybdenum Expect: [Ar] 4 s 2 3 d 4 Actual: [Ar] 4 s 1 3 d 5 Expect: [Kr] 5 s 2 4 d 4 Actual: [Kr] 5 s 1 4 d 5 Exceptions are explained, but not predicted! Atoms are more stable with half full sublevel

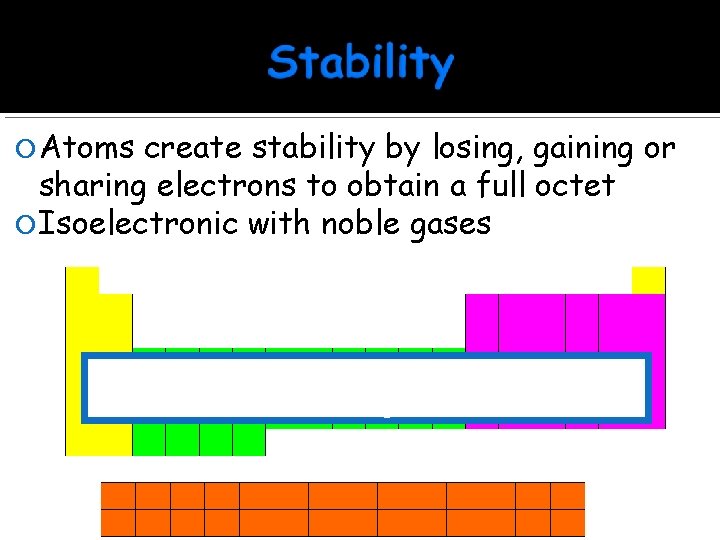
Atoms create stability by losing, gaining or sharing electrons to obtain a full octet Isoelectronic with noble gases +1 0 +2 +3 +4 -3 -2 -1 Atoms take electron configuration of the closest noble gas

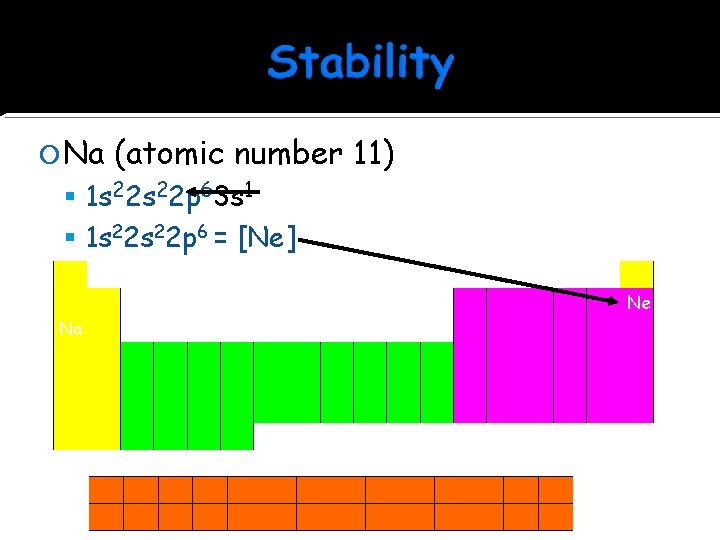
Na (atomic number 11) 1 s 22 p 63 s 1 1 s 22 p 6 = [Ne] 1 Valence electron Metal = Loses Ne Na

P-3 (atomic number 15) 1 s 22 p 63 s 23 p 6 Ca+2 (atomic number 20) 1 s 22 p 63 s 23 p 6 Full Octet Zn+2 (atomic number 30) 1 s 22 p 63 s 23 p 63 d 10 Last valence electrons (s and p)

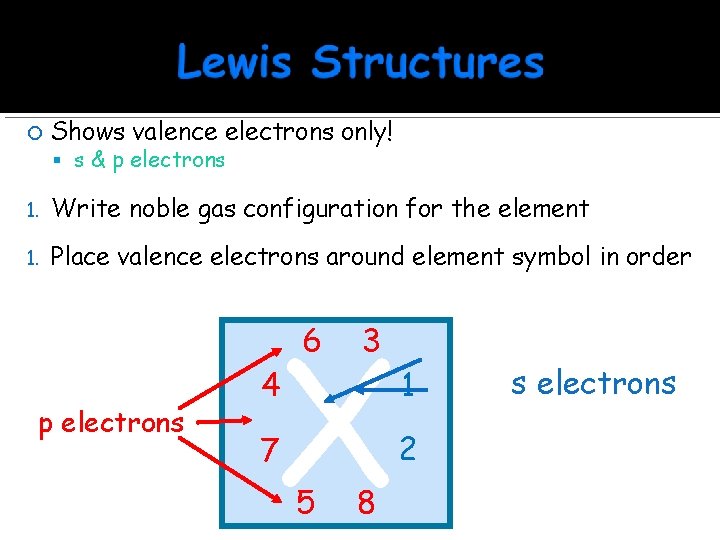
Shows valence electrons only! 1. Write noble gas configuration for the element 1. Place valence electrons around element symbol in order s & p electrons X 4 6 3 7 5 8 1 2 s electrons
![Write the Lewis Oxygen O He Iron structures for 2 s 2 2 Write the Lewis Oxygen (O) [He] Iron structures for: 2 s 2 2](https://slidetodoc.com/presentation_image_h2/2164d3aa788131e9c14d8d8b62c3f479/image-48.jpg)
Write the Lewis Oxygen (O) [He] Iron structures for: 2 s 2 2 p 4 (Fe) [Ar] 4 s 2 3 d 6 Bromine (Br) [Ar] 4 s 2 3 d 10 4 p 5 • • O • • Valence electrons • • Fe • • Br • • •