ELECTRON Orbital Notation Noble Gas Notation and Valence

ELECTRON Orbital Notation, Noble Gas Notation, and Valence Electrons DIAGRAMS

Noble Gases � Method of representing electron configurations of noble gases �Elements in the last column of the periodic table �Have 8 electrons in their outermost orbital �Extremely stable

Nobel Gas Notation � Recall, the electron configuration for Na is: Na: 1 s 2 2 p 6 3 s 1 � We can abbreviate the electron configuration by indicating the innermost electrons with the symbol of the preceding noble gas. � The preceding noble gas with an atomic number less than sodium is neon, Ne. We rewrite the electron configuration: Na: [Ne] 3 s 1

Orbital Notation In electron configurations, we wrote the orbitals together as one sublevel � Example: � � 2 px 2 py 2 pz all became the 2 p sublevel � We do not get to see how the individual electrons line up � In orbital notations, each sublevel is written to show the electron’s spins in each direction � Orbital notation shows you where each specific electron is placed (in order) and what it’s “spin” is.

Reference Sheet � This reference sheet shows each orbital split apart into its 3 dimensional axes

Electron Configuration for Na � Na, 11 e� 1 s 2 2 p 6 3 s 1 � We are now going to represent each electron as a “half arrow” � We must fill in the electrons as we did previously but show them using arrows on the orbital notation diagram � The first electron in an orbital is placed up (+1/2 spin)
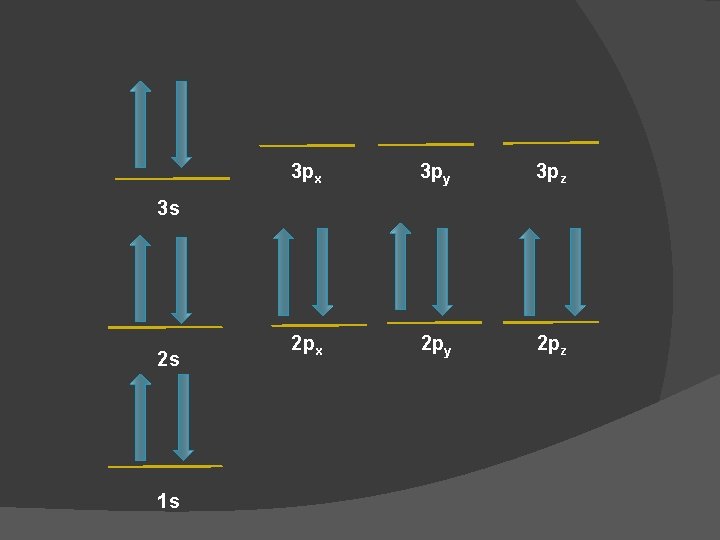
3 px 3 py 3 pz 2 px 2 py 2 pz 3 s 2 s 1 s
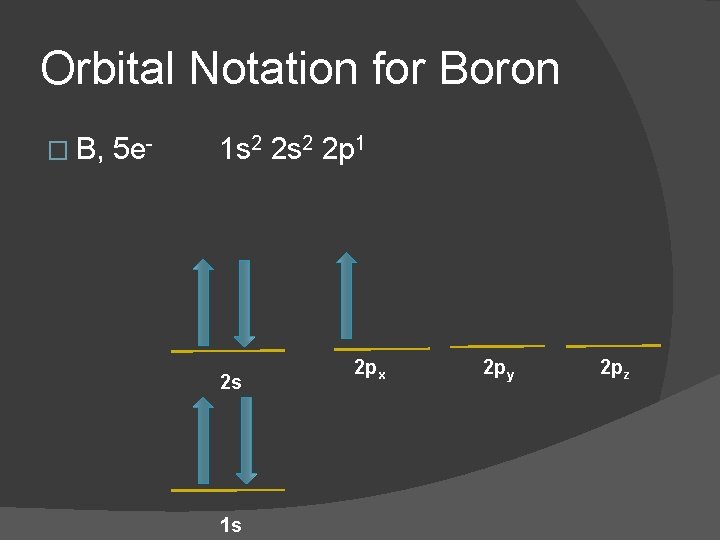
Orbital Notation for Boron � B, 5 e- 1 s 2 2 p 1 2 s 1 s 2 px 2 py 2 pz

Orbital Notation Practice � Helium � Boron � Carbon � Oxygen � Sodium � Manganese
![Exceptions to Predicted Configurations � Chromium �Incorrect: [Ar] 4 s 23 d 4 �Correct: Exceptions to Predicted Configurations � Chromium �Incorrect: [Ar] 4 s 23 d 4 �Correct:](http://slidetodoc.com/presentation_image/3a4f5499081c4685bd45cd54e6d1e9d9/image-10.jpg)
Exceptions to Predicted Configurations � Chromium �Incorrect: [Ar] 4 s 23 d 4 �Correct: [Ar] 4 s 13 d 5 � Copper: �Incorrect: [Ar] 4 s 23 d 9 �Correct: [Ar] 4 s 13 d 10 � Elements in group 6 (s 1 d 5) and group 11 (s 1 d 10) have greater stability with halffilled and filled sets of s and d orbitals

Valence Electrons � Only certain electrons determine the chemical properties of an element � Valence Electrons: � Electrons in the atom’s outermost orbitals – generally those orbitals associated with the atom’s highest principal energy level orbitals (s and p) � Example: � Sulfur contains 16 electrons � 6 electrons occupy the outermost 3 s and 3 p orbitals � Therefore, Sulfur has 6 valence electrons

Electron Dot Structures � Chemist often represent valence electrons in a short hand method �Termed Electron-Dot Structures � Structures consist of the element’s symbol surrounded by dots, representing the valence electrons
- Slides: 12