GENERAL CHEMISTRY CHEM 110 REVISION CHAPTER 7 Energy
![[1] Calculate the energy, in joules, required to excite a hydrogen atom by causing [1] Calculate the energy, in joules, required to excite a hydrogen atom by causing](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-6.jpg)
![[3] What is the four quantum numbers for the last electron in Aluminum(Al 13) [3] What is the four quantum numbers for the last electron in Aluminum(Al 13)](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-7.jpg)
![[6] Which one of the following electrons have the same energy as an electron [6] Which one of the following electrons have the same energy as an electron](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-8.jpg)
![[9] Which statement about the four quantum numbers which describe electrons in atoms is [9] Which statement about the four quantum numbers which describe electrons in atoms is](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-9.jpg)
![[12] A possible set of quantum numbers for the last electron added to complete [12] A possible set of quantum numbers for the last electron added to complete](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-10.jpg)
![[15] Which one of the following sets of quantum number is not possible? A. [15] Which one of the following sets of quantum number is not possible? A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-11.jpg)
![[18] Choose the incorrect quantum numbers for electron in an atom? A. (3 [18] Choose the incorrect quantum numbers for electron in an atom? A. (3](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-12.jpg)
![[21] The maximum number of electron for one orbital is? A. 2 electron B. [21] The maximum number of electron for one orbital is? A. 2 electron B.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-13.jpg)
![[24] Atoms of which two elements have the same number of unpaired electrons? A. [24] Atoms of which two elements have the same number of unpaired electrons? A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-14.jpg)
![[27]The ground-state electron configuration for an atom of indium (In) is A. B. C. [27]The ground-state electron configuration for an atom of indium (In) is A. B. C.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-15.jpg)
![[30] Atoms of which two elements have the same number of unpaired electrons? A. [30] Atoms of which two elements have the same number of unpaired electrons? A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-16.jpg)
![[33] Which of the following is paramagnetic? (A) K+ (B) Sn+2 (C) Sc+3 (D) [33] Which of the following is paramagnetic? (A) K+ (B) Sn+2 (C) Sc+3 (D)](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-17.jpg)
![[36] A ground –state atom of Mn+2 has -----unpaired electrons and is -----. A. [36] A ground –state atom of Mn+2 has -----unpaired electrons and is -----. A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-18.jpg)
![[38] Which two electron configurations represent elements that would have similar chemical properties? 1. [38] Which two electron configurations represent elements that would have similar chemical properties? 1.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-19.jpg)
![[40] Which of the orbital diagrams is not follow the pauli exclusion principle? ↑ [40] Which of the orbital diagrams is not follow the pauli exclusion principle? ↑](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-20.jpg)
![[42]Choose the incorrect quantum numbers for electron in an atom? A. (3 , [42]Choose the incorrect quantum numbers for electron in an atom? A. (3 ,](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-21.jpg)
![[45] The electron configuration of a neutral atom is[Ne] 3 s 23 p 3. [45] The electron configuration of a neutral atom is[Ne] 3 s 23 p 3.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-22.jpg)



- Slides: 25

GENERAL CHEMISTRY CHEM 110 REVISION CHAPTER 7

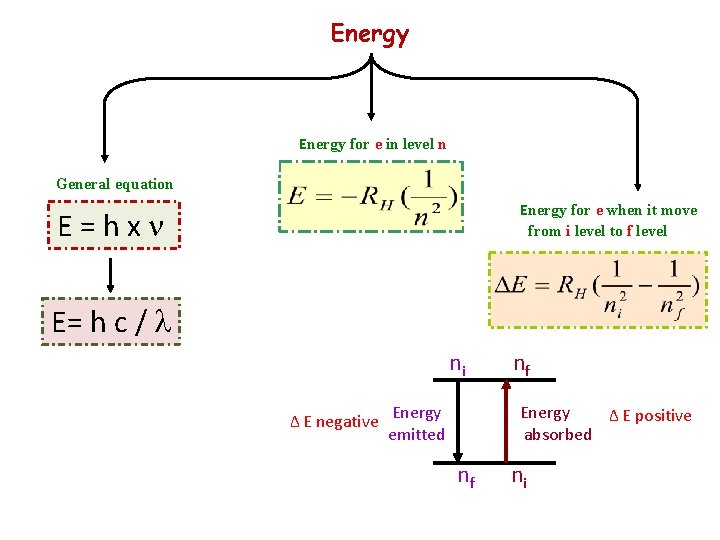
Energy for e in level n General equation Energy for e when it move from i level to f level E = h x n E= h c / l ni ∆ E negative Energy emitted nf Energy ∆ E positive absorbed nf ni

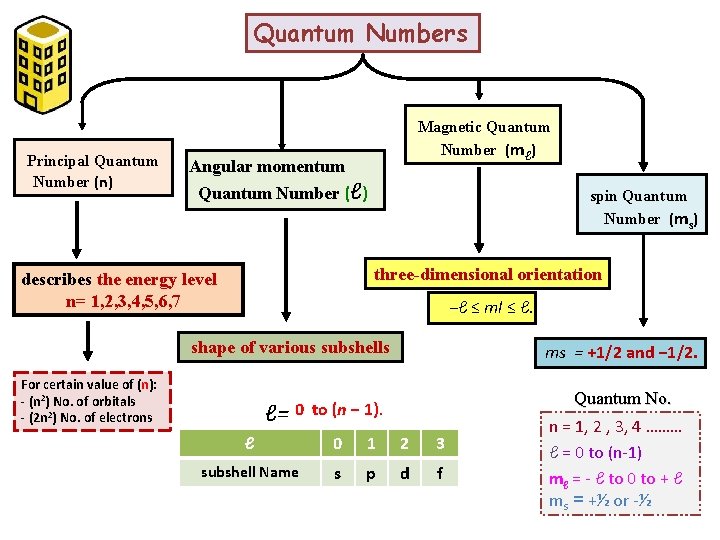
Quantum Numbers Principal Quantum Number (n) Magnetic Quantum Number (mℓ) Angular momentum Quantum Number (ℓ) spin Quantum Number (ms) three-dimensional orientation describes the energy level n= 1, 2, 3, 4, 5, 6, 7 −ℓ ≤ ml ≤ ℓ. shape of various subshells For certain value of (n): - (n 2) No. of orbitals - (2 n 2) No. of electrons ℓ= ms = +1/2 and − 1/2. Quantum No. 0 to (n − 1). ℓ 0 1 2 3 subshell Name s p d f n = 1, 2 , 3, 4 ……… ℓ = 0 to (n-1) mℓ = - ℓ to 0 to + ℓ ms = +½ or -½

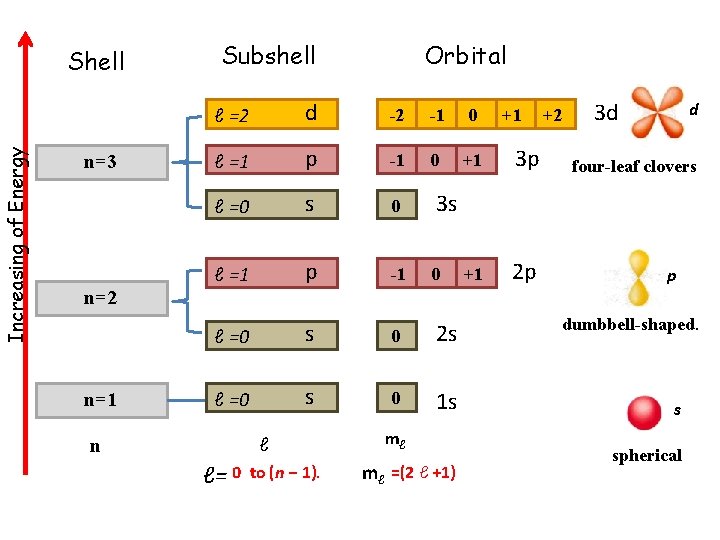
Increasing of Energy Shell n=3 Subshell Orbital ℓ =2 d -2 -1 0 ℓ =1 p -1 0 +1 3 p ℓ =0 s 0 3 s ℓ =1 p -1 0 +1 2 p ℓ =0 s 0 2 s ℓ =0 s 0 1 s +1 +2 3 d d four-leaf clovers p n=2 n=1 ℓ n ℓ= 0 to (n − 1). mℓ mℓ =(2 ℓ +1) dumbbell-shaped. s spherical

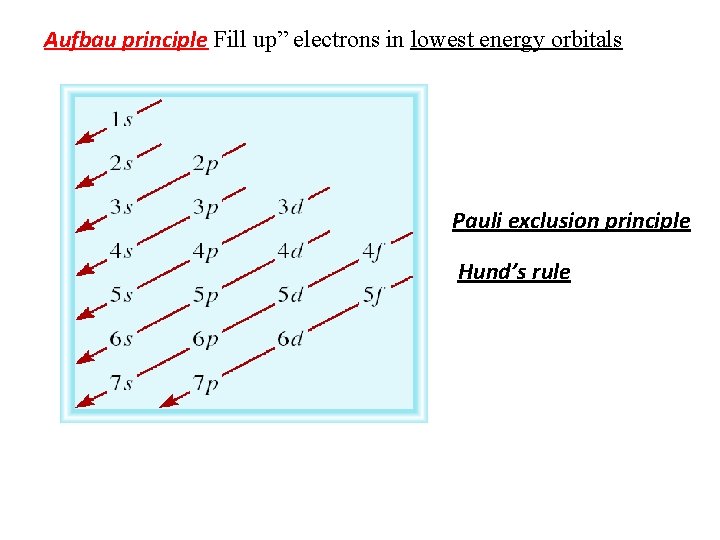
Aufbau principle Fill up” electrons in lowest energy orbitals Pauli exclusion principle Hund’s rule
![1 Calculate the energy in joules required to excite a hydrogen atom by causing [1] Calculate the energy, in joules, required to excite a hydrogen atom by causing](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-6.jpg)
[1] Calculate the energy, in joules, required to excite a hydrogen atom by causing an electronic transition from the n = 1 to the n = 4 principal energy level. A. 2. 04 10 - 18 J B. 2. 19 105 J C. 2. 04 10 -29 J D. 3. 27 10 -17 J [2] What is the wavelength of radiation that has a frequency of 7. 5× 1014 s-1? A. 2. 3× 106 nm B. 4. 3× 10 -7 nm C. 2. 0× 1023 nm D. 4. 0× 102 nm
![3 What is the four quantum numbers for the last electron in AluminumAl 13 [3] What is the four quantum numbers for the last electron in Aluminum(Al 13)](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-7.jpg)
[3] What is the four quantum numbers for the last electron in Aluminum(Al 13) atom A. n=2, ℓ= 1, mℓ=-1, ms=+1/2 B. n=3, ℓ=3, mℓ=-1, ms=+1/2 C. n=3, ℓ=1, mℓ=-1, ms=+1/2 D. n=2, ℓ= 1, mℓ=-1, ms=-1/2 [4] Which one of the following electrons has the same energy as an electron with quantum numbers n = 5, l = 2, ml = -1 , ms = +1/2? A. n = 5, l = 2, ml = 0, ms = +1/2 B. n = 4, l = 3, ml = -1, ms = +1/2 C. n = 4, l = 2, ml = 3, ms = -1/2 D. None of these [5] What is the four quantum numbers for the last electron in Sodium (Na 11) atom A. B. C. D. n = 3, ℓ = 1, mℓ = 0, ms = -½ n = 3, ℓ = 0, ms =+ ½ n = 2, ℓ = 1, mℓ = -1, ms = +½ n = 2, ℓ = 0, mℓ = -1, ms =+ ½
![6 Which one of the following electrons have the same energy as an electron [6] Which one of the following electrons have the same energy as an electron](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-8.jpg)
[6] Which one of the following electrons have the same energy as an electron with quantum numbers n = 4, l = 3, ml = -1 , ms = +1/2? A. n = 5, l = 1, ml = -1 , ms = ½ B. n = 4, l = 3, ml = -1 , ms = ½ C. n = 4, l = 2, ml = 2 , ms = -½ D. None of these [7 ]Which one of the following electrons have the same energy as an electron with quantum numbers n = 4, l = 2, ml = -1 , ms = +1/2? A. n=5, l = 1, ml = -1, ms= +1/2 B. n=4, l = 3, ml = -1, ms= +1/2 C. n=4, l = 2, ms= -1/2 D. None of these [8] What is the four quantum numbers for the last electron in Silicon (Si) atom A. B. C. D. n= 3, l = 2, ml = 0, ms = - ½ n= 3, l = 1, ml = 0, ms = +½ n= 2, l = 1, ml = -1, ms = -½ n= 2, l = 0, ml = -1, ms = +½
![9 Which statement about the four quantum numbers which describe electrons in atoms is [9] Which statement about the four quantum numbers which describe electrons in atoms is](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-9.jpg)
[9] Which statement about the four quantum numbers which describe electrons in atoms is incorrect? A. n = principal quantum number, n = 1, 2, 3, . . . B. l = subsidiary quantum number, l = 1, 2, 3, . . . , (n+1) C. ml = magnetic quantum number, ml = (-l), . . , 0, . . , (+l) D. ms = spin quantum number, ms = +1/2 or -1/2. [10] What is the total number of orbitals in the n = 3 level? (A) 2 (B) 8 (C) 9 (D) 3 [11] Which of the following statements about the quantum numbers is incorrect? (A) n has integral values from 1 to ∞ (B) L has values from 1 to ∞ (C) m. L has values of -L to +L, including zero (D) ms has values of + ,
![12 A possible set of quantum numbers for the last electron added to complete [12] A possible set of quantum numbers for the last electron added to complete](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-10.jpg)
[12] A possible set of quantum numbers for the last electron added to complete an atom of gallium Ga in its ground state is n L ms (A) 4 0 -1/2 (B) 3 1 0 +1/2 (C) 4 1 +1/2 (D) 3 1 +1 -1/2 [13]The number of orbitals in a d subshell is? A. 1 B. 3 C. 5 D. 7 [14] The maximum number of electron for one orbital is? A. 2 electron B. 1 electron C. 3 electron D. 6 electron
![15 Which one of the following sets of quantum number is not possible A [15] Which one of the following sets of quantum number is not possible? A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-11.jpg)
[15] Which one of the following sets of quantum number is not possible? A. (3 , 0 , +1/2 ) B. (4 , 3 , -2 , -1/2 ) C. (1 , 0 , +1/2 ) D. (3 , 2 , 1 ) [16]The last two electrons in C 6 atom should differ in the value of the quantum number: A. n B. l C. ml D. ms [17] The representative elements are those with unfilled energy levels in which the "last electron" was added to A. an s or p orbital. . B. an s orbital. C. a p or d orbital. D. an f orbital.
![18 Choose the incorrect quantum numbers for electron in an atom A 3 [18] Choose the incorrect quantum numbers for electron in an atom? A. (3](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-12.jpg)
[18] Choose the incorrect quantum numbers for electron in an atom? A. (3 , 0 , +1/2) B. (3 , 5 , 0 , +1/2) C. (1 , 0 , +1/2) D. (4 , 3 , -2 , -1/2) [19 The number of orbital in a p subshell is? A. 1 B. 7 C. 5 D. 3 [20] The maximum number of electrons that can occupy an energy level described by the principal quantum number, n, is? A. n+1 B. 2 n C. n 2 D. 2 n 2
![21 The maximum number of electron for one orbital is A 2 electron B [21] The maximum number of electron for one orbital is? A. 2 electron B.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-13.jpg)
[21] The maximum number of electron for one orbital is? A. 2 electron B. 3 electron C. 1 electron D. 6 electron [22] The electron configuration of a neutral atom is[Ne] 3 s 23 p 3. The four quantum numbers of the last electron are: A. (3 , 1 , +1/2 ) B. (2 , 0 , +1/2 ) C. (2 , 0 , -1 , +1/2 ) D. (3 , 2 , -1 , +1/2 ) [23] Atoms of which two elements have the same number of unpaired electrons? A. Na and C B. O and C C. Mg and C D. Al and C
![24 Atoms of which two elements have the same number of unpaired electrons A [24] Atoms of which two elements have the same number of unpaired electrons? A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-14.jpg)
[24] Atoms of which two elements have the same number of unpaired electrons? A. Si and Cl B. Al and Cl C. Mg and Cl D. N and Cl [25] Which anion contains the most number of electrons? A. P 3 B. Se 2 C. Si 4 D. S 2[26] Atoms of which two elements have the same number of unpaired electrons? A. Si and F B. B and F C. Ca and F D. N and F
![27The groundstate electron configuration for an atom of indium In is A B C [27]The ground-state electron configuration for an atom of indium (In) is A. B. C.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-15.jpg)
[27]The ground-state electron configuration for an atom of indium (In) is A. B. C. D. [Ar]4 s 23 d 104 p 1. [Ar]4 s 24 p 63 d 5. [Kr]5 s 25 p 64 d 5. [Kr]5 s 24 d 105 p 1. [28] The [Ar]4 s 24 p 5 element are found in _______ of the periodic table. A. B. C. D. Group 2 A Group 7 A Period 7 Group 8 A [29] How many electrons are found in the (Sn 2+) ion? A. 48 B. 119 C. 22 D. 69
![30 Atoms of which two elements have the same number of unpaired electrons A [30] Atoms of which two elements have the same number of unpaired electrons? A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-16.jpg)
[30] Atoms of which two elements have the same number of unpaired electrons? A. Na and S B. Si and S C. Mg and S D. Al and S [31] Which ground-state atom has an electron configuration described by the following orbital diagram? (A) phosphorus (B) germanium (C) selenium (D) tellurium [32] All the following electronic ground-state configurations are correct except (A) 20 Ca: [Ar] 4 s 2 (B) 25 Mn: [Ar] 4 s 2 4 d 5 (C) 29 Cu: [Ar] 4 s 13 d 10 (D) 54 Xe: [Kr] 5 s 24 d 10 5 p 6
![33 Which of the following is paramagnetic A K B Sn2 C Sc3 D [33] Which of the following is paramagnetic? (A) K+ (B) Sn+2 (C) Sc+3 (D)](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-17.jpg)
[33] Which of the following is paramagnetic? (A) K+ (B) Sn+2 (C) Sc+3 (D) Cr+3 [34] The general electron configuration for atoms of all elements in Group 5 A is (A) ns 2 np 1 (B) ns 2 np 5 (C) ns 2 np 4 (D) ns 2 np 3 [35] What is the electron configuration of the Cr 24? A. ( 1 s 2 2 p 6 3 s 2 3 p 5 4 s 2 3 d 5 ) B. ( 1 s 2 2 p 6 3 s 3 3 p 6 4 s 2 3 d 3 ) C. ( 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 5 ) D. ( 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 4 )
![36 A ground state atom of Mn2 has unpaired electrons and is A [36] A ground –state atom of Mn+2 has -----unpaired electrons and is -----. A.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-18.jpg)
[36] A ground –state atom of Mn+2 has -----unpaired electrons and is -----. A. 2, paramagnetic B. 4, diamagnetic C. 0, diamagnetic D. 5, paramagnetic [37] The orbital diagram for a ground state O 2 - atom is: A. ↓↑ ↓↑ ↓ ↑ ↑ 1 s 2 s 2 p B. ↓↑ ↓↑ ↑ ↑ 1 s 2 s 2 p C. ↓↑ ↓↑ ↑ 1 s 2 s 2 p D. ↓↑ ↓↑ ↓↑ 1 s 2 s 2 p
![38 Which two electron configurations represent elements that would have similar chemical properties 1 [38] Which two electron configurations represent elements that would have similar chemical properties? 1.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-19.jpg)
[38] Which two electron configurations represent elements that would have similar chemical properties? 1. 1 s 22 p 4 2. 1 s 22 p 5 3. [Ar]4 s 23 d 104 p 3 4. [Ar]4 s 23 d 104 p 4 A. (2) and (4) B. (1) and (2) C. (2) and (3) D. (1) and (4) [39] What is the electron configuration of the Mo 42? A. ( 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 104 p 65 s 14 d 9 ) B. ( 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 104 p 6 5 s 1 4 d 5) C. ( 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 104 p 64 d 9 5 s 2) D. ( 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 104 p 6 5 s 2 4 d 4
![40 Which of the orbital diagrams is not follow the pauli exclusion principle [40] Which of the orbital diagrams is not follow the pauli exclusion principle? ↑](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-20.jpg)
[40] Which of the orbital diagrams is not follow the pauli exclusion principle? ↑ ↑ ↑↑ ↑ ↑ ↓ ↑↓ ↑ ↓↓ ↑↓ ↑↓ ↑ ↑ ↑↓ A. (1) and(6) B. (2) and(5) C. (1) , (2)and(6) D. (3) , (4)and(5) [41] Which of the orbital diagrams is not follow Hand's rule? ↑ ↑ ↑↑ ↑ ↓ ↓ ↑↓ A. (1) and(4) B. (1) and(5) C. (2) and(3) D. (2) , (4)and(6) ↑↓ ↑↓ ↑ ↓↓ ↑↓ ↑ ↑
![42Choose the incorrect quantum numbers for electron in an atom A 3 [42]Choose the incorrect quantum numbers for electron in an atom? A. (3 ,](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-21.jpg)
[42]Choose the incorrect quantum numbers for electron in an atom? A. (3 , 0 , +1/2) B. (3 , 5 , 0 , +1/2) C. (1 , 0 , +1/2) D. (4 , 3 , -2 , -1/2) [43] The number of orbital in a p subshell is? A. 1 B. 7 C. 5 D. 3 [44] The maximum number of electrons that can occupy an energy level described by the principal quantum number , n, is? A. n+1 B. 2 n C. n 2 D. 2 n 2
![45 The electron configuration of a neutral atom isNe 3 s 23 p 3 [45] The electron configuration of a neutral atom is[Ne] 3 s 23 p 3.](https://slidetodoc.com/presentation_image_h/82ed1d9f49899ec3f58648da61e27878/image-22.jpg)
[45] The electron configuration of a neutral atom is[Ne] 3 s 23 p 3. The four quantum numbers of the last electron are: A. (3 , 1 , +1/2 ) B. (2 , 0 , +1/2 ) C. (2 , 0 , -1 , +1/2 ) D. (3 , 2 , -1 , +1/2 ) 46 What is the energy (in joules) a photon must have in order to excite an electron from E 2 to E 3? A) 5 × 10– 10 J B) 10 × 10– 19 J C) 5 × 10– 19 J D) 15 × 10– 19 J 47. The atomic number of an element is 73. Is this element diamagnetic or paramagnetic? (A) Diamagnetic (B) Paramagnetic

48. Give the values of the quantum numbers associated with the 2 p subshell. A) n = 2, l = 2, ml = – 2, – 1, 0, 1, 2 B) n = 2, l = 1, ml = 0 C) n = 2, l = 1, ml = 1 D) n = 2, l = 1, ml = – 1, 0, 1 49. State the total number of: p electrons in N (Z = 7); s electrons in Si (Z = 14); and 3 d electrons in S (Z = 16). A) N, 3 p electrons; Si, 6 s electrons; S, 5 d electrons B) N, 2 p electrons; Si, 6 s electrons; S, 5 d electrons C) N, 3 p electrons; Si, 6 s electrons; S, 0 d electrons D) N, 6 p electrons; Si, 6 s electrons; S, 0 d electrons 50. The electron configuration of a neutral atom is 1 s 22 p 63 s 2. Name the element. A) Si B) Na C) Mg D) Al

51. Which of the following species has the most unpaired electrons? S+, S, or S–? (A) S+ (B) S (C) S– (D) They all have the same number of unpaired electrons. 52. Use the Aufbau principle to obtain the ground-state electron configuration of selenium. A) Se: [Ar]4 s 23 d 104 p 3 B) Se: [Ar]4 s 23 d 104 p 4 C) Se: [Ar]4 s 23 d 104 p 5 D) Se: [Ar]4 s 23 d 104 p 6 54. How many orbitals are allowed in a subshell if the angular momentum quantum number for electrons in that subshell is 3? A. 1 B. 3 C. 5 D. 7 E. 9

55. How many electrons in a ground-state cadmium atom are in orbitals labeled by ml = -1? A. 2 B. 10 C. 12 D. 18 E. 36 56. A ground-state atom of iron has ___ unpaired electrons and is _____. A. 0, diamagnetic B. 6, diamagnetic C. 3, paramagnetic D. 5, paramagnetic E. 4, paramagnetic 57. Transition metal elements have atoms or ions with partially filled A. s subshells. B. p subshells. C. d subshells. D. f subshells. E. g subshells.