Periodic table Electron configuration of carbon atoms and

Periodic table. Electron configuration of carbon atoms and molecules. John Summerscales Jim Clark's excellent Chemguide website http: //www. chemguide. co. uk/ is recommended to support this lecture

Fundamental particles in atom Proton Neutron Electron Charge Mass Position +1 1673 x 10 -27 g in nucleus zero -1 1675 x 10 -27 g 0. 91 x 10 -27 g in nucleus in orbit Atomic number = number of protons • for balanced charge (in atom) = number of electrons • value is characteristic of a specific element Atomic weight = number of (protons + neutrons) • can have partial values as isotopes have different numbers of neutrons and different proportions
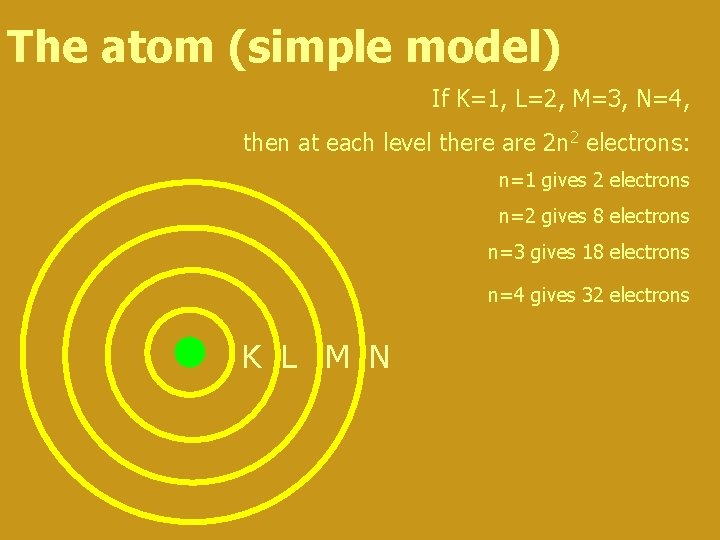
The atom (simple model) If K=1, L=2, M=3, N=4, then at each level there are 2 n 2 electrons: n=1 gives 2 electrons n=2 gives 8 electrons n=3 gives 18 electrons n=4 gives 32 electrons K L M N
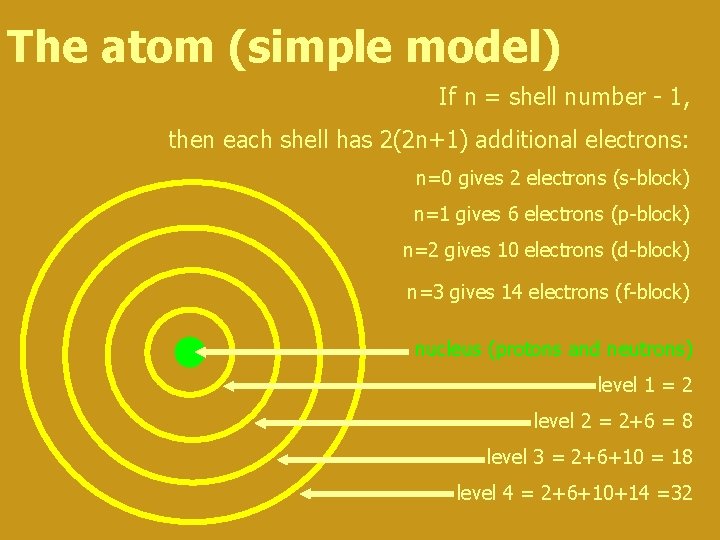
The atom (simple model) If n = shell number - 1, then each shell has 2(2 n+1) additional electrons: n=0 gives 2 electrons (s-block) n=1 gives 6 electrons (p-block) n=2 gives 10 electrons (d-block) n=3 gives 14 electrons (f-block) nucleus (protons and neutrons) level 1 = 2 level 2 = 2+6 = 8 level 3 = 2+6+10 = 18 level 4 = 2+6+10+14 =32
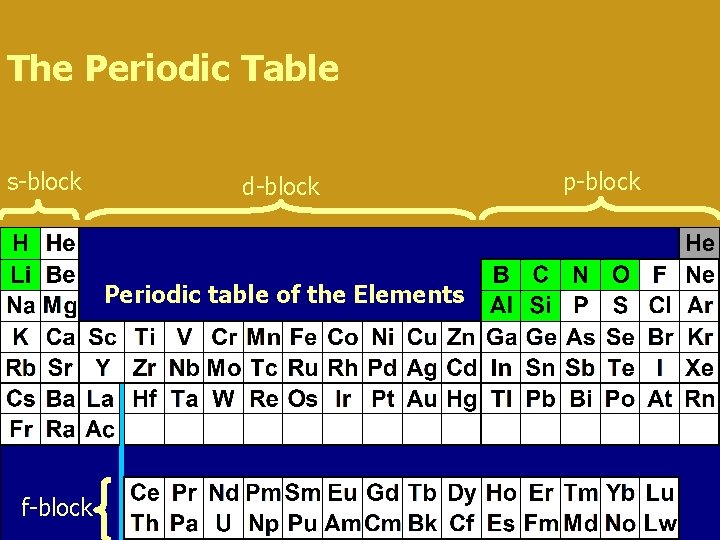
The Periodic Table s-block d-block Periodic table of the Elements f-block p-block

Inert gases Fibres - glass: B, O, Al, Si aramid: H, C, N, O Resins - H, C, N, O This column has a full electron shell - the most stable configuration Periodic table of the Elements
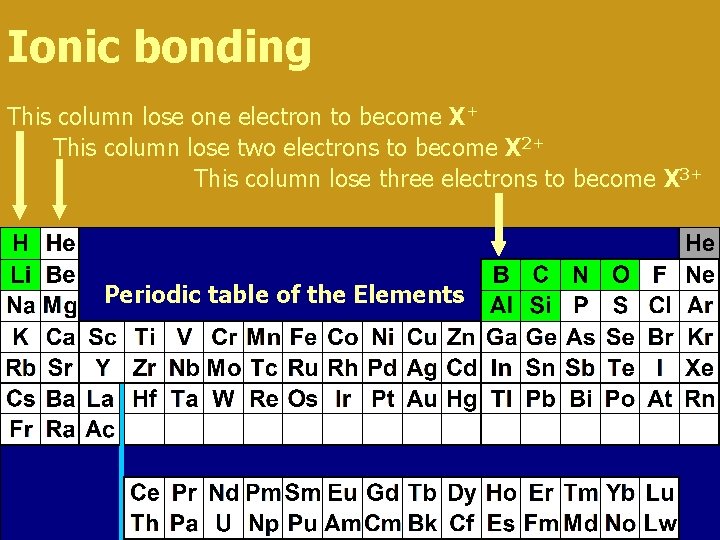
Ionic bonding This column lose one electron to become X+ This column lose two electrons to become X 2+ This column lose three electrons to become X 3+ Periodic table of the Elements
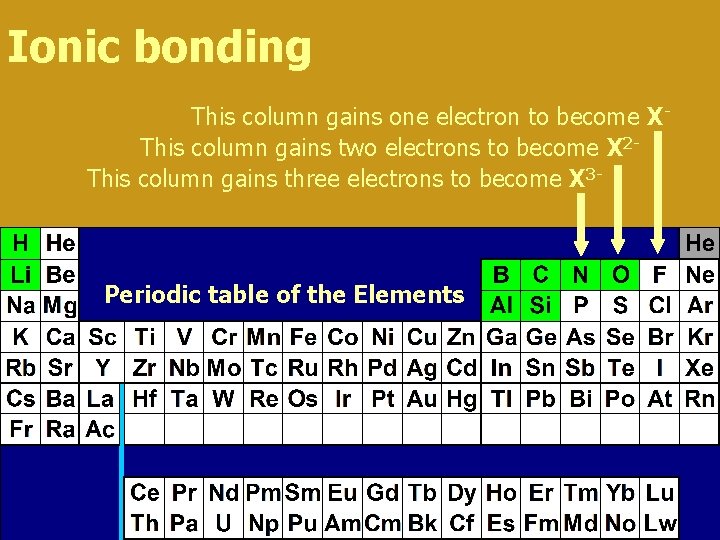
Ionic bonding This column gains one electron to become XThis column gains two electrons to become X 2 This column gains three electrons to become X 3 - Periodic table of the Elements
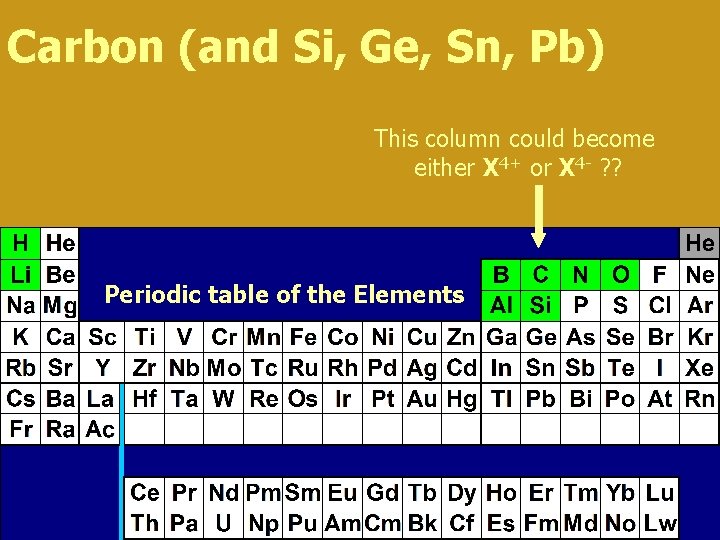
Carbon (and Si, Ge, Sn, Pb) This column could become either X 4+ or X 4 - ? ? Periodic table of the Elements

Carbon 4+ or 4 - ? ? • In practice, six protons holding three electrons > strong force/electron (difficult to remove 4 th e-) o six protons holding ten electrons > weak force/electron (difficult to retain 10 th e-) o • so, carbon shares electrons > covalent bonding • one bond. . . is. . . two shared electrons

Carbon - covalent bonding • carbon has 4 electrons in the outer shell needs four electrons to fill shell • can share with 2, 3 or 4 other atoms o 4 other atoms v o = ≡ = 3 σ and 1 double (π) bond equals used to represent a double (1σ and 1π) bond 2 other atoms v 4 single (σ) bonds hyphen used to represent a single bond between two atoms 3 other atoms v o - = = 2 σ and 2 π bonds equivalent used to represent a triple (1σ and 2π) bond but. .

Carbon - electron orbitals • electron shells divide into electron orbitals each has up to two electrons of opposite spin o electrons enter empty orbitals first o at level 2 of Periodic Table, maximum of: o 2 v 2 v electrons in in a a spherical orbital dumbbell orbit on x-axis dumbbell orbit on y-axis dumbbell orbit on z-axis
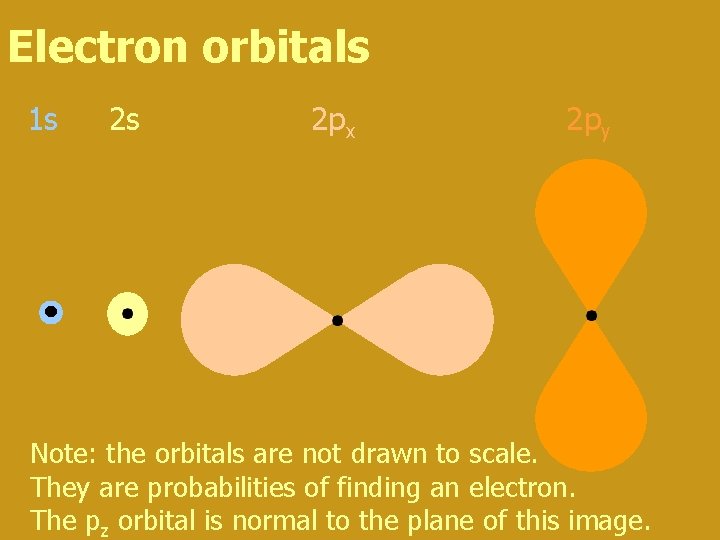
Electron orbitals 1 s 2 s 2 px 2 py Note: the orbitals are not drawn to scale. They are probabilities of finding an electron. The pz orbital is normal to the plane of this image.
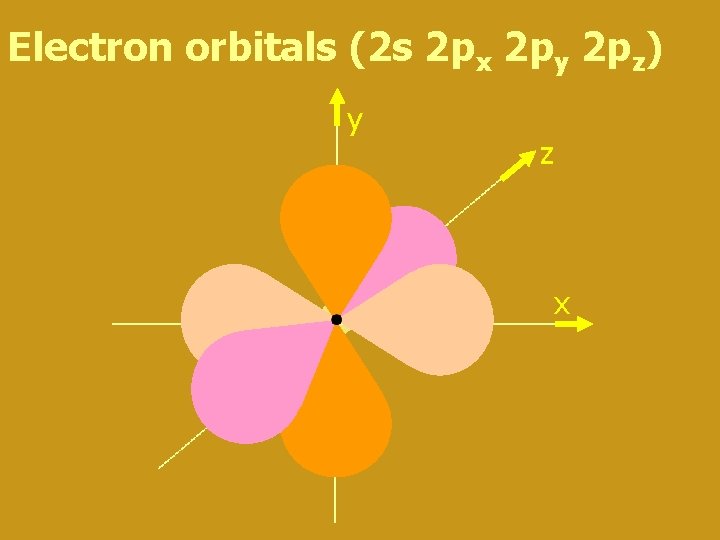
Electron orbitals (2 s 2 px 2 py 2 pz) y z x
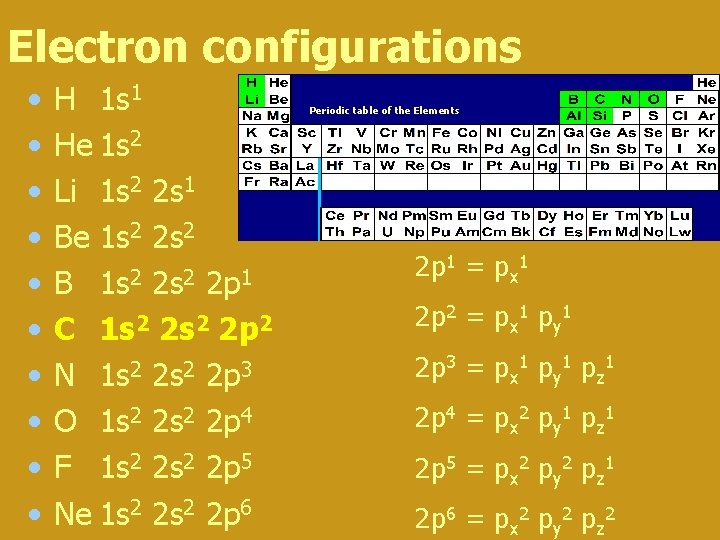
Electron configurations • • • H 1 s 1 He 1 s 2 Li 1 s 2 2 s 1 Be 1 s 2 2 s 2 B 1 s 2 2 p 1 C 1 s 2 2 p 2 N 1 s 2 2 p 3 O 1 s 2 2 p 4 F 1 s 2 2 p 5 Ne 1 s 2 2 p 6 Periodic table of the Elements 2 p 1 = px 1 2 p 2 = px 1 py 1 2 p 3 = px 1 py 1 pz 1 2 p 4 = px 2 py 1 pz 1 2 p 5 = px 2 py 2 pz 1 2 p 6 = px 2 py 2 pz 2

Methane (CH 4) • carbon bonded to four hydrogen atoms • if each H bonds to a different electron orbital the resulting molecule is asymmetric • symmetrical molecules have lowest energy and are thus the most stable form • so (2 s + 2 px +2 py + 2 pz) reorganise to four hybrid sp 3 orbitals (think s 1 p 3 !!) oriented along each line from the apex to the centre of a tetrahedron
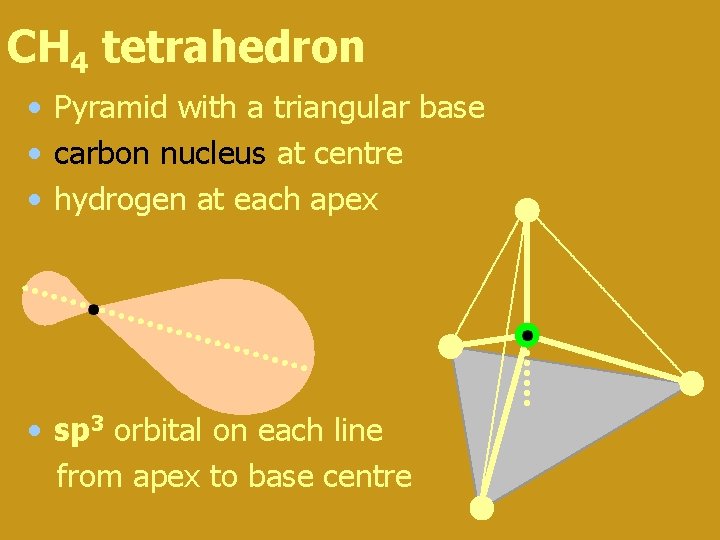
CH 4 tetrahedron • Pyramid with a triangular base • carbon nucleus at centre • hydrogen at each apex • sp 3 orbital on each line from apex to base centre
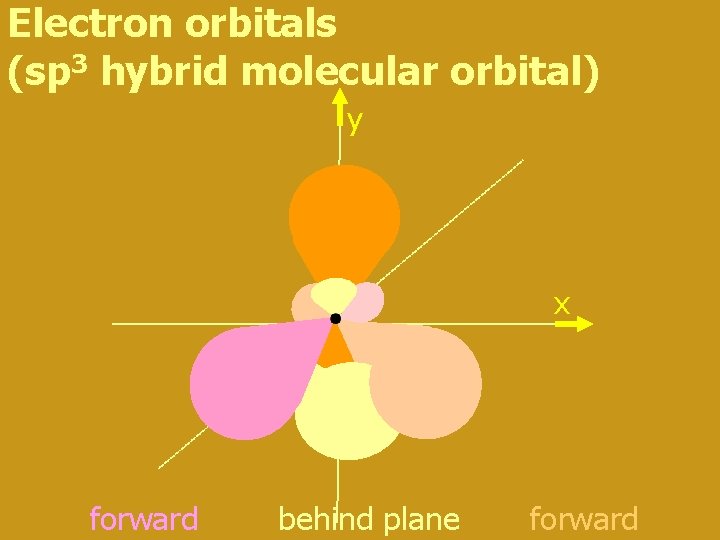
Electron orbitals (sp 3 hybrid molecular orbital) y x forward behind plane forward
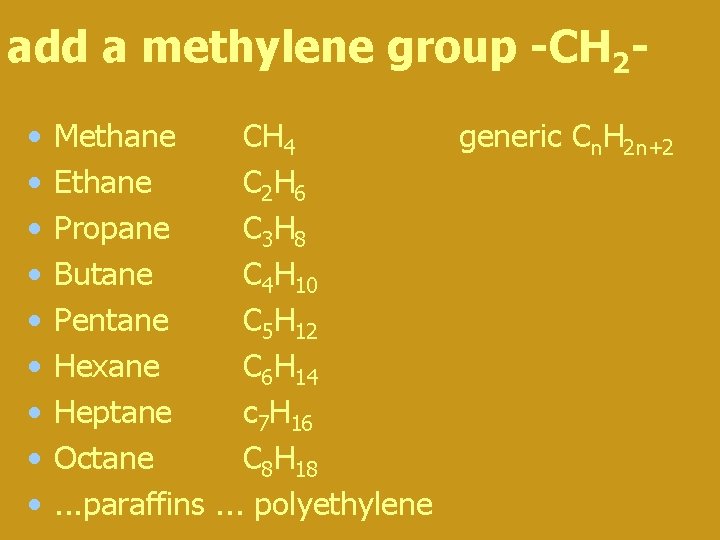
add a methylene group -CH 2 • • • Methane CH 4 generic Cn. H 2 n+2 Ethane C 2 H 6 Propane C 3 H 8 Butane C 4 H 10 Pentane C 5 H 12 Hexane C 6 H 14 Heptane c 7 H 16 Octane C 8 H 18. . . paraffins. . . polyethylene

. . . with one double bond: • • • Methene Ethene Propene Butene Pentene Hexene Heptene Octene etcetera. . n/a C 2 H 4 C 3 H 6 C 4 H 8 C 5 H 10 C 6 H 12 c 7 H 14 C 8 H 16 generic Cn. H 2 n a. k. a. ethylene a. k. a. propylene a. k. a. butylene

. . . with one triple bond: • • • Methyne Ethyne Propyne Butyne Pentyne Hexyne Heptyne Octyne etcetera. . n/a C 2 H 2 C 3 H 4 C 4 H 6 C 5 H 8 C 6 H 10 c 7 H 12 C 8 H 14 generic Cn. H 2 n-2 a. k. a. acetylene
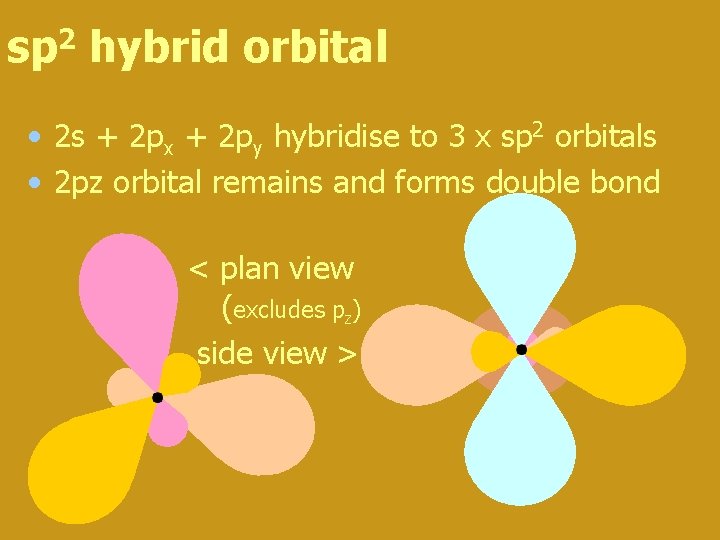
sp 2 hybrid orbital • 2 s + 2 px + 2 py hybridise to 3 x sp 2 orbitals • 2 pz orbital remains and forms double bond < plan view (excludes pz) side view > pz
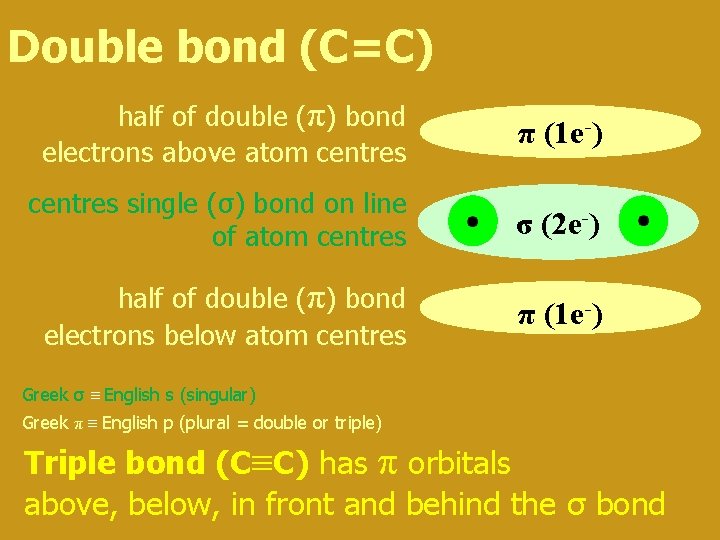
Double bond (C=C) half of double (π) bond electrons above atom centres π (1 e-) centres single (σ) bond on line of atom centres σ (2 e-) half of double (π) bond electrons below atom centres π (1 e-) Greek σ English s (singular) Greek π English p (plural = double or triple) Triple bond (C C) has π orbitals above, below, in front and behind the σ bond
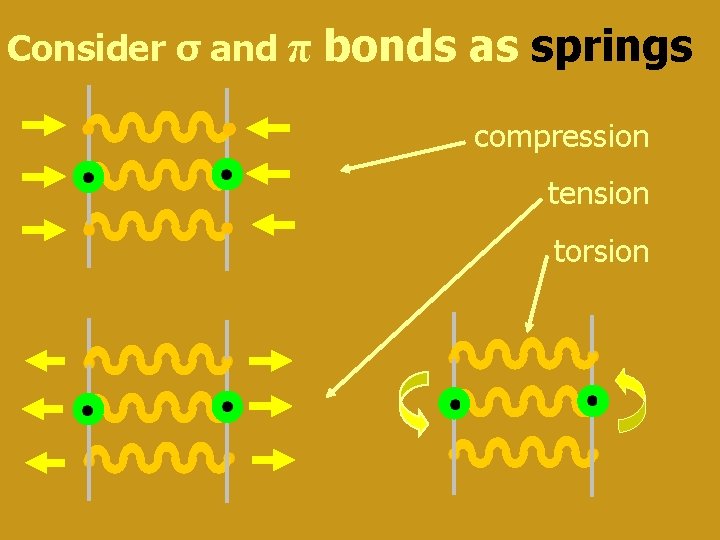
Consider σ and π bonds as springs compression tension torsion

Hybrid orbitals - summary • sp 3 bonds to 4 other atoms 4 σ (single) bonds bond angle = 109° 28’ (tetrahedral molecule) • sp 2 bonds to 3 other atoms 3 σ and 1 π bond angle = 120° (triangular molecule) 1σ and 1π bond = the double bond (i. e. 1+1 = 2) • sp bonds to 2 other atoms 2 σ and 2 π bonds bond angle = 180° (linear molecule) 1σ and 2π bonds = the triple bond (i. e. 1+2 = 3)
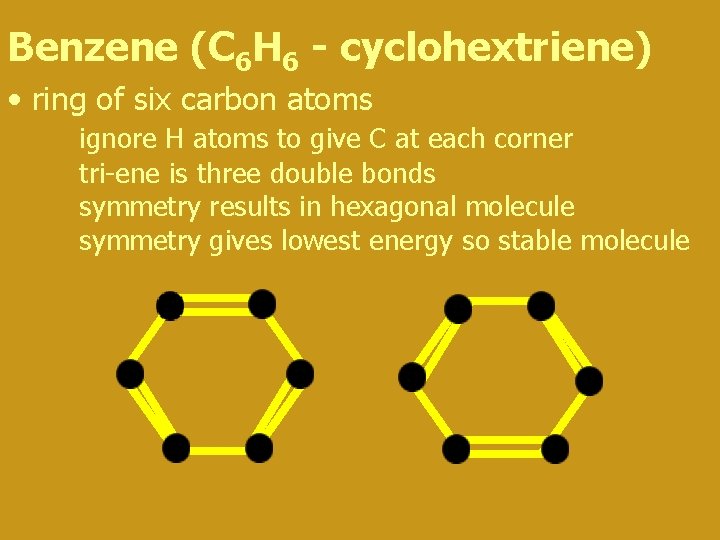
Benzene (C 6 H 6 - cyclohextriene) • ring of six carbon atoms ignore H atoms to give C at each corner tri-ene is three double bonds symmetry results in hexagonal molecule symmetry gives lowest energy so stable molecule
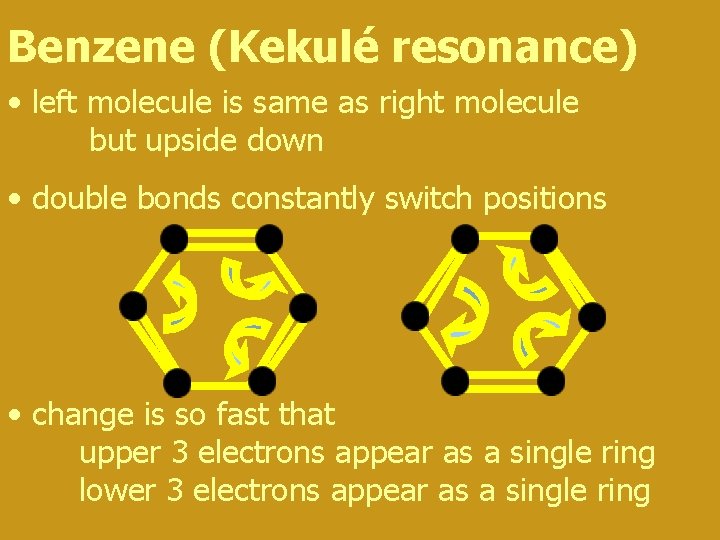
Benzene (Kekulé resonance) • left molecule is same as right molecule but upside down • double bonds constantly switch positions • change is so fast that upper 3 electrons appear as a single ring lower 3 electrons appear as a single ring
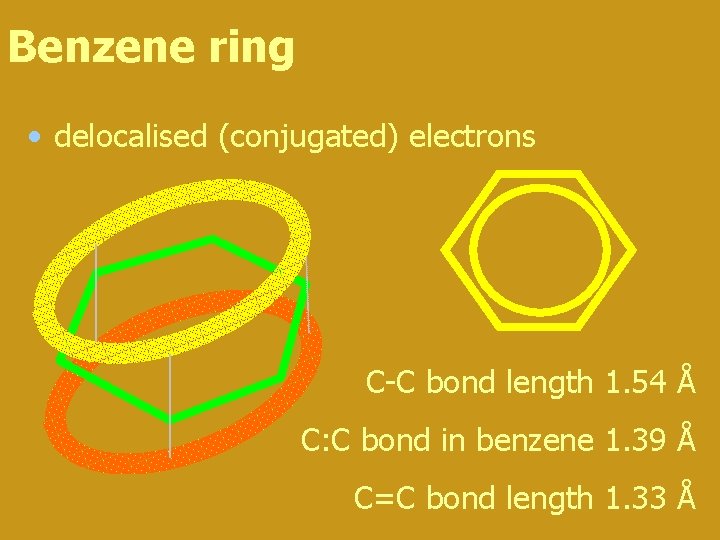
Benzene ring • delocalised (conjugated) electrons C-C bond length 1. 54 Å C: C bond in benzene 1. 39 Å C=C bond length 1. 33 Å

Graphite (E in-plane ~ ? ? ? GPa)

Elastic modulus • • • Material epoxy resin glass aluminium steel graphite (in-plane) Modulus Rel. density 3 GPa 1. 15 70 GPa 2. 56 70 GPa 2. 7 210 GPa 8 1000 GPa 2. 266

Conclusion Chemical bond type and chemical bond density each determine material stiffness/strength and chemical durability
- Slides: 31