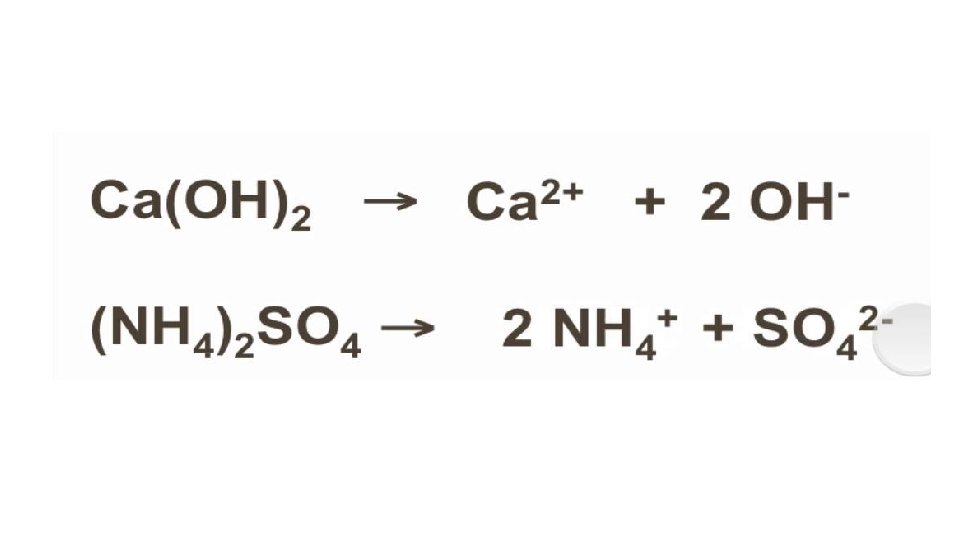Solution Chemistry Solution Chemistry Chemical reactions that occur

















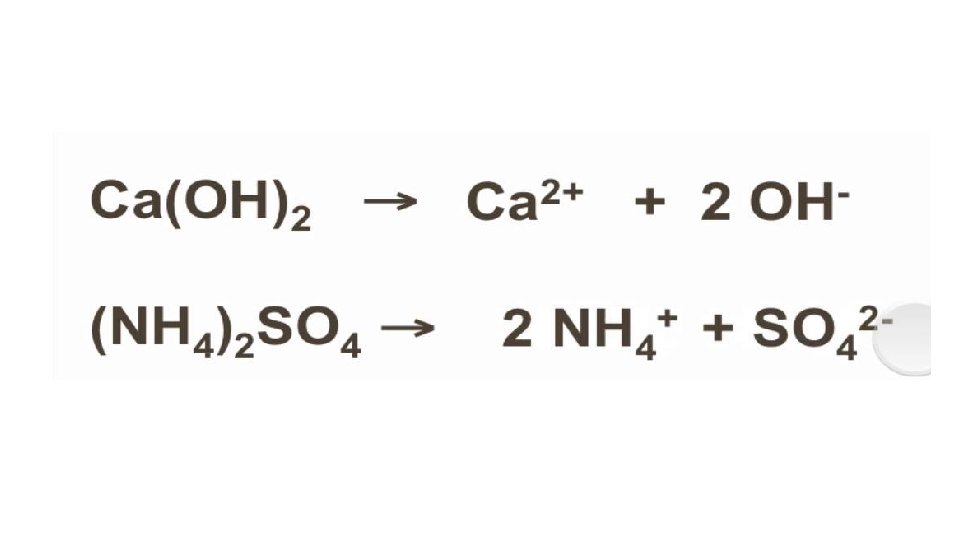
- Slides: 32

Solution Chemistry

Solution Chemistry • Chemical reactions that occur in solutions • Convenient • Easily handled • Concentrations can be changed • Concentrations easily determined • Many solvents • Definitions: • • Solution – homogenous mixture Solvent – component in the mixture in the greatest quantity Solute - component in the mixture in the smaller quantity Soluble – if the solute and solvent for a homogenous mixture – Insoluble –little or no solute dissolves in solvent

Definitions Cont’d • Saturated – solvent is saturated when the solute has dissolved as much as possible • Must be a constant amount of dissolved solute if kept in a sealed container and a constant temperature • Unsaturated – solvent is unsaturated when it can dissolve more solute

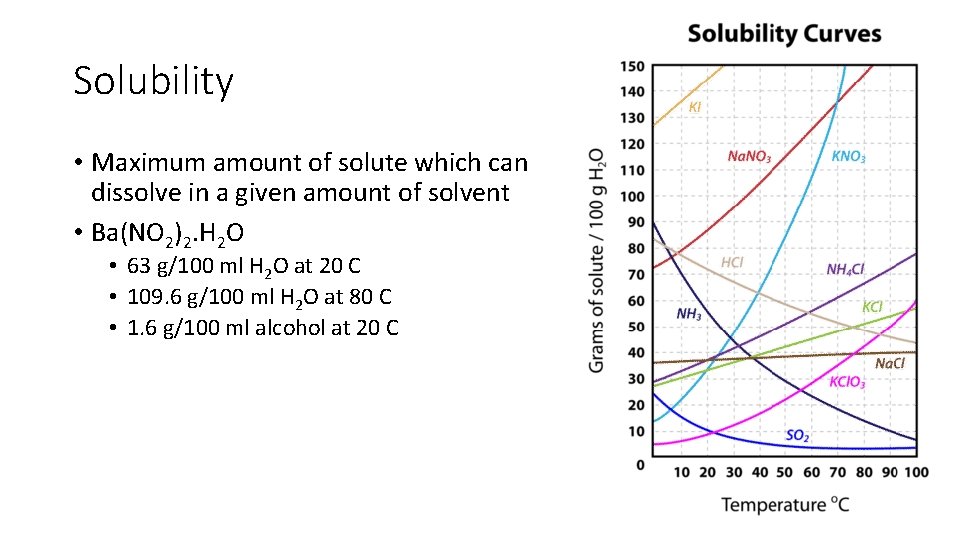
Solubility • Maximum amount of solute which can dissolve in a given amount of solvent • Ba(NO 2)2. H 2 O • 63 g/100 ml H 2 O at 20 C • 109. 6 g/100 ml H 2 O at 80 C • 1. 6 g/100 ml alcohol at 20 C

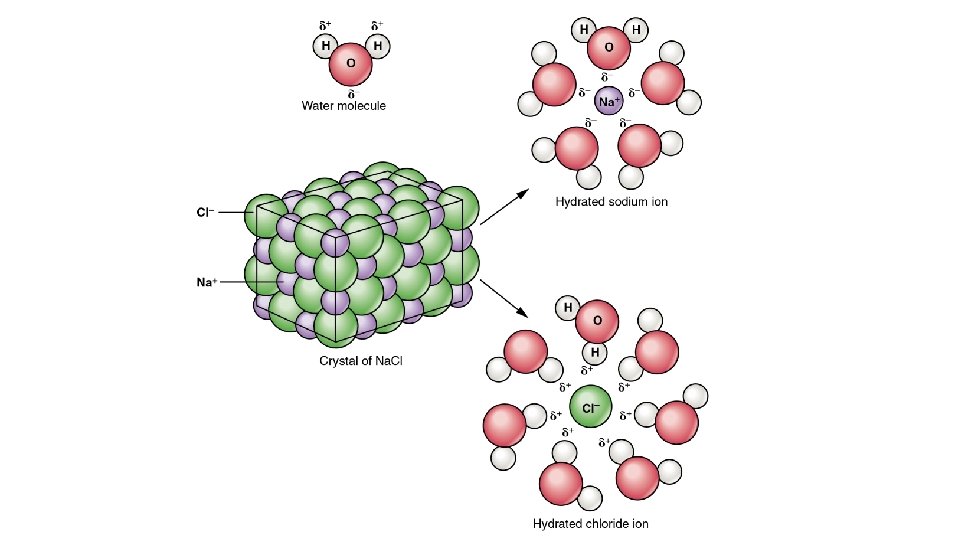
Conductivity of Aqueous Solutions • Ions – have electric charge • Ionic solution conduct electricity • Generally: • Conducting solutions contain ions, the greater the concentration the more conductivity • Nonmetal and nonmetal solution will not form an ionic solution. Little to no conductivity • Carbon compounds do not form ionic solutions in water. Except organic acids


Substance vs Conductivity

State vs Conductivity

Concentration vs Conductivity


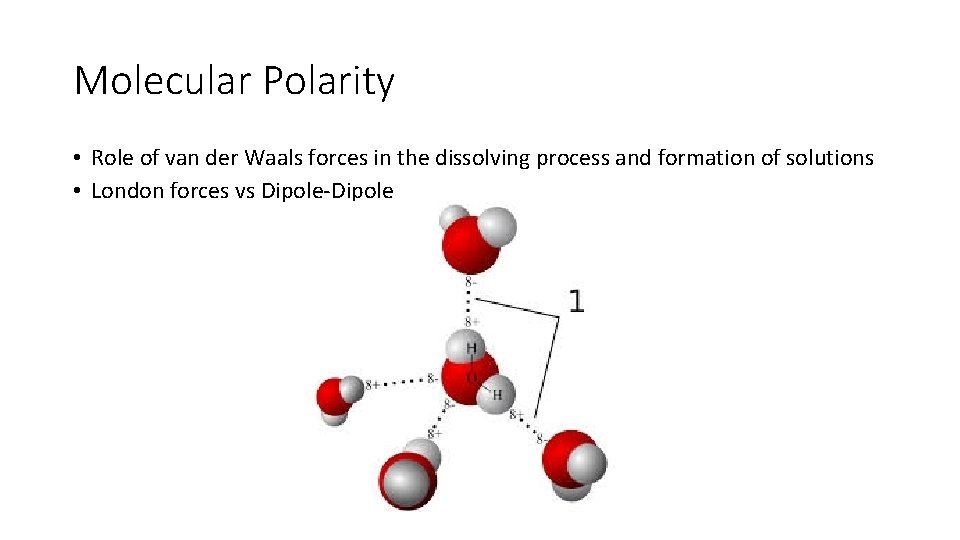
Molecular Polarity • Role of van der Waals forces in the dissolving process and formation of solutions • London forces vs Dipole-Dipole

London Forces • Weak attractive forces as a result of temporary attractions between neighboring molecules • If permanent dipole is absent then only van der Waals present

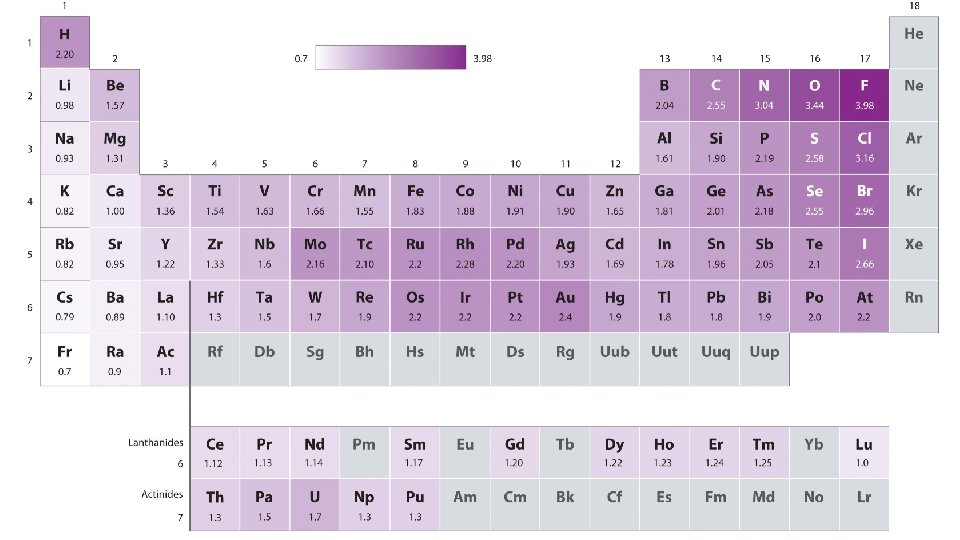
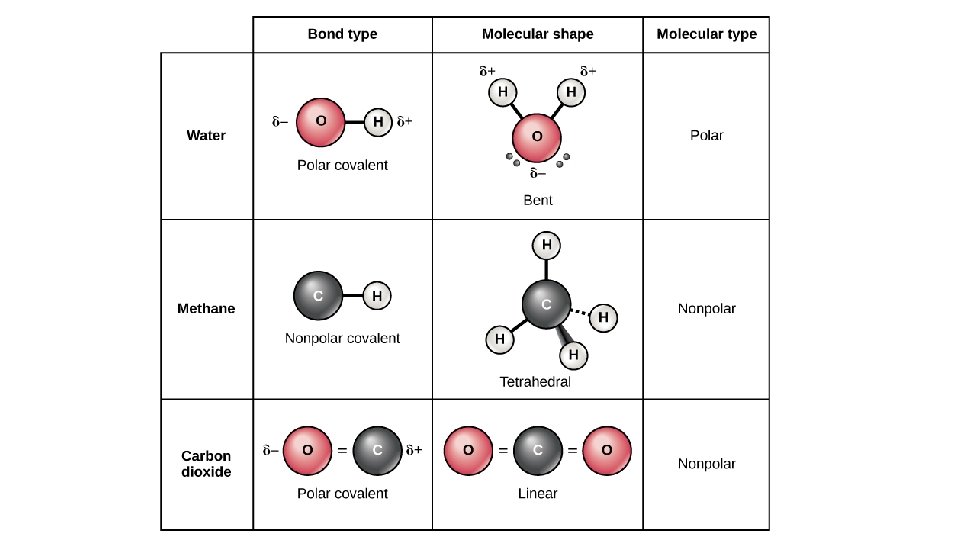
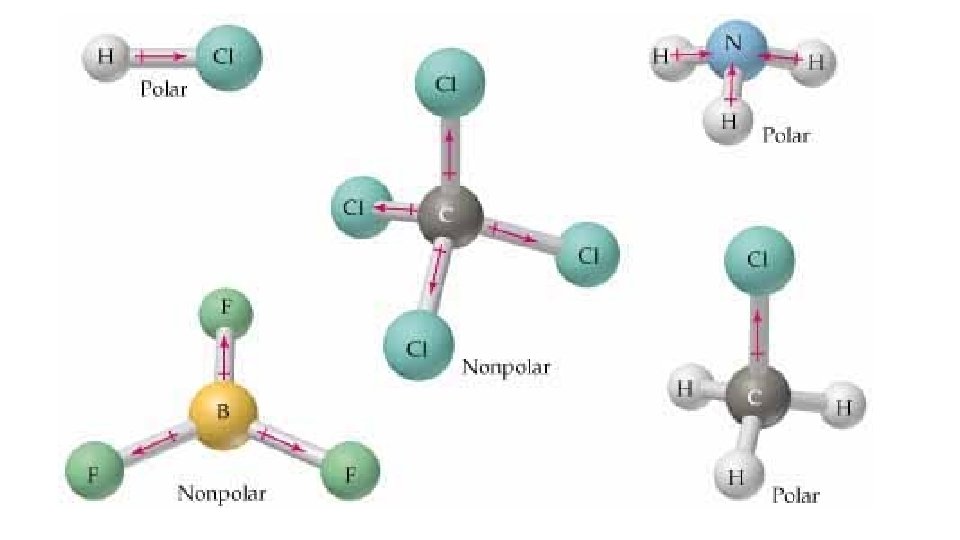
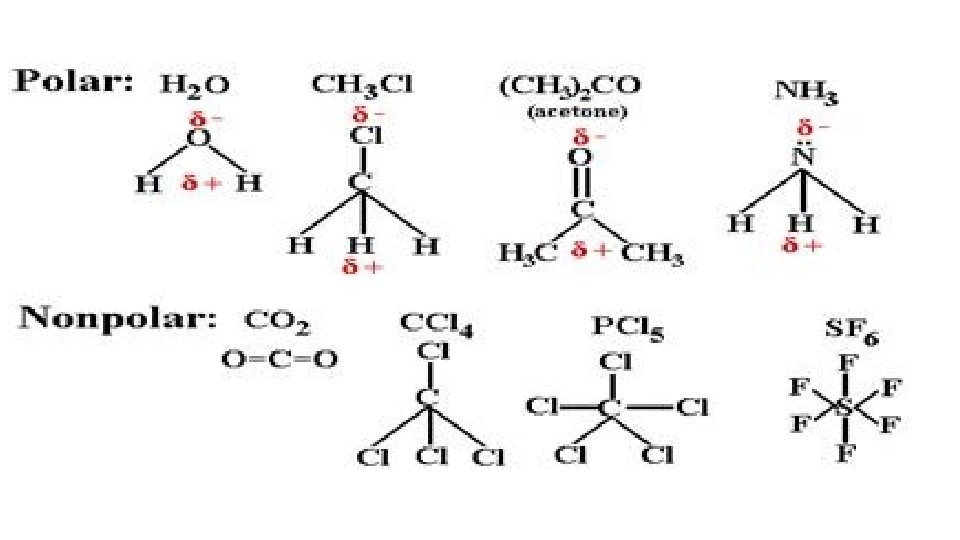
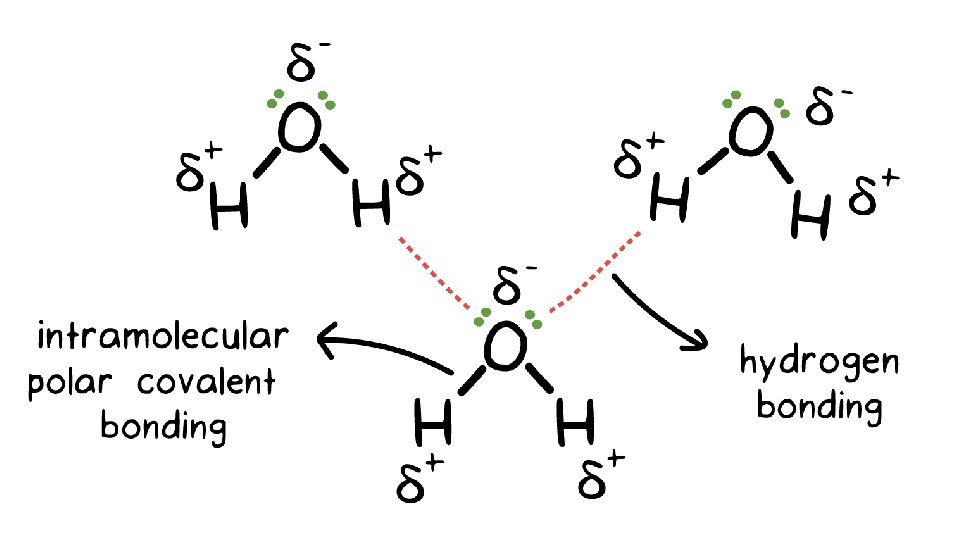
Dipole-Dipole (permanent) • Dipole is a partial separation of charge when one end of a molecule has a slight excess of negative charge and the other end has a slight excess of positive charge. • Dipole-Dipole Forces – bonding forces which exist as a result to the electrostatic attraction of molecules with permanent dipoles. • Bonding of atoms with different electronegativities leads to dipoles. • BUT, must be asymmetrical molecule • Note – London Forces add to Dipole-Dipole





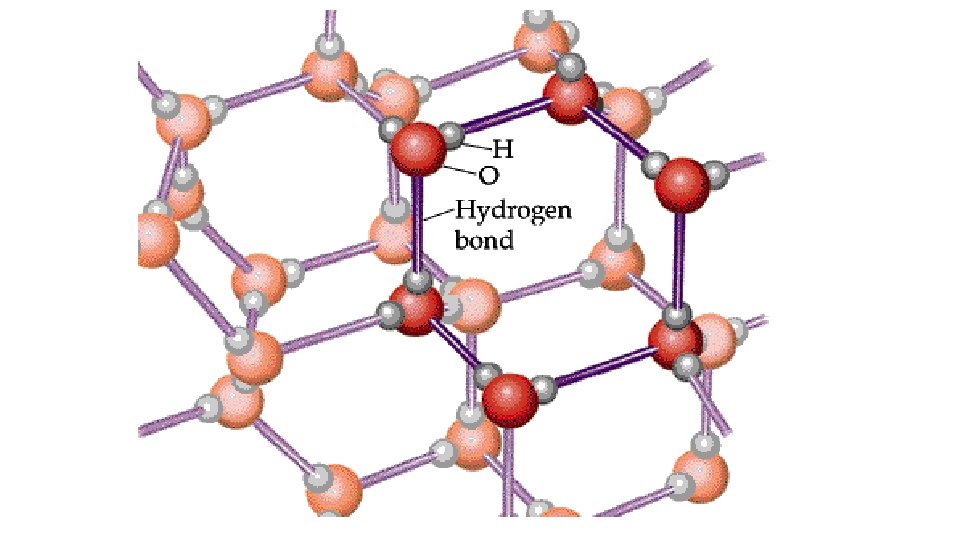
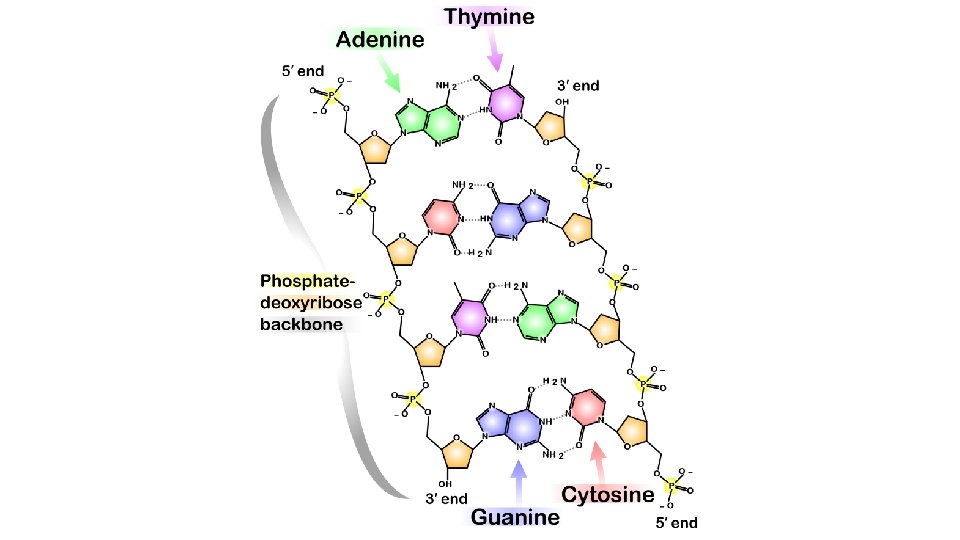
Hydrogen Bonding • A fairly strong dipole-dipole attraction exists when H is bonded to one of N, O, or F • Examples: a. HF b. NH 3, NH 2, NH (i. e. CH 3 NH 2 , (CH 3)2 NH c. H 2 O, H 2 O 2, OH (i. e. CH 3 OH)




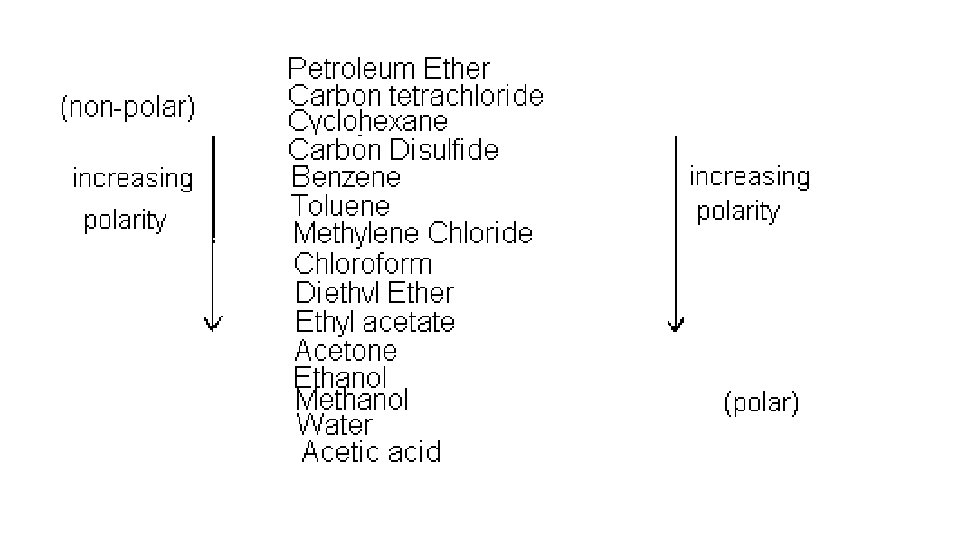
Polar and Non-Polar Solvents • Key Point: Like Dissolves Like



Why Like Dissolves Like: Polar • Ionic compounds - + and – ions bound together strongly as a solid • Three types of attractions • Attraction of solvent-solvent molecules • Attraction of solvent-solute particles • Attraction of solute-solute particle • To break bonds, energy is needed and energy is released when a bond forms • If the solvent forms strong enough bond with the solute then a solute can separate. • Solvent-solvent attraction allows solvent-solute to mix in the general population of solvent molecules • Conclusion: polar and ionic solutes have low solubility in non-polar solvents

Solubility of Non-Polar Solutes • Non-polar solvents do not possess charges – no polar attraction of polar solute = no dipoles • Only attracted to solutes by London Forces • Non-polar solvent London forces over come the weak London forces of solute = dissolves

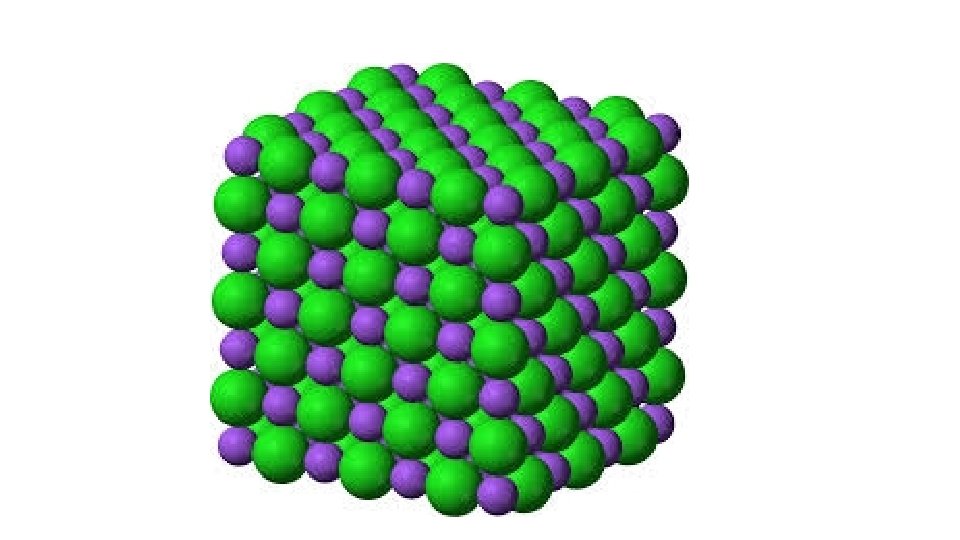
Solution of Ions • Complex! • Definitions: • Solvation – interaction between solute and solvent • Ionic Solid – solid whose crystal structure is made up of ions • Molecular Solid – a solid whose crystal structure is made up of neutral ions



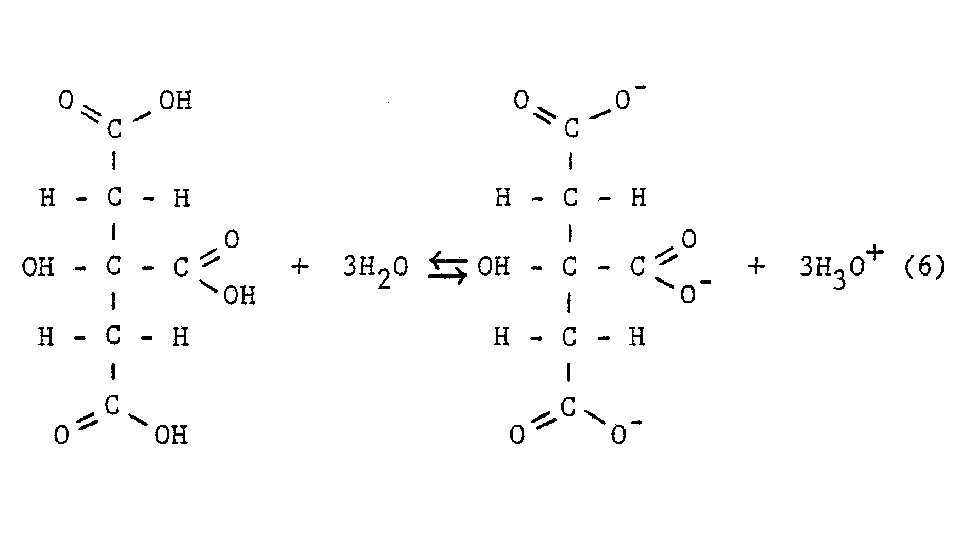
Definitions cont’d • Dissociation – • Separation of previously existing ions as ionic solid • Ions are already ions in which the solvent separates them from each other • Ionization Reaction – • Ions do not exist until the solvent is able to react with the molecules to break it apart into ions