Organic Chemistry Sh Javanshir Faculty of Chemistry Iran











- Slides: 41

Organic Chemistry Sh. Javanshir Faculty of Chemistry Iran University of Science & Technology 1

Chapter 4. Alkenes: Structure and Reactivity Based on Mc. Murry’s Organic Chemistry, 6 th edition 2

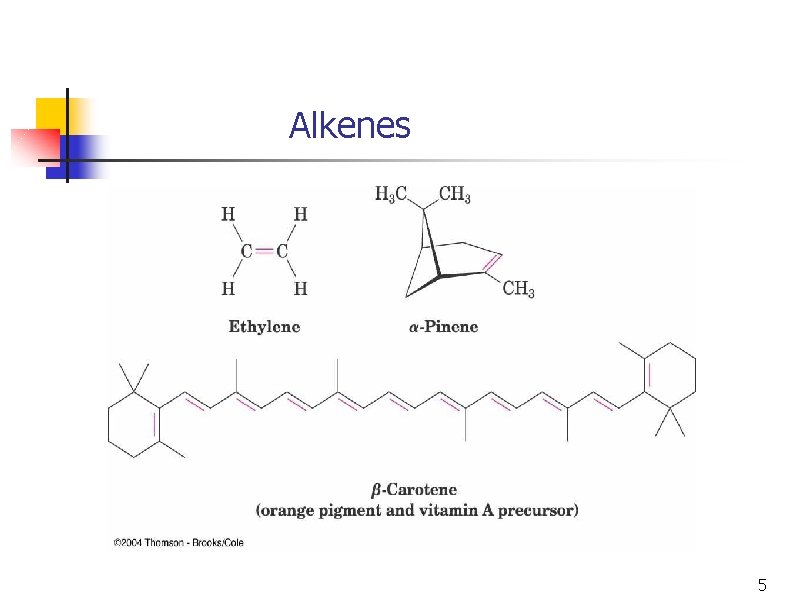
Alkene - Hydrocarbon With Carbon-Carbon Double Bond n n Includes many naturally occurring materials n Flavors, fragrances, vitamins Important industrial products n These are feedstocks for industrial processes 3

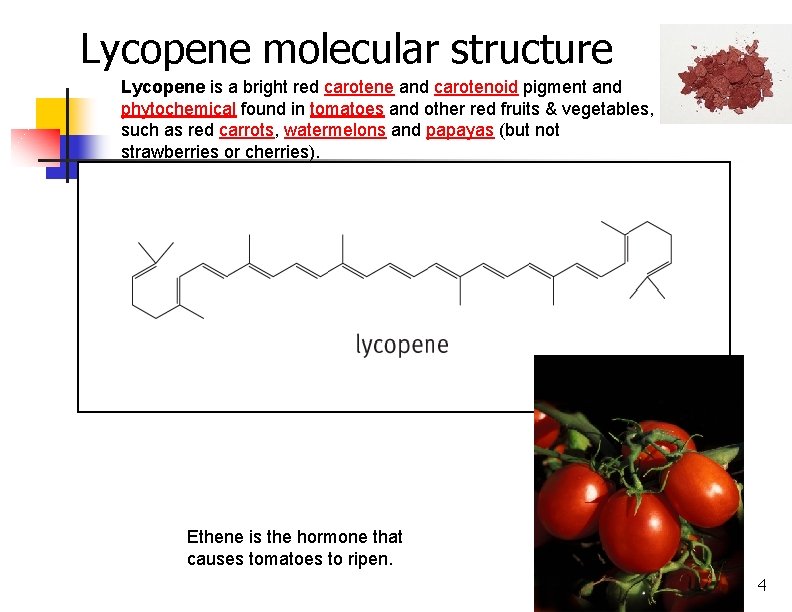
Lycopene molecular structure Lycopene is a bright red carotene and carotenoid pigment and phytochemical found in tomatoes and other red fruits & vegetables, such as red carrots, watermelons and papayas (but not strawberries or cherries). Ethene is the hormone that causes tomatoes to ripen. 4

Alkenes 5

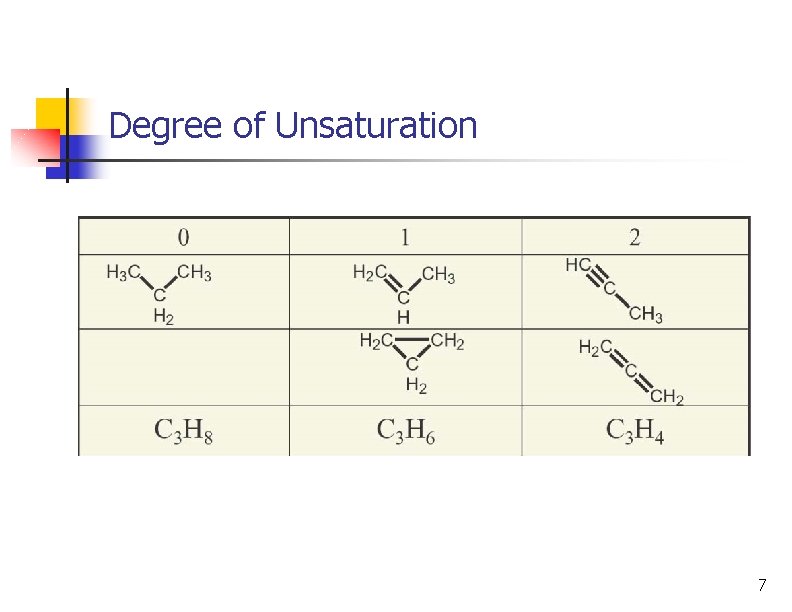
n Relates molecular formula to possible structures Degree of Unsaturation n Degree of unsaturation: number of multiple bonds or rings Formula for saturated a acyclic compound is Cn. H 2 n+2 Each ring or multiple bond replaces 2 H's 6

Degree of Unsaturation 7

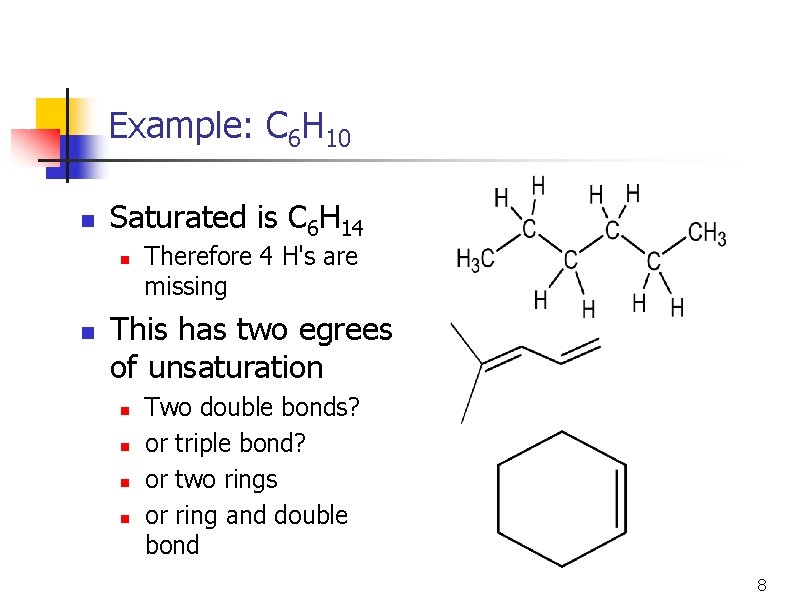
Example: C 6 H 10 n Saturated is C 6 H 14 n n Therefore 4 H's are missing This has two egrees of unsaturation n n Two double bonds? or triple bond? or two rings or ring and double bond 8

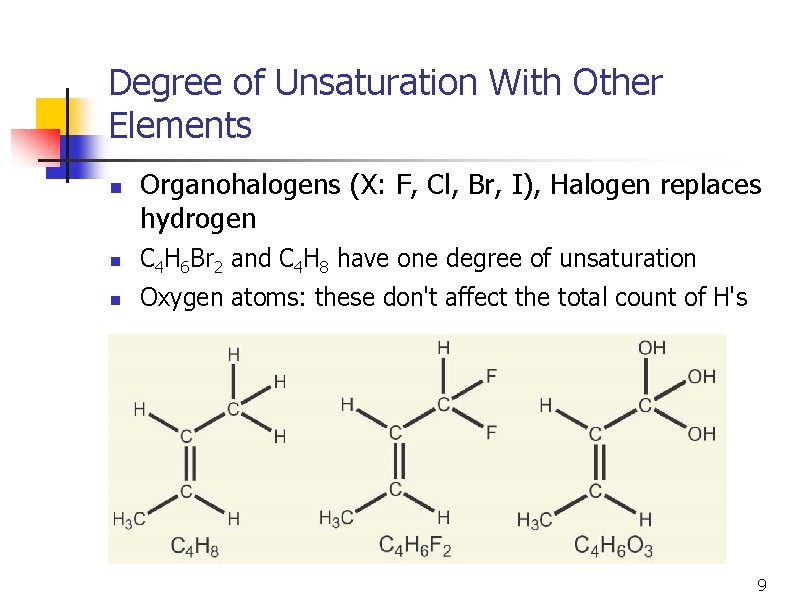
Degree of Unsaturation With Other Elements n Organohalogens (X: F, Cl, Br, I), Halogen replaces hydrogen n C 4 H 6 Br 2 and C 4 H 8 have one degree of unsaturation n Oxygen atoms: these don't affect the total count of H's 9

Degree of Unsaturation With Other Elements 10

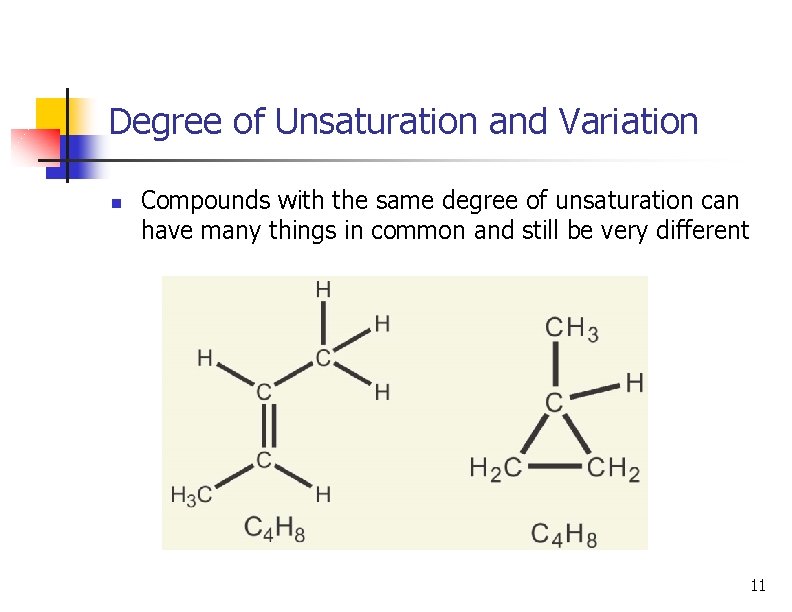
Degree of Unsaturation and Variation n Compounds with the same degree of unsaturation can have many things in common and still be very different 11

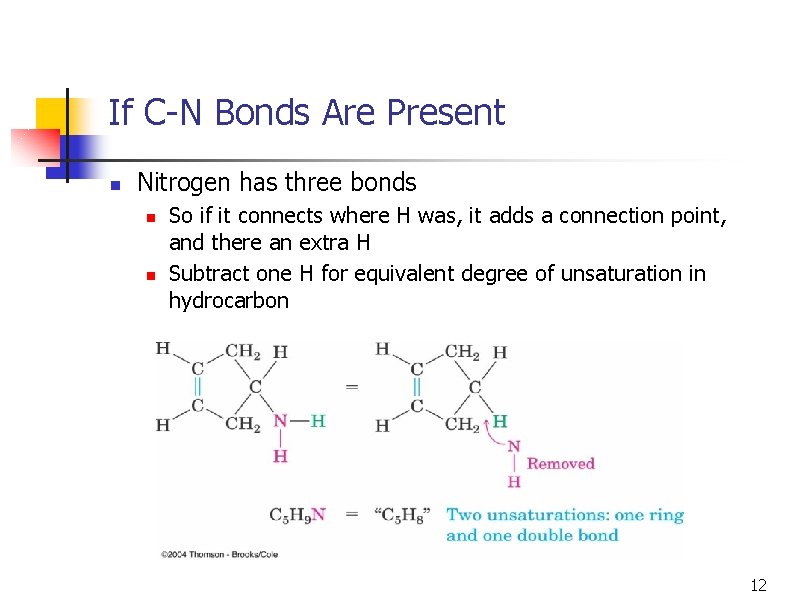
If C-N Bonds Are Present n Nitrogen has three bonds n n So if it connects where H was, it adds a connection point, and there an extra H Subtract one H for equivalent degree of unsaturation in hydrocarbon 12

Summary - Degree of Unsaturation n Count pairs of H's below Cn. H 2 n+2 Add number of halogens to number of H's (X equivalent to H) Don't count oxygens (oxygen links H) Subtract NH's DU=X-(Y/2 -Z/2)+1 ﺩﺭﺟﻪ ﻏﻴﺮ ﺍﺷﺒﺎﻋی n X=nbr of C , y=nbr of H, Z=nbr of N n n 13

Naming of Alkenes n n n Find longest continuous carbon chain for root Number carbons in chain so that double bond carbons have lowest possible numbers Rings have “cyclo” prefix 14

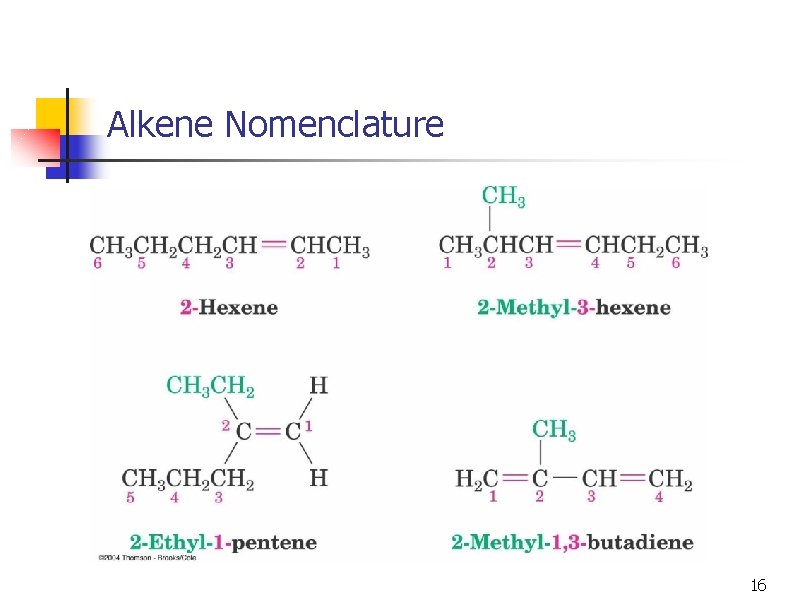
Alkene Nomenclature: longest chain must contain the C=C 15

Alkene Nomenclature 16

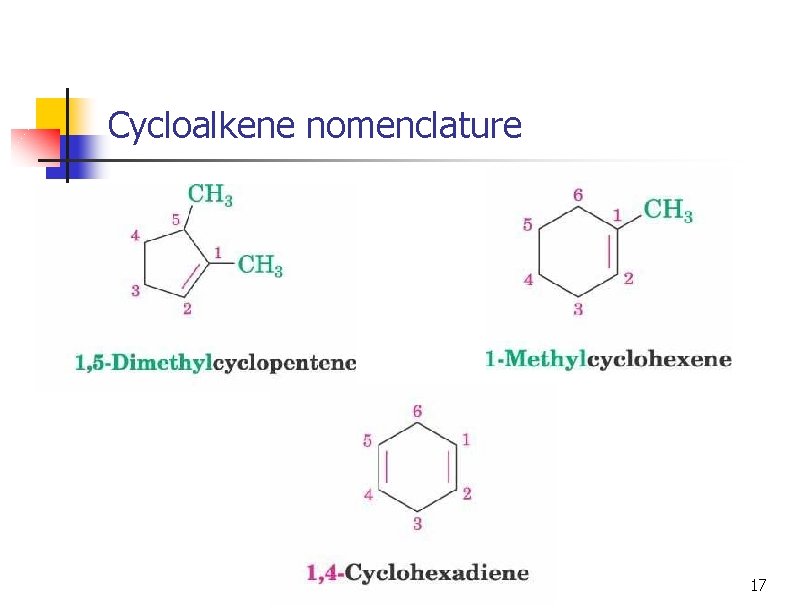
Cycloalkene nomenclature 17

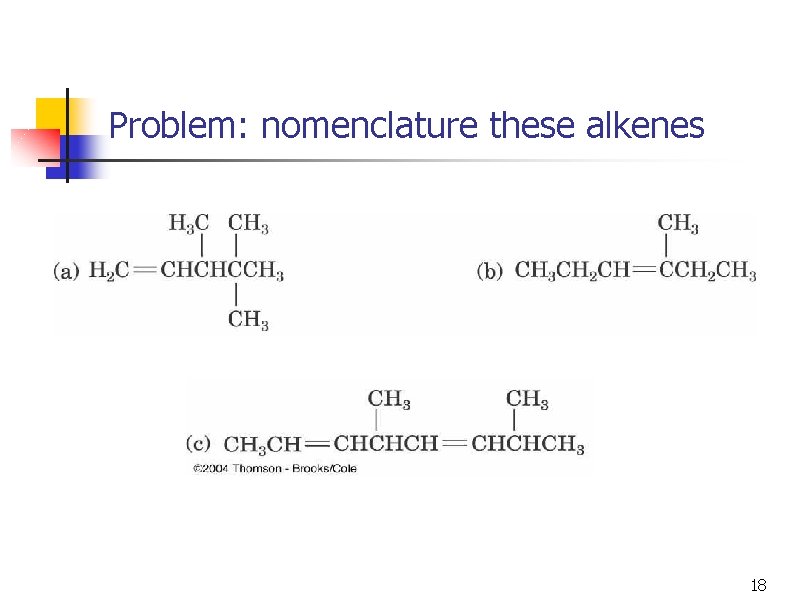
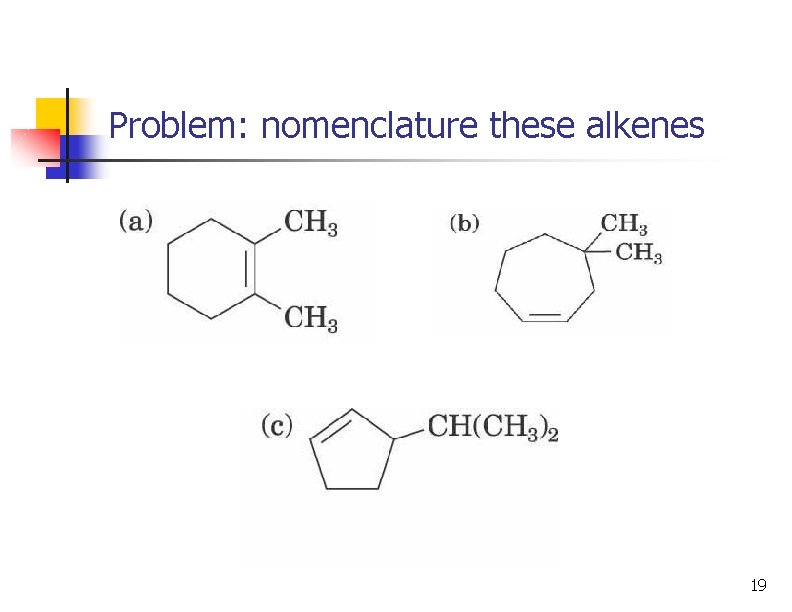
Problem: nomenclature these alkenes 18

Problem: nomenclature these alkenes 19

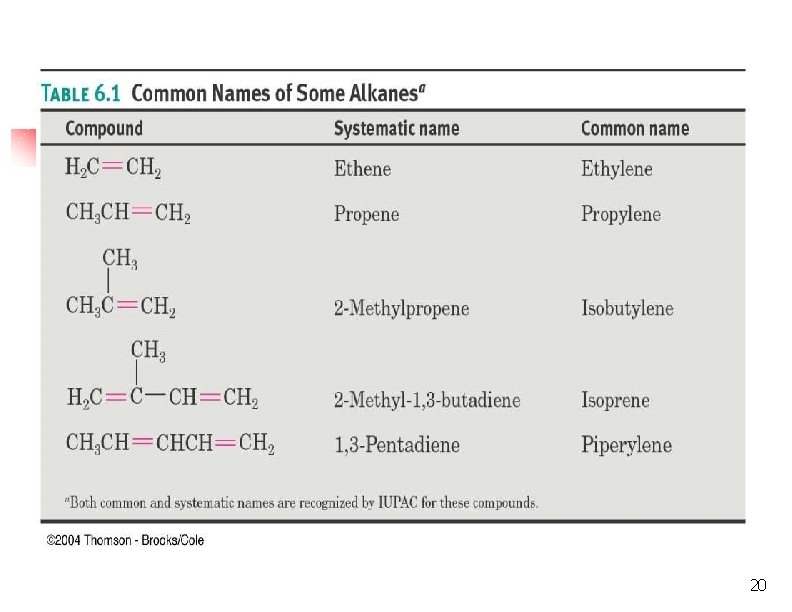
20

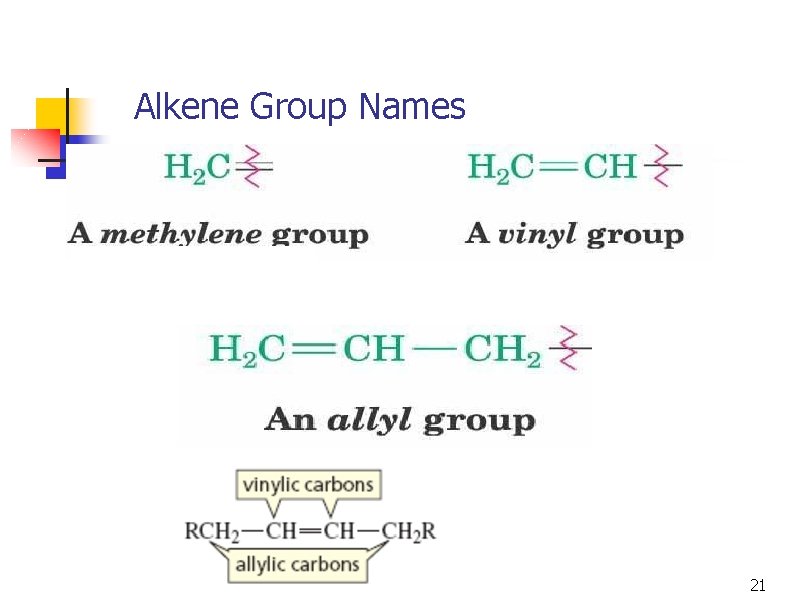
Alkene Group Names 21

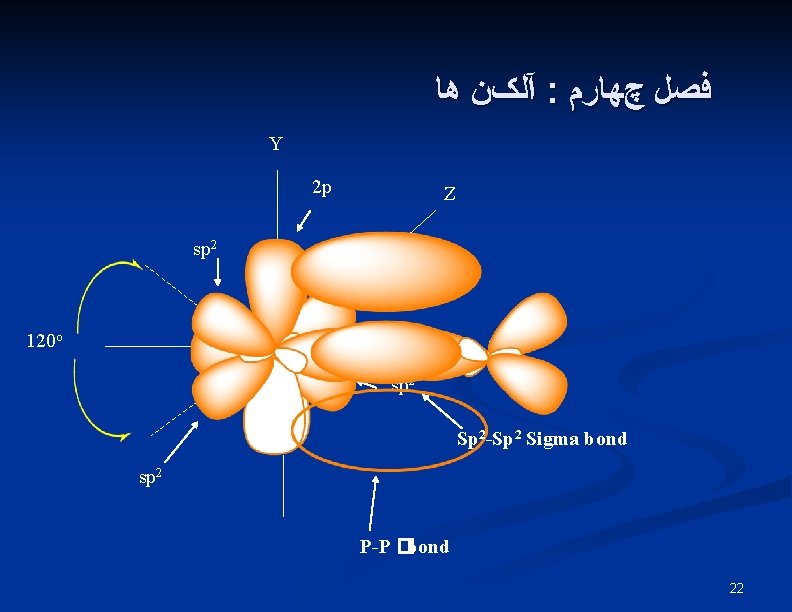
آﻠکﻦ ﻫﺎ : ﻓﺼﻞ چﻬﺎﺭﻡ Y 2 p Z sp 2 120 o X sp 2 Sp 2 -Sp 2 Sigma bond sp 2 P-P �bond 22



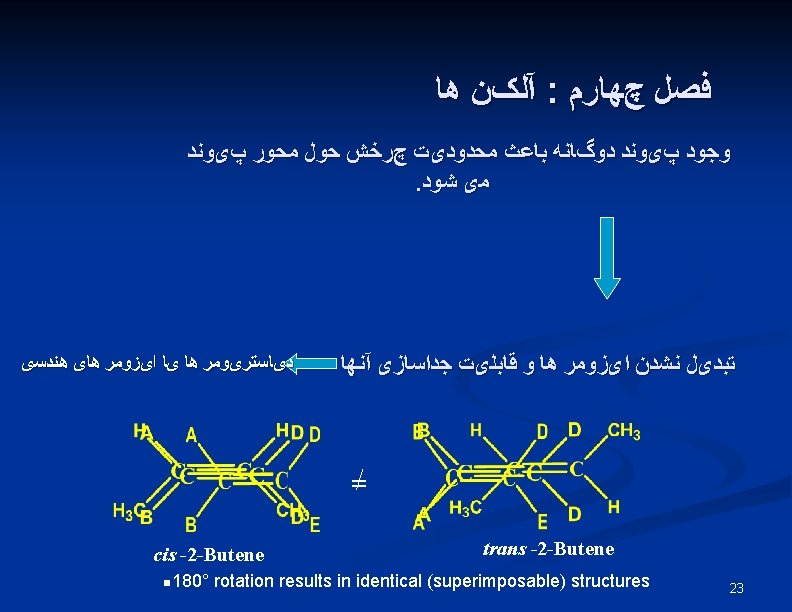
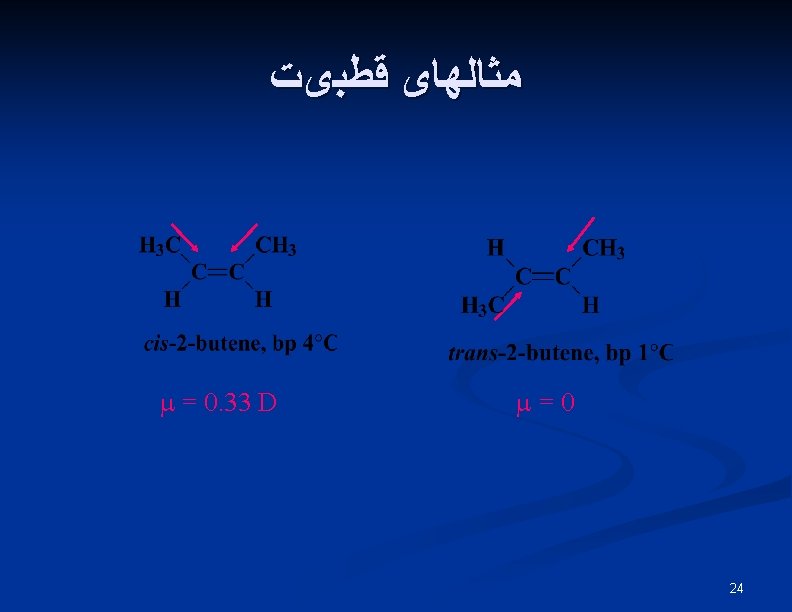
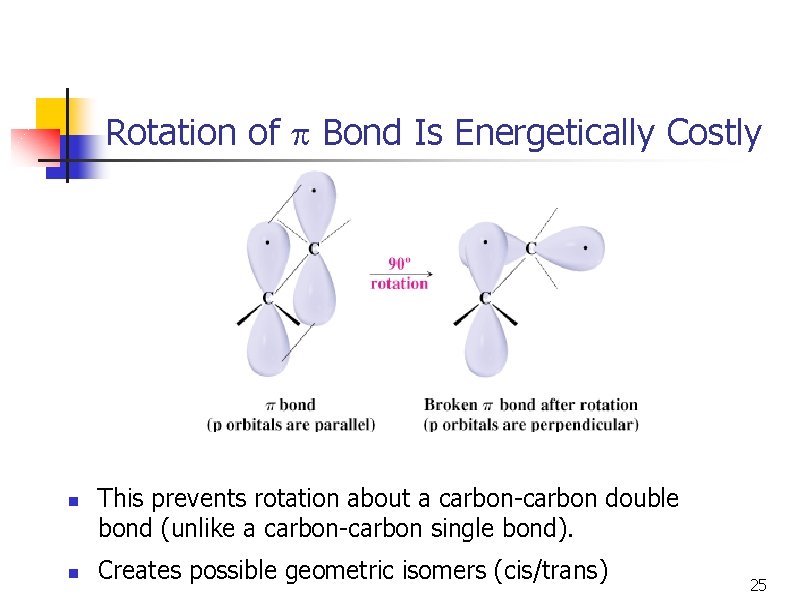
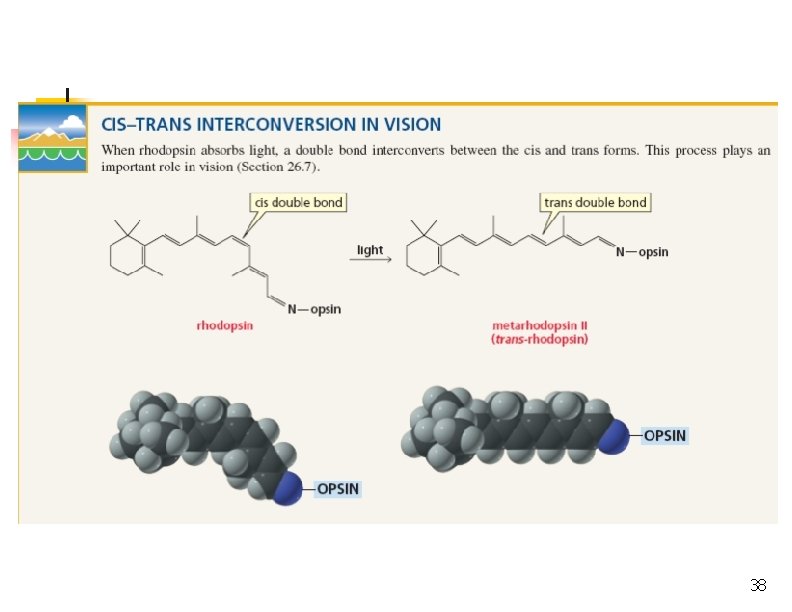
Rotation of Bond Is Energetically Costly n n This prevents rotation about a carbon-carbon double bond (unlike a carbon-carbon single bond). Creates possible geometric isomers (cis/trans) 25

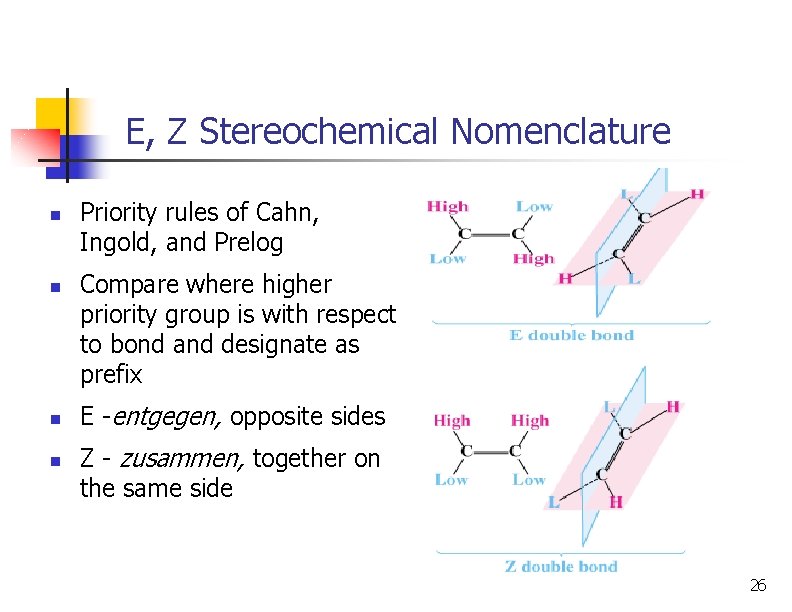
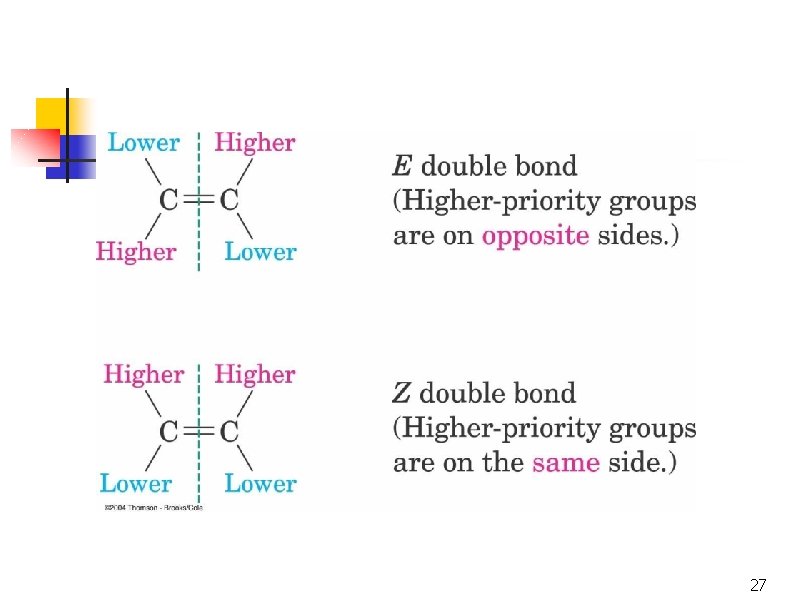
E, Z Stereochemical Nomenclature n n Priority rules of Cahn, Ingold, and Prelog Compare where higher priority group is with respect to bond and designate as prefix E -entgegen, opposite sides Z - zusammen, together on the same side 26

27

Ranking Priorities: Cahn-Ingold-Prelog Rules n n Must rank atoms that are connected at comparison point Higher atomic number gets higher priority n Br > Cl > O > N > C > H In this case, The higher priority groups are opposite: (E )-1 -bromo-1 -chloro-propene 28

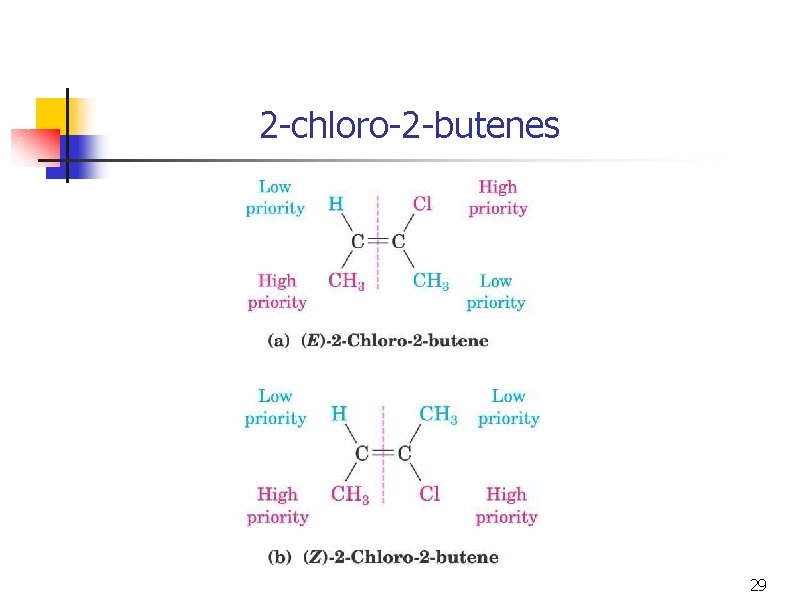
2 -chloro-2 -butenes 29

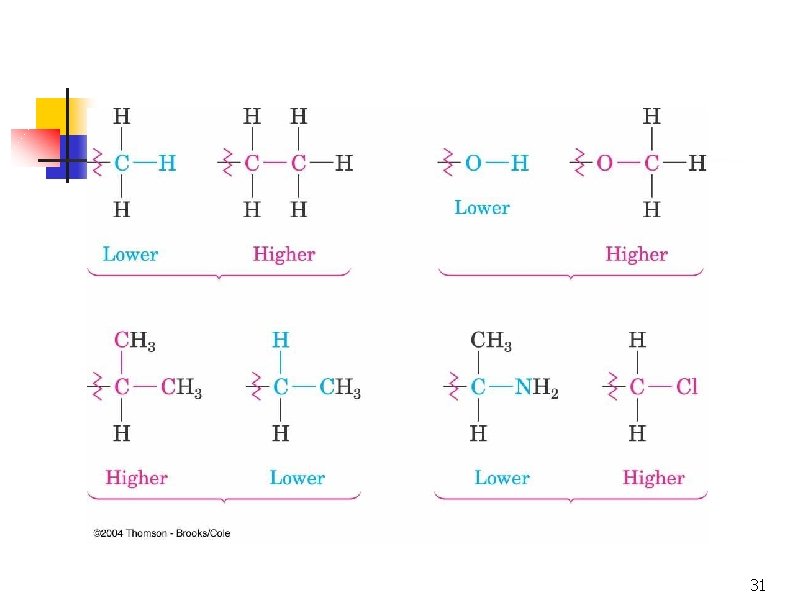
Extended Comparison n If atomic numbers are the same, compare at next connection point at same distance Compare until something has higher atomic number Do not combine – always compare 30

31

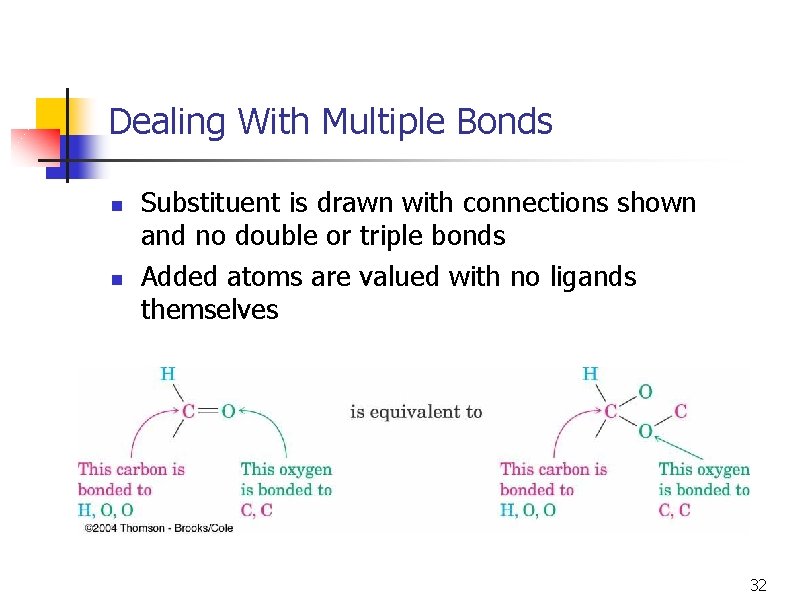
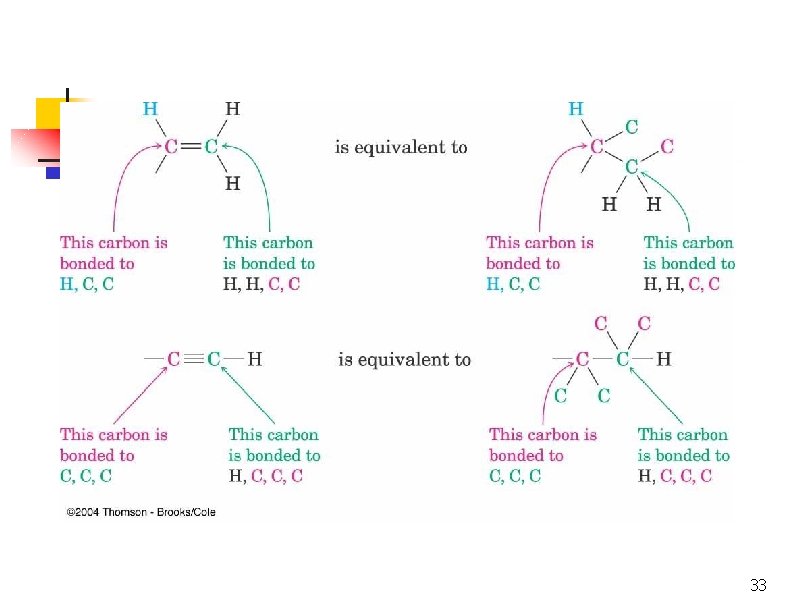
Dealing With Multiple Bonds n n Substituent is drawn with connections shown and no double or triple bonds Added atoms are valued with no ligands themselves 32

33

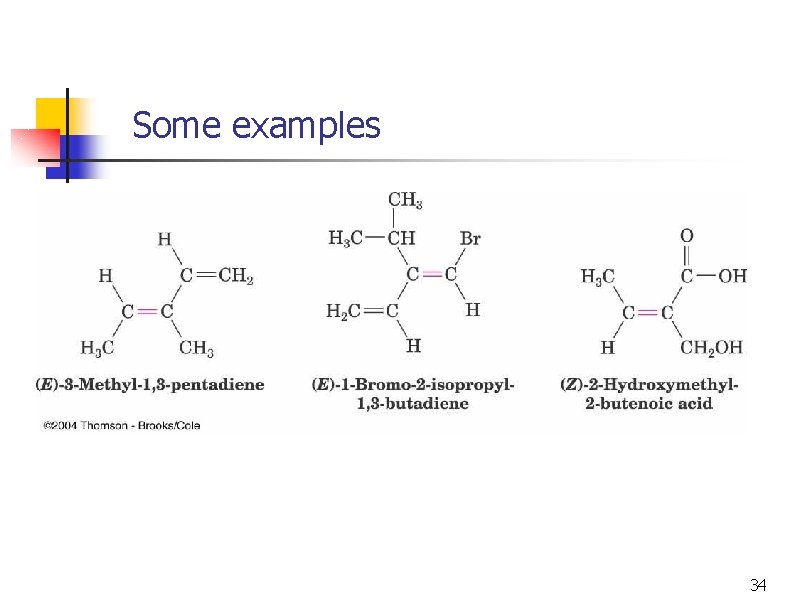
Some examples 34

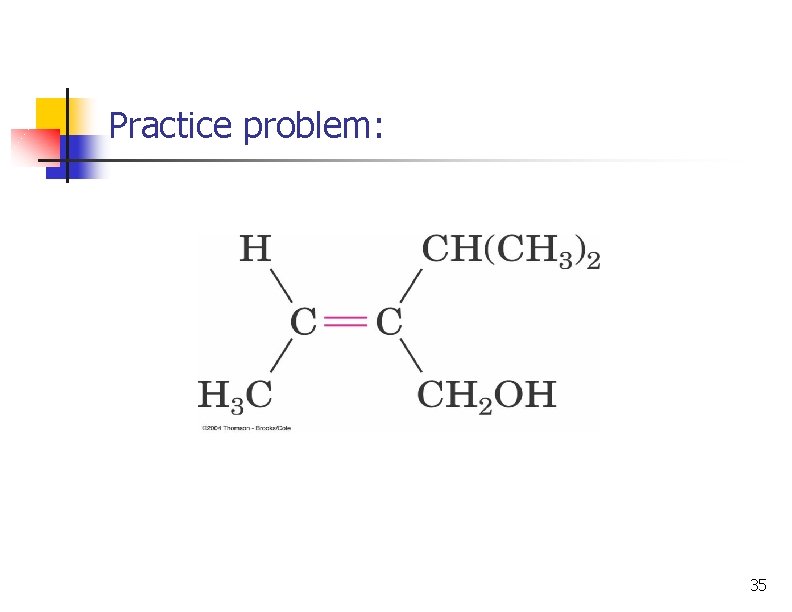
Practice problem: 35

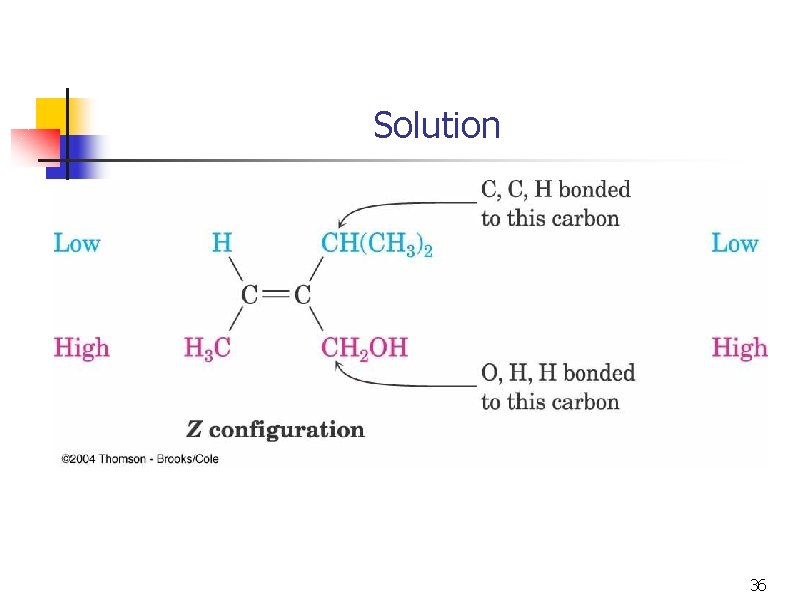
Solution 36

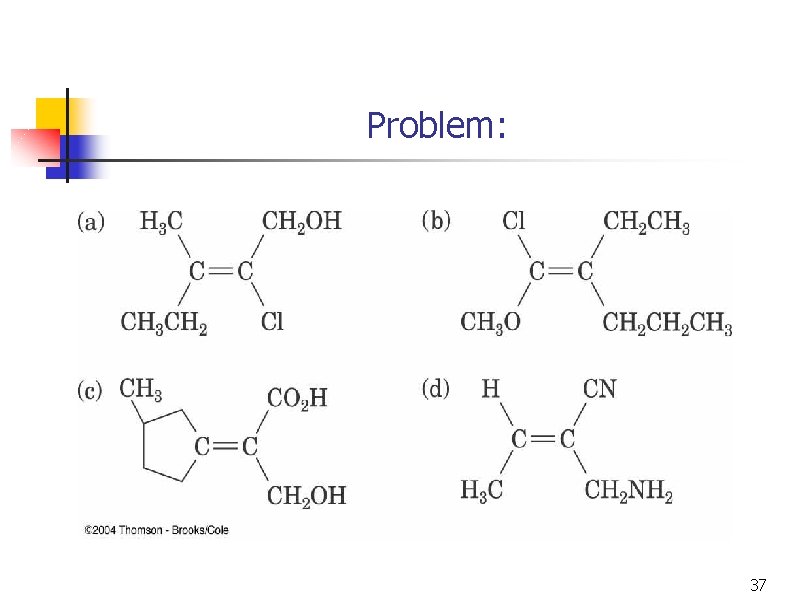
Problem: 37

38

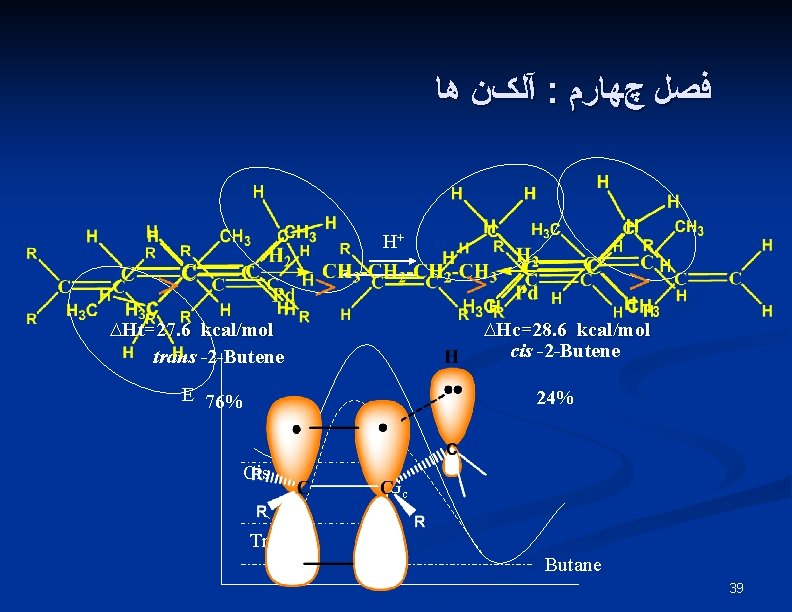
آﻠکﻦ ﻫﺎ : ﻓﺼﻞ چﻬﺎﺭﻡ H+ ∆Ht=27. 6 kcal/mol trans -2 -Butene ∆Hc=28. 6 kcal/mol cis -2 -Butene E 76% 24% Cis Trans ∆Gc ∆Gt Butane 39

Reaction of alkenes: Electrophilic Addition of HX to Alkenes n n n General reaction mechanism: electrophilic addition Attack of electrophile (such as HBr) on bond of alkene produces carbocation and bromide ion Carbocation is itself an electrophile, reacting with nucleophilic bromide ion 40

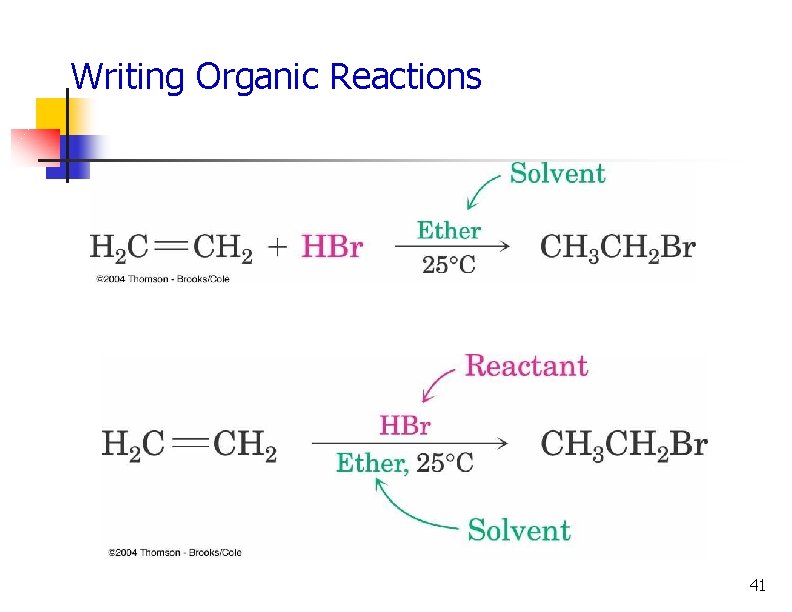
Writing Organic Reactions 41