Enzymes Regulatory enzymes are usually the enzymes that













- Slides: 28

Enzymes �Regulatory enzymes are usually the enzymes that are the rate-limiting, or committed step, in a pathway, meaning that after this step a particular reaction pathway will go to completion �There are five primary forms of enzyme regulation: substrate availability, allosteric, post-translational modification, interaction with control proteins

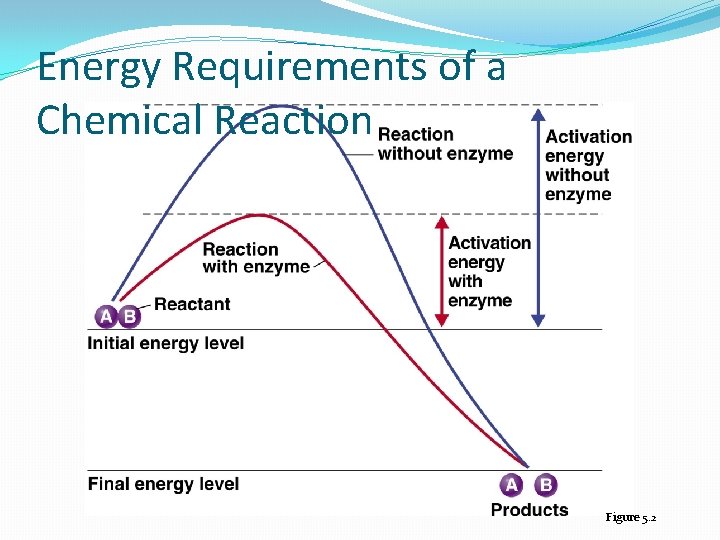
Properties of Enzymes �In general, chemical reactions that release energy can occur without input of energy �The oxidation of glucose releases energy, but the reaction does not occur without an input of energy �Activation energy: the energy required to start such a reaction �Enzymes lower the activation energy so reactions can occur at mild temperatures in living cells

Enzymes �Provide a surface on which reactions take place �Active site: the area on the enzyme surface where the enzyme forms a loose association with the substrate �Substrate: the substance on which the enzyme acts �Enzyme-substrate complex: formed when the substrate molecule collides with the active site of its enzyme �Enzymes generally have a high degree of specificity �Endoenzymes (intracellular)/exoenzymes (extracellular)

Energy Requirements of a Chemical Reaction Figure 5. 2

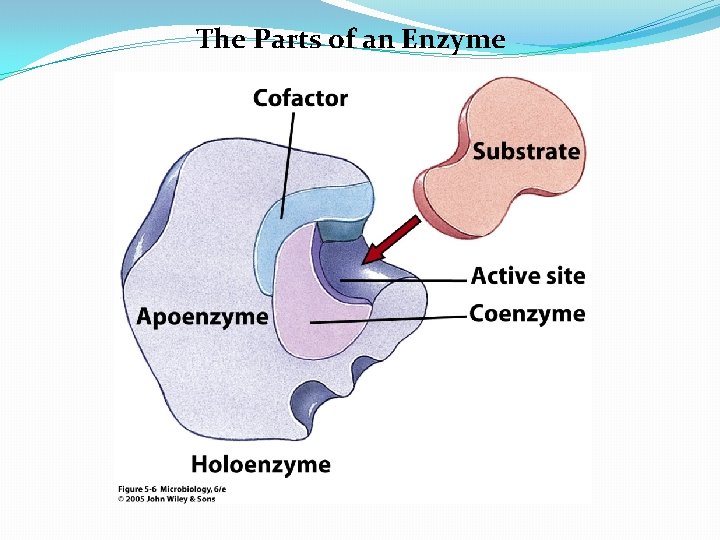
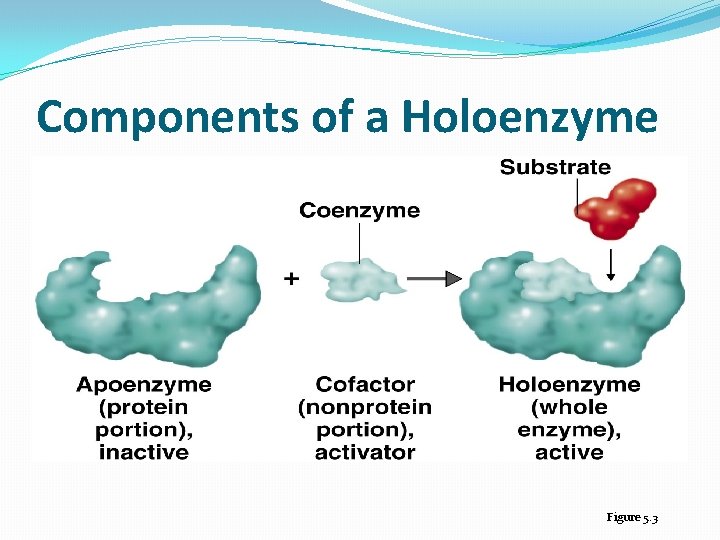
Enzyme Components �Biological catalysts �Specific for a chemical reaction; not used up in that reaction �Apoenzyme: Protein �Cofactor: Nonprotein component �Coenzyme: Organic cofactor �Holoenzyme: Apoenzyme plus cofactor

The Parts of an Enzyme

Properties of Coenzymes and Cofactors �Many enzymes can catalyze a reaction only if substances called coenzymes, or cofactors are present �Apoenzyme: protein portion of such enzymes �Holoenzyme: nonprotein coenzyme or cofactor that is active when combined with apoenzyme �Coenzyme: nonprotein organic molecule bound to or loosely associated with an enzyme �Cofactor: an inorganic ion (e. g. magnesium, zinc) that often improve the fit of an enzyme with its substrate

Components of a Holoenzyme Figure 5. 3

Important Coenzymes �NAD+ �NADP+ �FAD �Coenzyme A

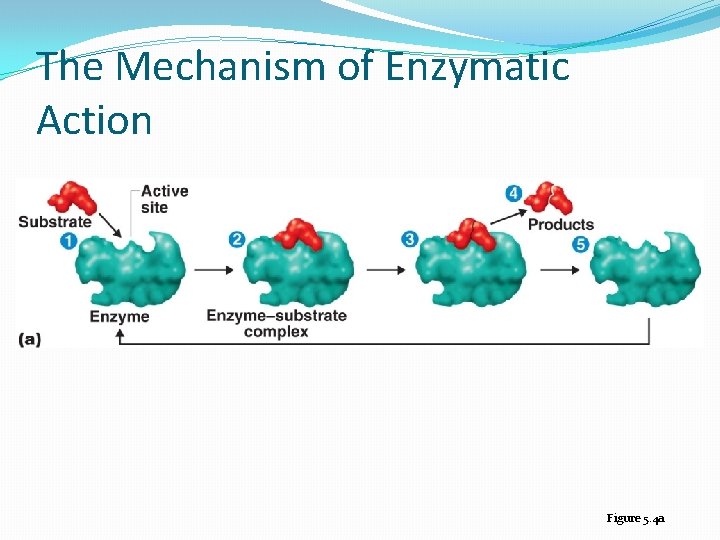
mechanism 1. 2. 3. 4. Substrate binding Formation enzyme substrate complex Production formation and dissociation Enzyme recovery

The Mechanism of Enzymatic Action Figure 5. 4 a

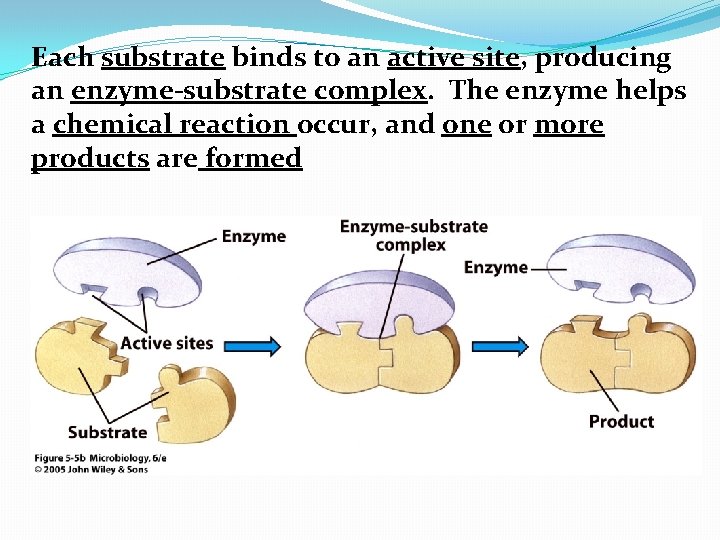
Each substrate binds to an active site, producing an enzyme-substrate complex. The enzyme helps a chemical reaction occur, and one or more products are formed

Enzyme Classification �Oxidoreductase: Oxidation-reduction reactions �Transferase: Transfer functional groups �Hydrolase: Hydrolysis �Lyase: Removal of atoms without hydrolysis �Isomerase: Rearrangement of atoms �Ligase: Joining of molecules, uses ATP

Factors Influencing Enzyme Activity �Temperature �p. H �Substrate concentration �Inhibitors

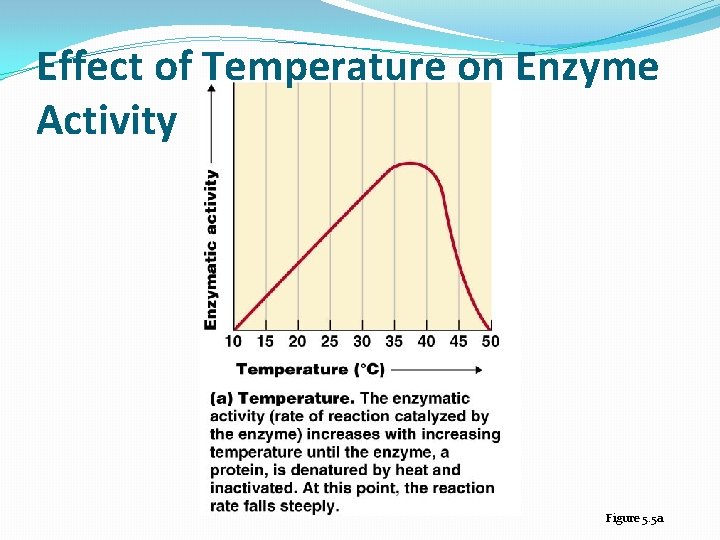
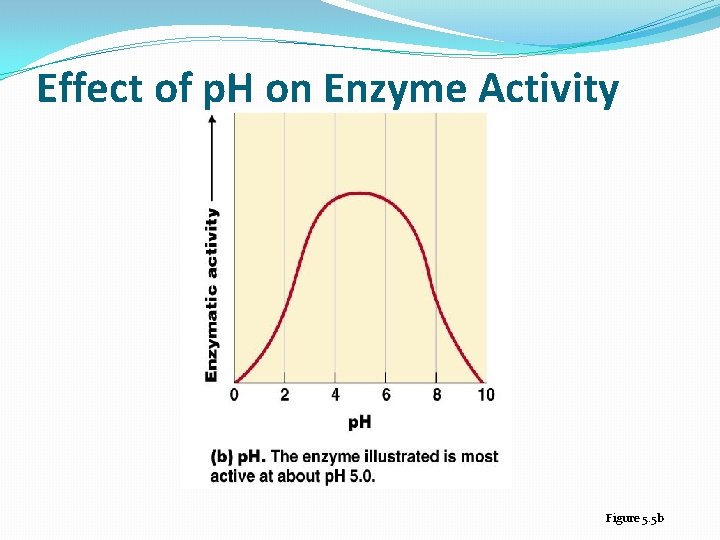
Temperature and p. H �Enzymes are affected by heat and extremes of p. H �Even small p. H changes can alter the electrical charges on various chemical groups in enzyme molecules, thereby altering the enzyme’s ability to bind its substrate and catalyze a reaction �Most enzymes have an optimum temperature, near normal body temperature, and an optimum p. H, near neutral, at which they catalyze a reaction most rapidly �The rate at which an enzyme catalyzes a reaction increases with temperature up to the optimum T

Effect of Temperature on Enzyme Activity Figure 5. 5 a

Effect of p. H on Enzyme Activity Figure 5. 5 b

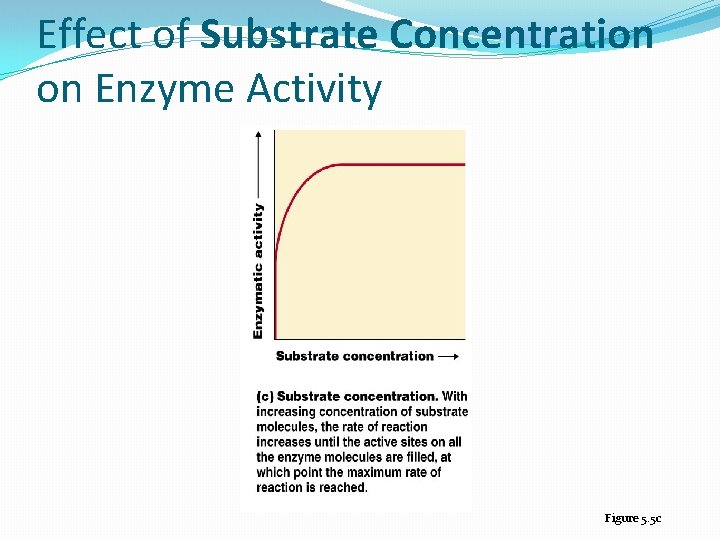
Effect of Substrate Concentration on Enzyme Activity Figure 5. 5 c

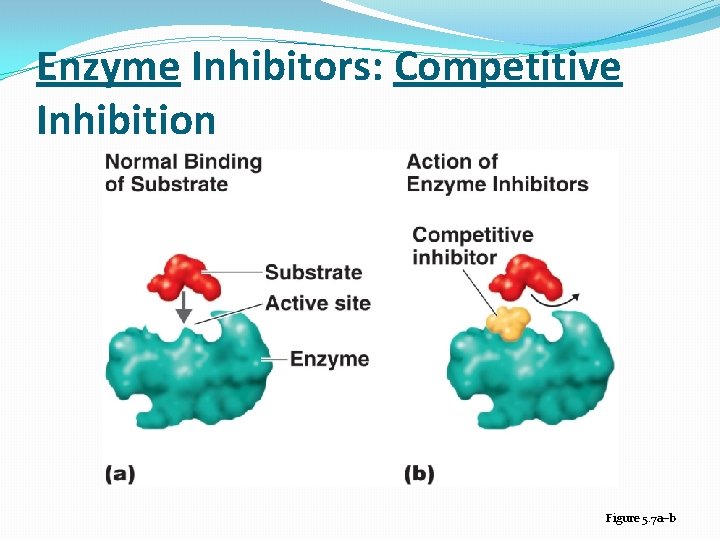
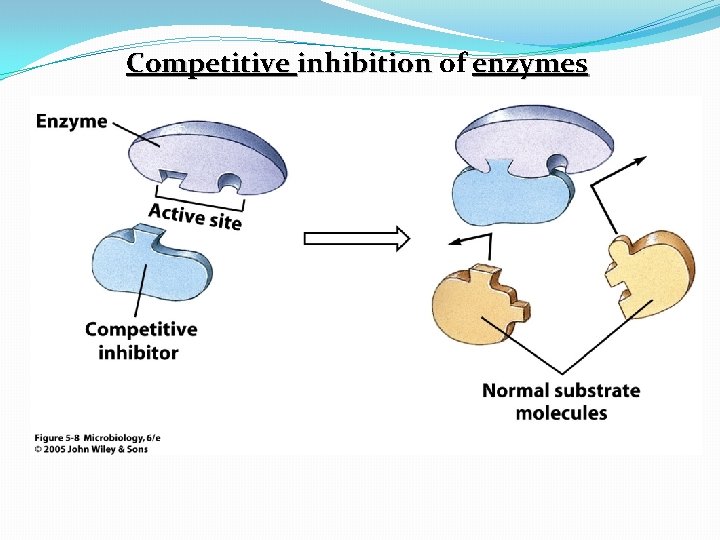
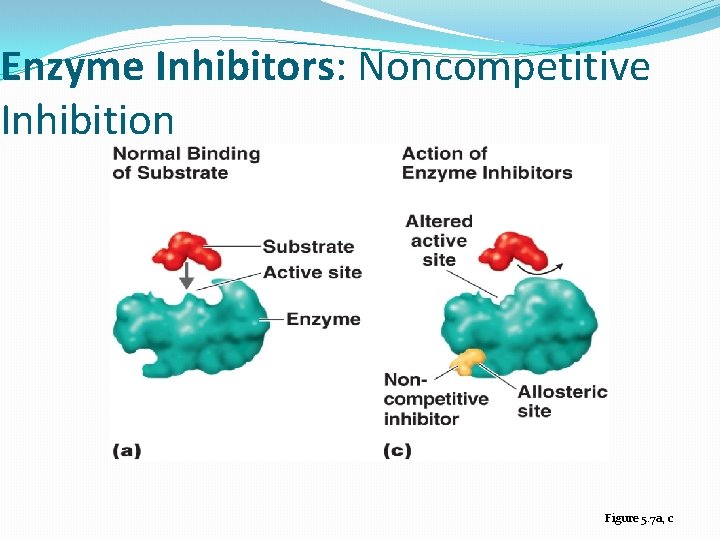
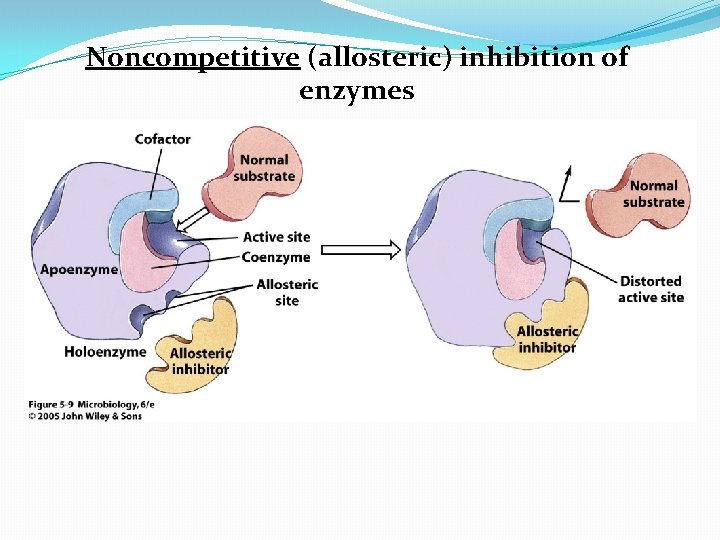
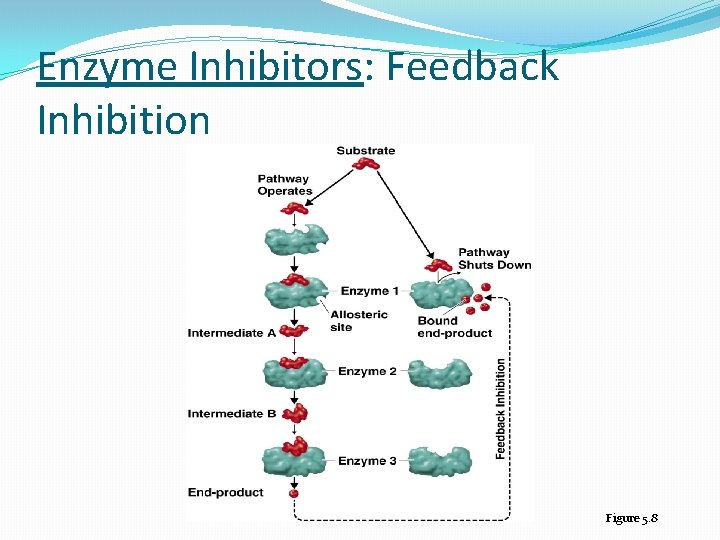
Enzyme Inhibition �Competitive inhibitor: A molecule similar in structure to a substrate can bind to an enzyme’s active site and compete with substrate �Noncompetitive inhibitors: attach to the enzyme at an allosteric site, which is a site other than the active site � noncompetitive inhibitors: distort the tertiary protein structure and alter the shape of the active site �Feedback inhibition: regulates the rate of many metabolic pathways when an end product of a pathway accumulates and binds to and inactivates the first enzyme in the metabolic pathway

Enzyme Inhibitors: Competitive Inhibition Figure 5. 7 a–b

Competitive inhibition of enzymes

Allosteric regulation of enzyme activity � Allosteric regulation = the activation or inhibition of an enzyme’s activity due to binding of an effectors molecule at a regulatory site that is distinct from the active site of the enzyme � Allosteric regulators generally act by increasing or decreasing the enzyme’s affinity for the substrate

Enzyme Inhibitors: Noncompetitive Inhibition Figure 5. 7 a, c

Noncompetitive (allosteric) inhibition of enzymes

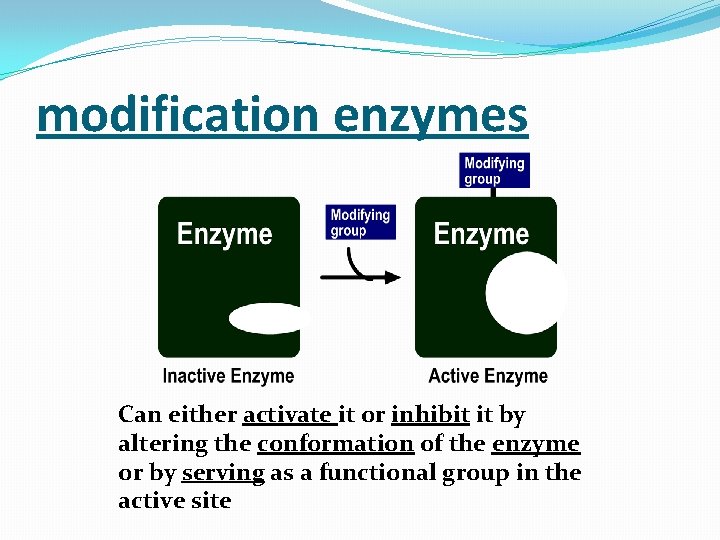
modification enzymes Can either activate it or inhibit it by altering the conformation of the enzyme or by serving as a functional group in the active site

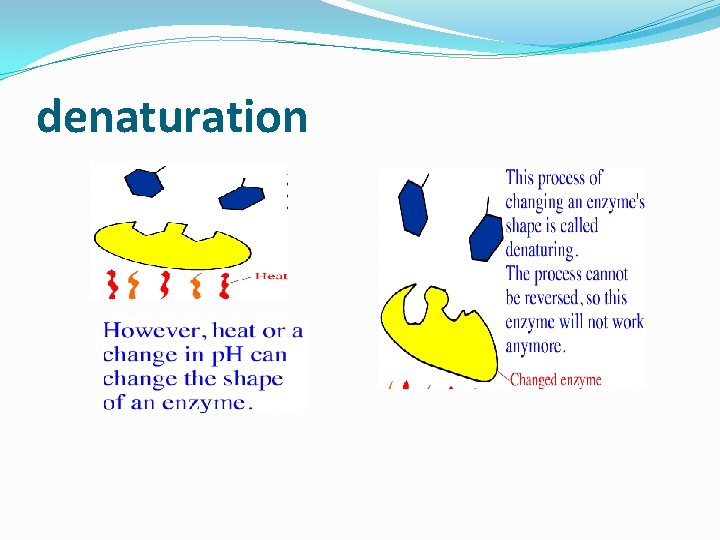
denaturation

denaturation

Enzyme Inhibitors: Feedback Inhibition Figure 5. 8