Chapter 3 Alkena dan Alkuna Nomenklatur dan Reaksinya



















- Slides: 46

Chapter 3. Alkena dan Alkuna: Nomenklatur dan Reaksinya Tutik Dwi Wahyuningsih Jurusan Kimia FMIPA UGM 2011

Alkena dan Alkuna Introduction: kegunaan alkena Struktur alkena Nomenklatur Alkena & Alkuna Nomenklatur E/Z Jenis/tipe ikatan rangkap dua Reaksi pada Alkena Adisi Substitusi Diels Alder Pemutusan 2

Example : a mixture of and bonds, but no triple bonds 3

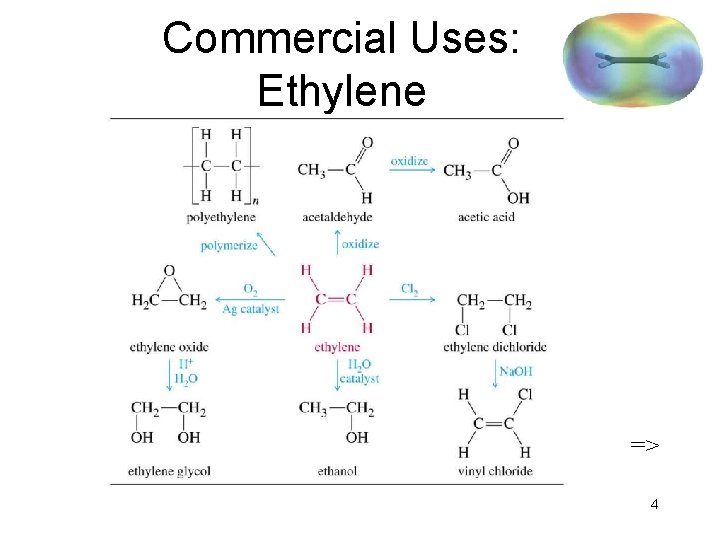
Commercial Uses: Ethylene => 4

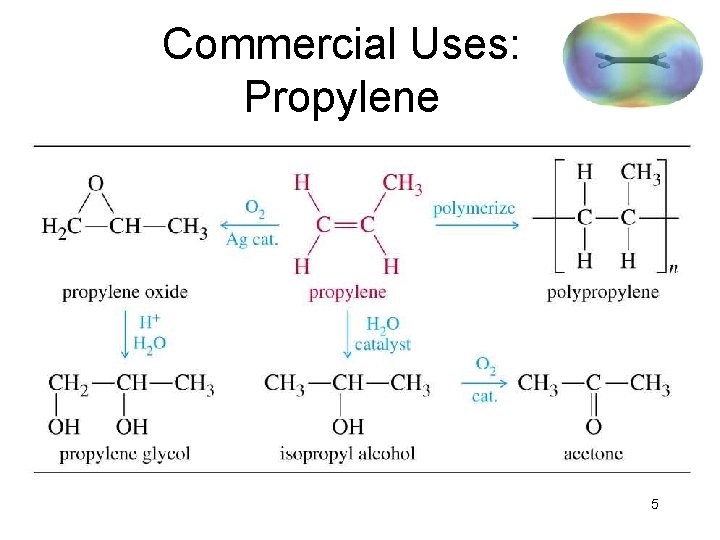
Commercial Uses: Propylene => 5

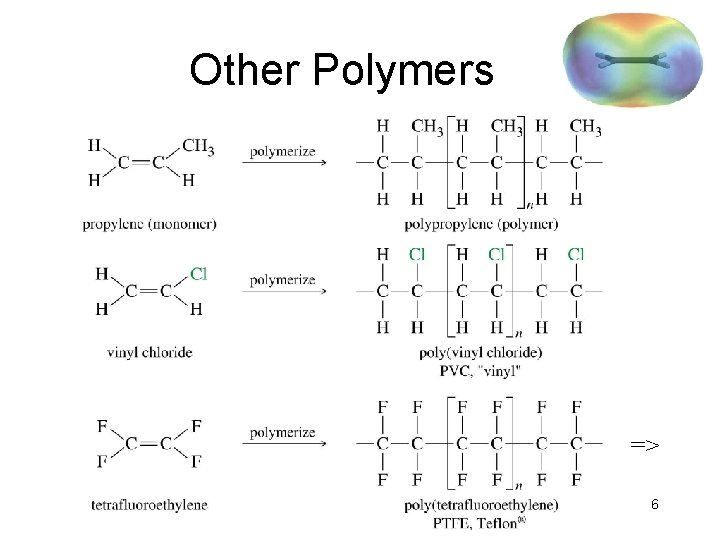
Other Polymers => 6

Industrial Methods • Catalytic cracking of petroleum ØLong-chain alkane is heated with a catalyst to produce an alkene and shorter alkane. ØComplex mixtures are produced. • Dehydrogenation of alkanes ØHydrogen (H 2) is removed with heat, catalyst. ØReaction is endothermic, but entropy-favored. • Neither method is suitable for lab synthesis => 7

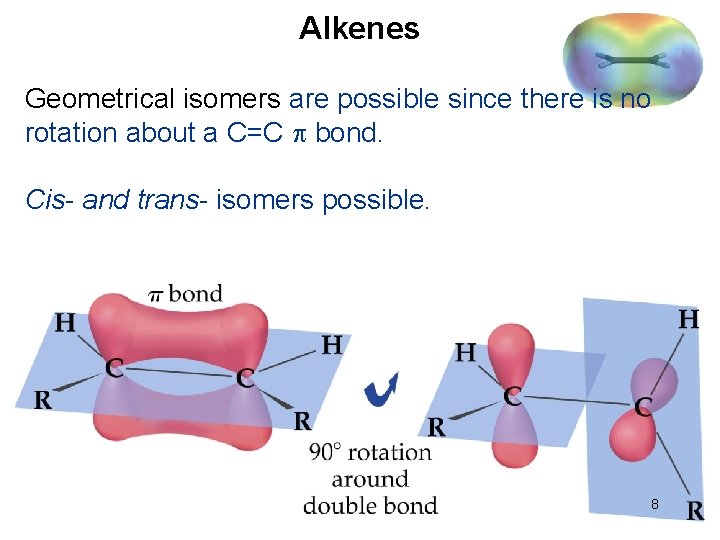
Alkenes Geometrical isomers are possible since there is no rotation about a C=C bond. Cis- and trans- isomers possible. 8

Functional Group • Pi bond is the functional group. • More reactive than sigma bond. 9

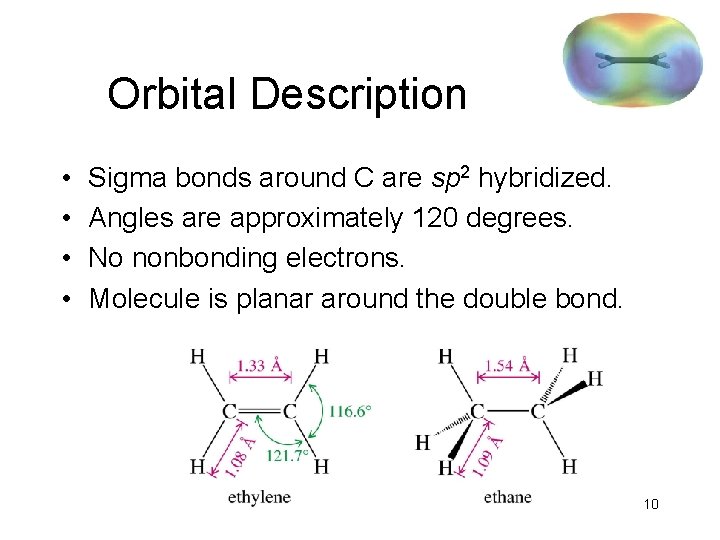
Orbital Description • • Sigma bonds around C are sp 2 hybridized. Angles are approximately 120 degrees. No nonbonding electrons. Molecule is planar around the double bond. 10

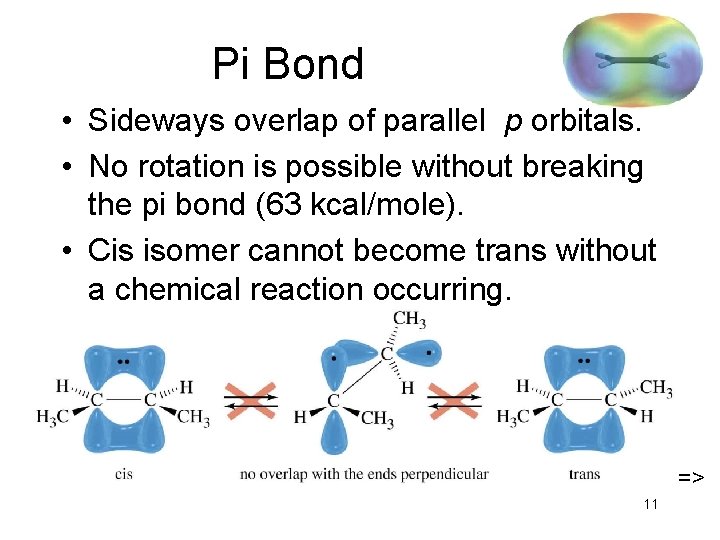
Pi Bond • Sideways overlap of parallel p orbitals. • No rotation is possible without breaking the pi bond (63 kcal/mole). • Cis isomer cannot become trans without a chemical reaction occurring. => 11

IUPAC Nomenclature • Parent is longest chain containing the double bond. • -ane changes to -ene. (or -diene, -triene) • Number the chain so that the double bond has the lowest possible number. • In a ring, the double bond is assumed to be between carbon 1 and carbon 2. => 12

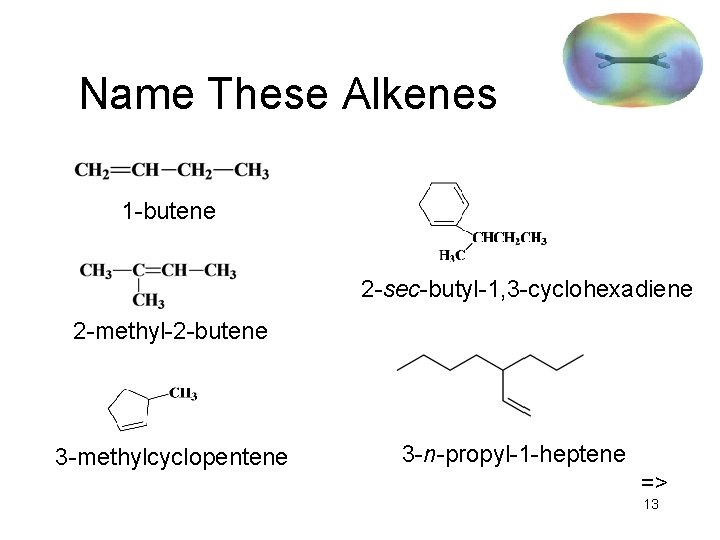
Name These Alkenes 1 -butene 2 -sec-butyl-1, 3 -cyclohexadiene 2 -methyl-2 -butene 3 -methylcyclopentene 3 -n-propyl-1 -heptene => 13

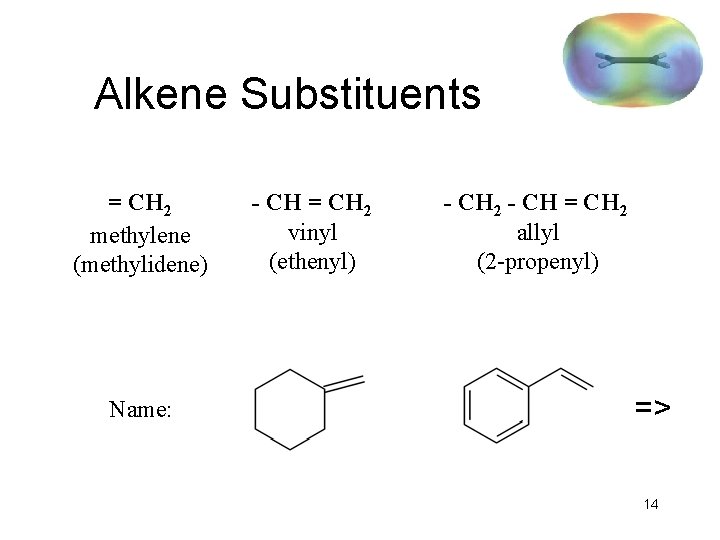
Alkene Substituents = CH 2 methylene (methylidene) Name: - CH = CH 2 vinyl (ethenyl) - CH 2 - CH = CH 2 allyl (2 -propenyl) => 14

Common Names • Usually used for small molecules. • Examples: => 15

Cis-trans Isomerism • Similar groups on same side of double bond, alkene is cis. • Similar groups on opposite sides of double bond, alkene is trans. • Cycloalkenes are assumed to be cis. • Trans cycloalkenes are not stable unless the ring has at least 8 carbons. => 16

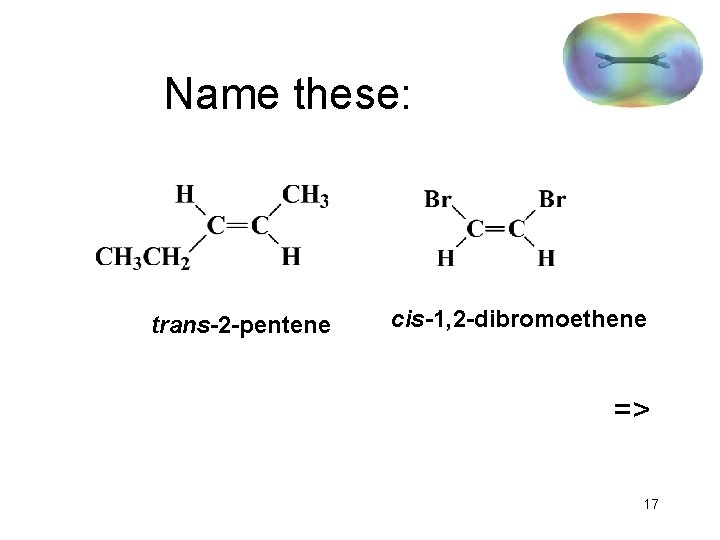
Name these: trans-2 -pentene cis-1, 2 -dibromoethene => 17

E-Z Nomenclature • Use the Cahn-Ingold-Prelog rules to assign priorities to groups attached to each carbon in the double bond. • If high priority groups are on the same side, the name is Z (for zusammen). • If high priority groups are on opposite sides, the name is E (for entgegen). => 18

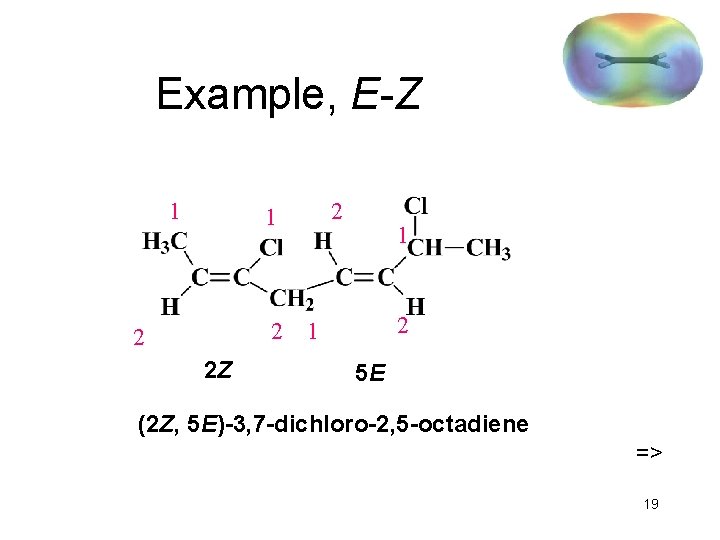
Example, E-Z 1 2 2 2 Z 1 2 1 5 E (2 Z, 5 E)-3, 7 -dichloro-2, 5 -octadiene => 19

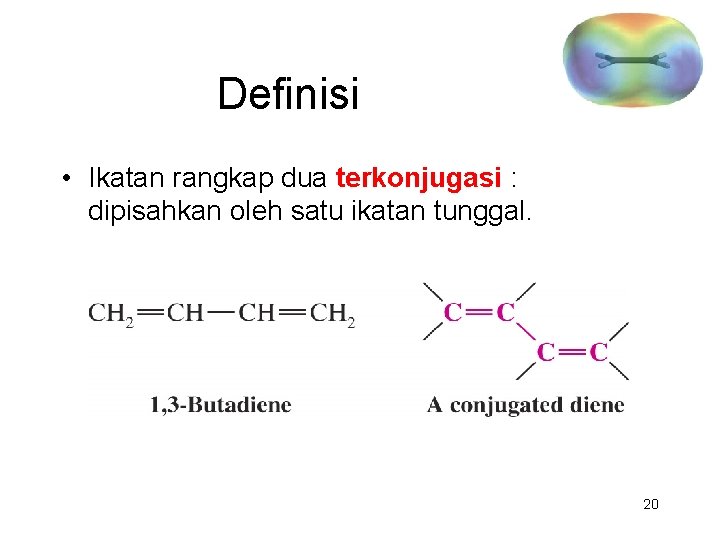
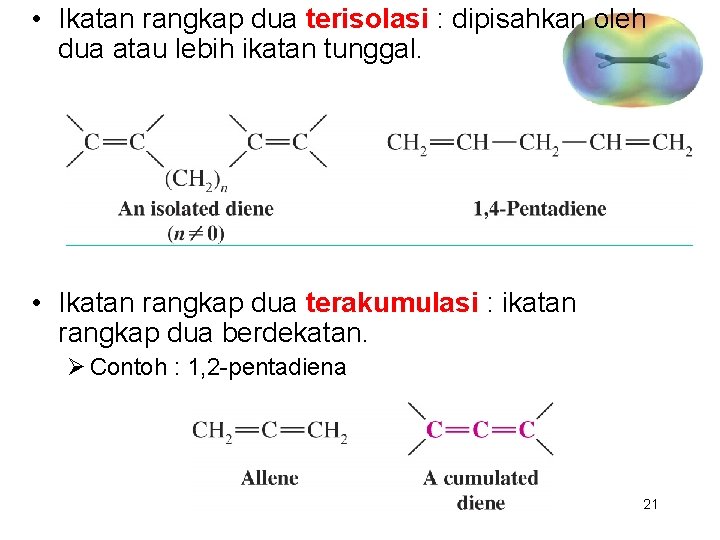
Definisi • Ikatan rangkap dua terkonjugasi : dipisahkan oleh satu ikatan tunggal. 20

• Ikatan rangkap dua terisolasi : dipisahkan oleh dua atau lebih ikatan tunggal. • Ikatan rangkap dua terakumulasi : ikatan rangkap dua berdekatan. Ø Contoh : 1, 2 -pentadiena 21

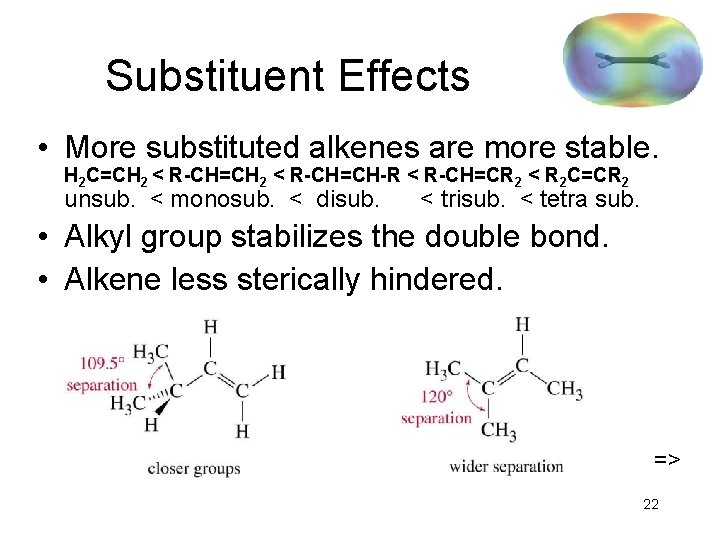
Substituent Effects • More substituted alkenes are more stable. H 2 C=CH 2 < R-CH=CH-R < R-CH=CR 2 < R 2 C=CR 2 unsub. < monosub. < disub. < trisub. < tetra sub. • Alkyl group stabilizes the double bond. • Alkene less sterically hindered. => 22

Alkenes 23

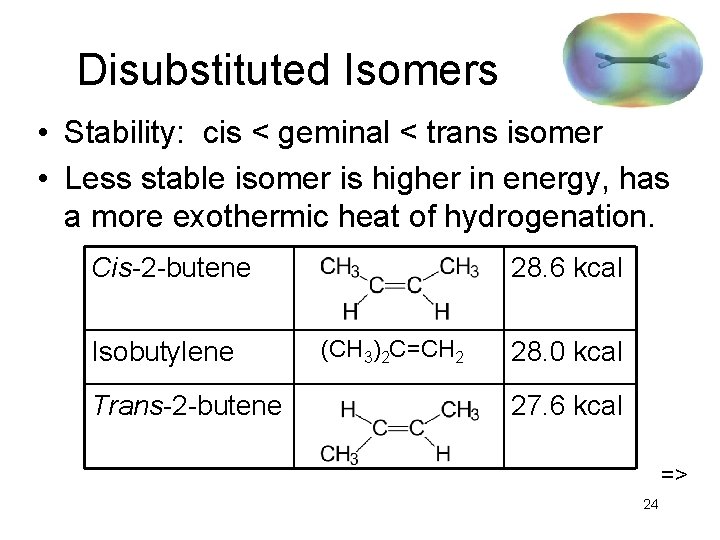
Disubstituted Isomers • Stability: cis < geminal < trans isomer • Less stable isomer is higher in energy, has a more exothermic heat of hydrogenation. Cis-2 -butene Isobutylene Trans-2 -butene 28. 6 kcal (CH 3)2 C=CH 2 28. 0 kcal 27. 6 kcal => 24

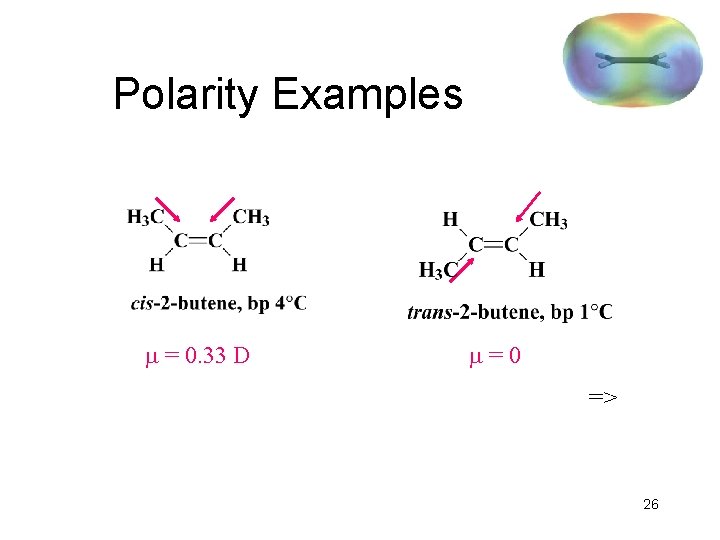
Physical Properties • • Low boiling points, increasing with mass. Branched alkenes have lower boiling points. Less dense than water. Slightly polar Ø Pi bond is polarizable, so instantaneous dipole interactions occur. Ø Alkyl groups are electron-donating toward the pi bond, so may have a small dipole moment. => 25

Polarity Examples = 0. 33 D =0 => 26

ADDITION REACTION An addition reaction is one in which the two reactants add together to make the product A + B AB with no other pieces lost or left over. 27

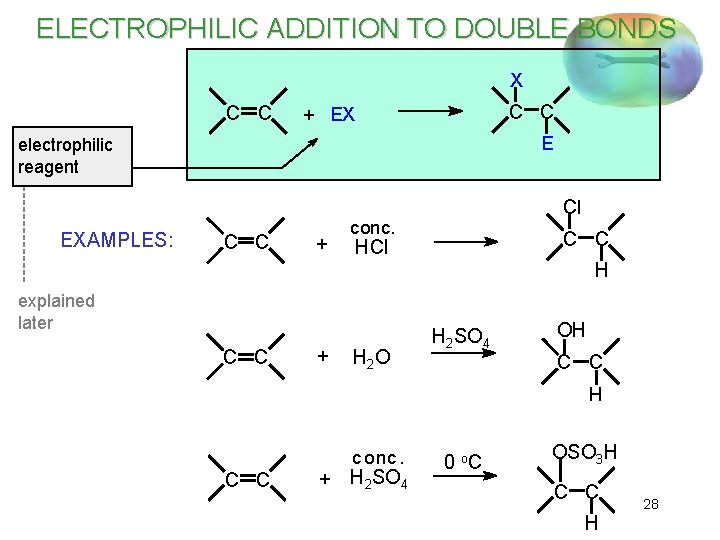
ELECTROPHILIC ADDITION TO DOUBLE BONDS X C C + EX E electrophilic reagent EXAMPLES: C C + Cl conc. C C HCl H explained later C C + H 2 O H 2 SO 4 OH C C H c onc. C C + H 2 SO 4 0 o. C OSO 3 H C C H 28

Addition Reactions of Alkenes and Alkynes A common addition reaction is hydrogenation: CH 3 CH=CHCH 3 + H 2 CH 3 CH 2 CH 3 Hydrogenation requires high temperatures and pressures as well as the presence of a catalyst (e. g. Ni). Note: hydrogenation forms alkanes from alkenes. 29

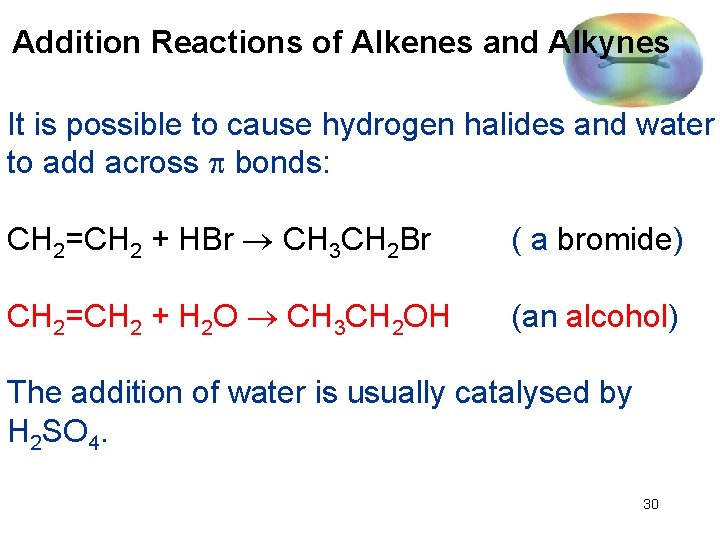
Addition Reactions of Alkenes and Alkynes It is possible to cause hydrogen halides and water to add across bonds: CH 2=CH 2 + HBr CH 3 CH 2 Br ( a bromide) CH 2=CH 2 + H 2 O CH 3 CH 2 OH (an alcohol) The addition of water is usually catalysed by H 2 SO 4. 30

Addition Reactions of Alkenes and Alkynes The most dominant reaction for alkenes and alkynes involves the addition of something to the two atoms which form the double bond: Note that the C-C bond has been replaced by two C-Br bonds. 31

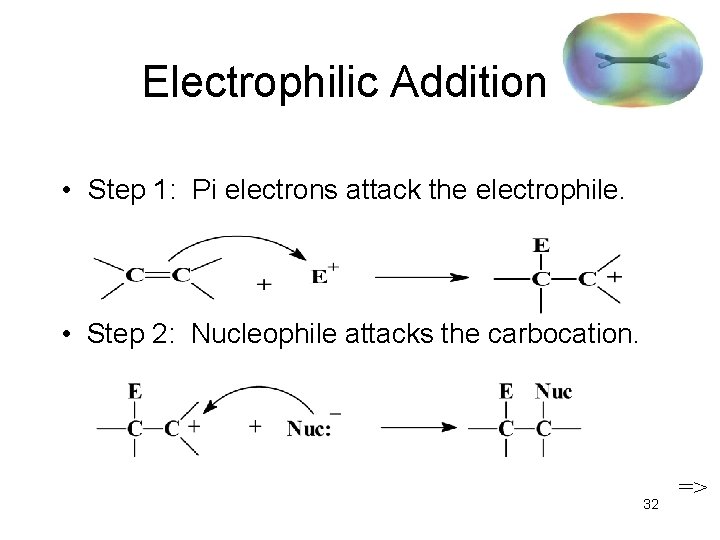
Electrophilic Addition • Step 1: Pi electrons attack the electrophile. • Step 2: Nucleophile attacks the carbocation. 32 =>

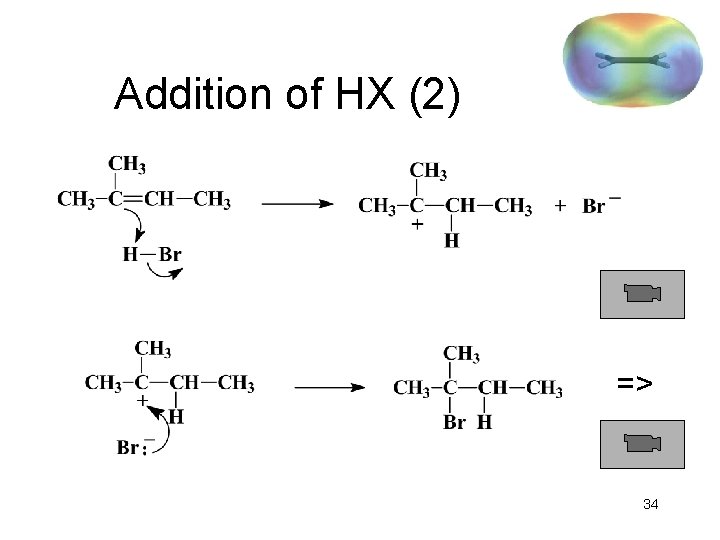
Addition of HX (1) Protonation of double bond yields the most stable carbocation. Positive charge goes to the carbon that was not protonated. X => 33

Addition of HX (2) => 34

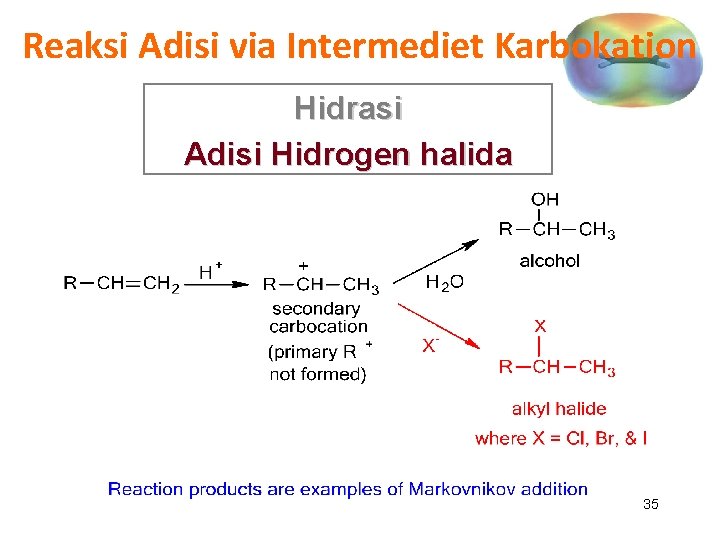
Reaksi Adisi via Intermediet Karbokation Hidrasi Adisi Hidrogen halida 35

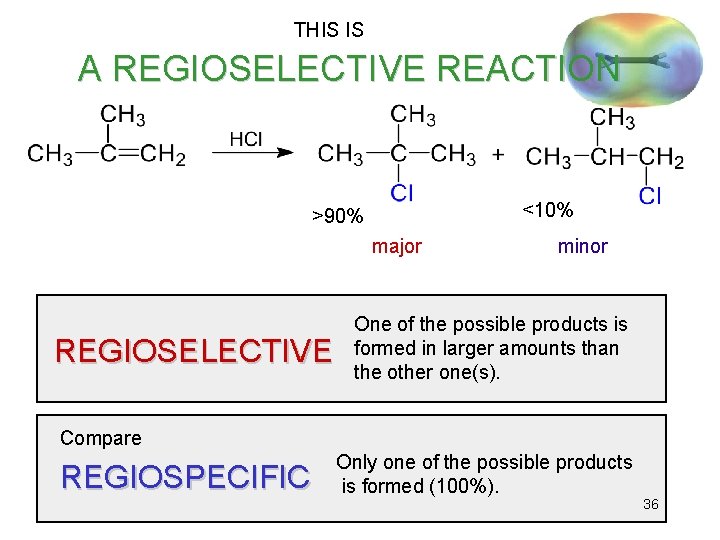
THIS IS A REGIOSELECTIVE REACTION <10% >90% major REGIOSELECTIVE minor One of the possible products is formed in larger amounts than the other one(s). Compare REGIOSPECIFIC Only one of the possible products is formed (100%). 36

Regiospecificity • Markovnikov’s Rule: The proton of an acid adds to the carbon in the double bond that already has the most H’s. “Rich get richer. ” • More general Markovnikov’s Rule: In an electrophilic addition to an alkene, the electrophile adds in such a way as to form the most stable intermediate. • HCl, HBr, and HI add to alkenes to form Markovnikov products. => 37

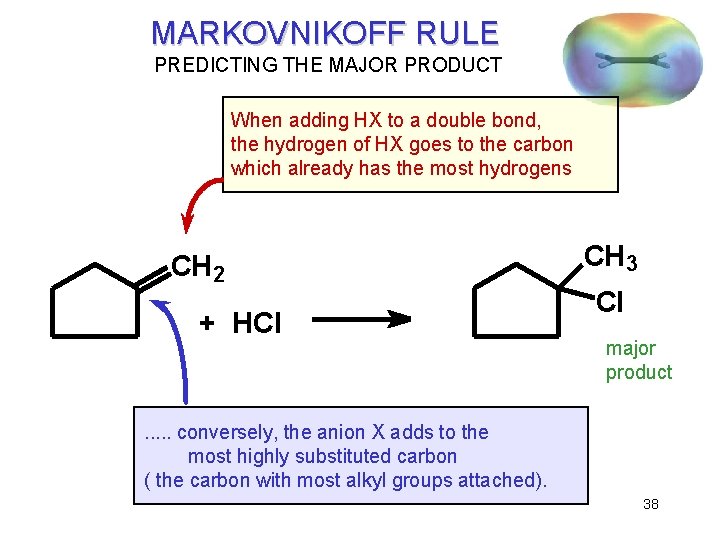
MARKOVNIKOFF RULE PREDICTING THE MAJOR PRODUCT When adding HX to a double bond, the hydrogen of HX goes to the carbon which already has the most hydrogens CH 2 + HCl CH 3 Cl major product . . . conversely, the anion X adds to the most highly substituted carbon ( the carbon with most alkyl groups attached). 38

AN “EMPIRICAL” RULE Markovnikoff formulated his rule by observing the results of hundreds of reactions that he performed. EMPIRICAL = DETERMINED BY OBSERVATION He had no idea why the reaction worked this way, only that as a general rule it did give the stated result. 39

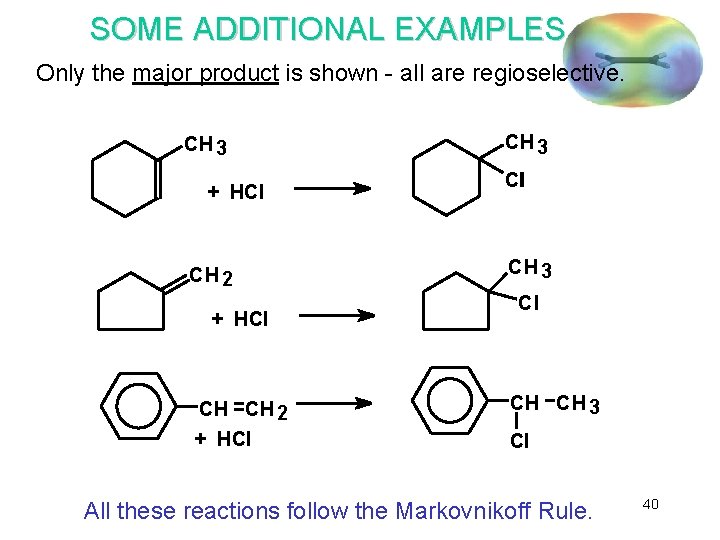
SOME ADDITIONAL EXAMPLES Only the major product is shown - all are regioselective. CH 3 + HCl CH 2 + HCl CH 3 Cll CH 3 Cl CH CH 3 Cl All these reactions follow the Markovnikoff Rule. 40

MARKOVNIKOFF RULE ANOTHER WAY TO STATE THE RULE When the reaction forms the carbocation intermediate, the most highly substituted carbocation is favored : tertiary > secondary > primary. least favored methyl carbocation primary carbocation secondary carbocation most favored (lowest energy) + CH 3 R R tertiary carbocation CH 2 + CH R + R R C + R 41

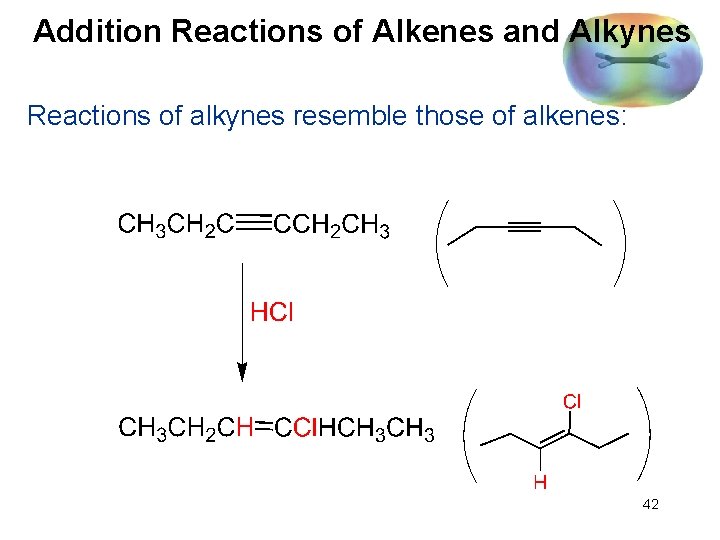
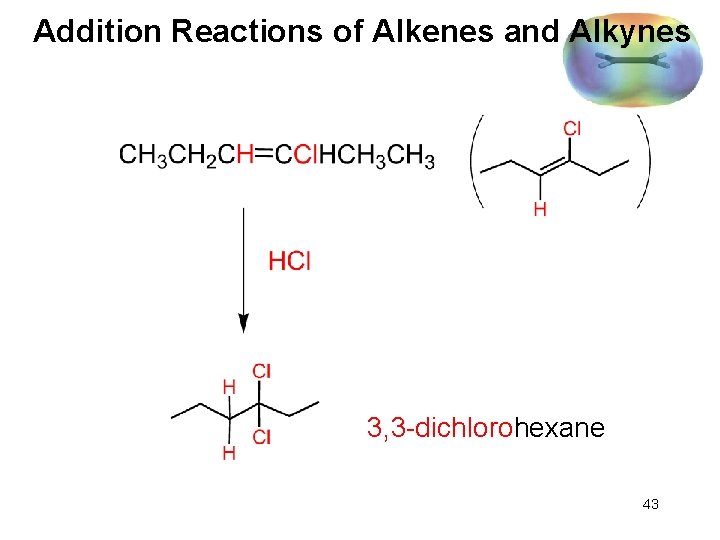
Addition Reactions of Alkenes and Alkynes Reactions of alkynes resemble those of alkenes: 42

Addition Reactions of Alkenes and Alkynes 3, 3 -dichlorohexane 43

Alkene Synthesis Overview • • E 2 dehydrohalogenation (-HX) E 1 dehydrohalogenation (-HX) Dehalogenation of vicinal dibromides (-X 2) Dehydration of alcohols (-H 2 O) => 44

Dehydration of Alcohols • Reversible reaction • Use concentrated sulfuric or phosphoric acid, remove low-boiling alkene as it forms. • Protonation of OH converts it to a good leaving group, HOH • Carbocation intermediate, like E 1 • Protic solvent removes adjacent H+ =>45

End of Chapter 3 46