Chapter 7 Ionic and Metallic Bonding Chemistry January

Chapter 7 “Ionic and Metallic Bonding” Chemistry January 14, 2011

Bellringer #55 (Jan 14 th, 2011) l Based on what you read for your homework: – What is the rule that atoms tend to follow when forming ions? – How can you determine the number of valence electrons in an atom of a representative element?

Agenda (Jan 14 th, 2011) Answer Bellringer #55 l Check Cornell notes 7. 1 l Write OBJECTIVES for section 7. 1 l Notes/Lecture 7. 1 l Answer “Pair to Pair” questions l Hmwk: Section 7. 1 assessment and 7. 1 Wkst l

Section 7. 1 - Ions l OBJECTIVES: –Determine the number of valence electrons in an atom of a representative element.

Section 7. 1 - Ions l OBJECTIVES: –Explain how the octet rule applies to atoms of metallic and nonmetallic elements.

Section 7. 1 - Ions l OBJECTIVES: –Describe how cations form.

Section 7. 1 - Ions l OBJECTIVES: –Explain how anions form.

Valence Electrons are…? l The electrons responsible for the chemical properties of atoms, and are those in the outer energy level. l Valence electrons - The s and p electrons in the outer energy level – the highest occupied energy level l Core electrons – are those in the energy levels below.

Keeping Track of Electrons l l l Atoms in the same column. . . 1) Have the same outer electron configuration. 2) Have the same valence electrons. The number of valence electrons are easily determined. It is the group number for a representative element Group 2 A: Be, Mg, Ca, etc. – have 2 valence electrons

Pair to Pair Question #1 l Determine how many valence electrons are in the following atoms: – Rubidium (Rb) – Barium (Ba) – Aluminum (Al) – Fluorine (F) – Argon (Ar) – Helium (He) – Nitrogen (N) – Sulfur (S)

Electron Dot diagrams are… l l l A way of showing & keeping track of valence electrons. How to write them? Write the symbol - it represents the nucleus and inner (core) electrons Put one dot for each valence electron (8 maximum) They don’t pair up until they have to…. . what rule does this sound like? Hund’s Rule X

The Electron Dot diagram for Nitrogen has 5 valence electrons to show. l First we write the symbol. l. Then add 1 electron at a time to each side. l. Now they are forced to pair up. l. We have now written the electron dot diagram for Nitrogen. l N

The Octet Rule In Chapter 6, we learned that noble gases are unreactive in chemical reactions l The Octet Rule: in forming compounds, atoms tend to achieve a noble gas configuration; 8 in the outer level is stable l Each noble gas has 8 electrons, except for one…. . which one is it? ? ? ? l Helium is an exception in Group 8 A because it only has 2 electrons l

Pair to Pair Question #2 l Draw the ELECTRON DOT DIAGRAM for the following atoms: – Aluminum – Silicon – Nitrogen – Sulfur – Fluorine – Argon – Barium – Rubidium

Formation of Cations l Metals lose electrons to attain a noble gas configuration. l They make positive ions (cations) l If we look at the electron configuration, it makes sense to lose electrons: l Na 1 s 22 p 63 s 1 1 valence electron l Na 1+ 1 s 22 p 6 This is a noble gas configuration with 8 electrons in the outer level.

Electron Dots For Cations l Metals will have few valence electrons (usually 3 or less); calcium has only 2 valence electrons Ca

Electron Dots For Cations Metals will have few valence electrons l Metals will lose the valence electrons l Ca

Electron Dots For Cations Metals will have few valence electrons l Metals will lose the valence electrons l Forming positive ions l 2+ Ca This is named the “calcium ion”. NO DOTS are now shown for the cation.

Electron Dots For Cations l Let’s do Beryllium, #4 l The electron configuration is: 2 2 1 s 2 s l Thus, it can lose 2 e- (making it 2+), to be like Helium Be = 2+ Be Beryllium ion

Pair to Pair Question #3 l Draw the ELECTRON DOT STRUCTURES for the following CATIONS: – Calcium (Ca) – Sodium (Na) – Gallium (Ga)

Electron Configurations: Anions l Nonmetals gain electrons to attain noble gas configuration. l They make negative ions (anions) l S = 1 s 22 p 63 s 23 p 4 = 6 valence electrons l S 2 - = 1 s 22 p 63 s 23 p 6 = noble gas configuration. l Halide ions are ions from chlorine or other halogens that gain electrons

Electron Dots For Anions Nonmetals will have many valence electrons (usually 5 or more) l They will gain electrons to fill outer shell. l P 3 - (This is called the “phosphide ion”, and should show dots)

Electron Dots For Anions l Let’s do Chlorine, #17 l The electron configuration is: 2 2 6 2 5 1 s 2 s 2 p 3 s 3 p l Thus, it can gain 1 e- (making it 1 -), to be like Argon Cl = Cl 1 Chloride ion

Pair to Pair Question #4 l Draw ELECTRON DOT STRUCTURES for the following ANIONS: – Phosphorous (P) – Bromine (Br) – Sulfur (S)

Stable Electron Configurations All atoms react to try and achieve a noble gas configuration. l Noble gases have 2 s and 6 p electrons. l 8 valence electrons = already stable! l This is the octet rule (8 in the outer level is particularly stable). l Ar

Bellringer #56 (Jan 18 th, 2011) l Write the ELECTRON DOT DIAGRAMS of the NEUTRAL atom AND ION for the following elements: – Bromine – Boron – Strontium ****each element should have TWO electron dot diagrams

Agenda Objective: Review for Semester Exam l Bellringer #56 l Homework Check l Results of Semester Exam: 1 st attempt l Semester Exam Study Guide l Science Fair Make-Up Update l Homework: Cornell Notes 7. 2 l Semester Exam 2 nd Attempt: Tomorrow l

Bellringer #57 (Jan 20 th, 2011) l According to Section 7. 2 – What is an IONIC COMPOUND? – What are three properties of ionic compounds?

Agenda (Jan 20 th, 2011) Bellringer #57 l Homework Check: 7. 2 Cornell Notes l Write Section 7. 2 Objectives l 7. 2 Notes/Lecture l Peer-to-Peer Questions (turn in at end of class) l Homework: 7. 2 Section Assessment (#14 -22) l

Section 7. 2 Ionic Bonds and Ionic Compounds l OBJECTIVES: –Explain the electrical charge of an ionic compound.

Section 7. 2 Ionic Bonds and Ionic Compounds l OBJECTIVES: –Describe three properties of ionic compounds.

Ionic Compounds 1) Also called SALTS 2) Made from: a CATION with an ANION (or literally from a metal combining with a nonmetal)

l Anions Ionic Bonding and cations are held together by opposite charges (+ and -) compounds are called salts. l Simplest ratio of elements in an ionic compound is called the formula unit. l The bond is formed through the transfer of electrons (lose and gain) l Electrons are transferred to achieve noble gas configuration. l Ionic

Ionic Bonding Example Na Cl The metal (sodium) tends to lose its one electron from the outer level. The nonmetal (chlorine) needs to gain one more to fill its outer level, and will accept the one electron that sodium is going to lose.

Ionic Bonding Example + Na Cl - Note: Remember that NO DOTS are now shown for the cation!

Ionic Bonding Example Lets do an example by combining calcium and phosphorus: Ca l P All the electrons must be accounted for, and each atom will have a noble gas configuration (which is stable).
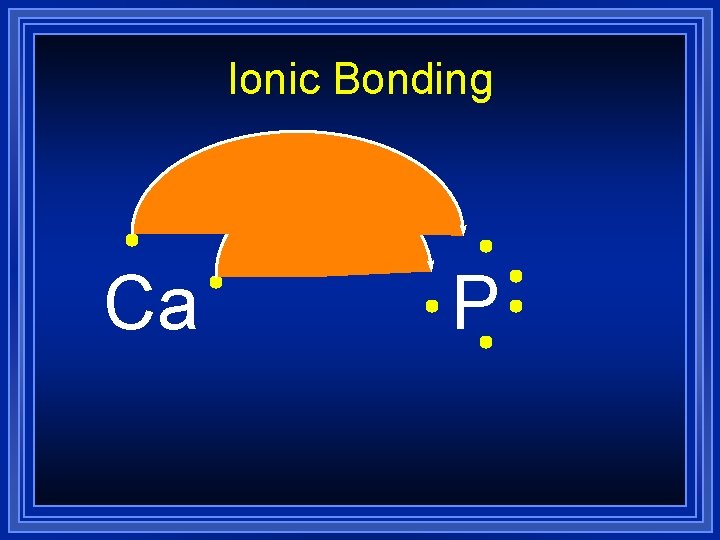
Ionic Bonding Ca P
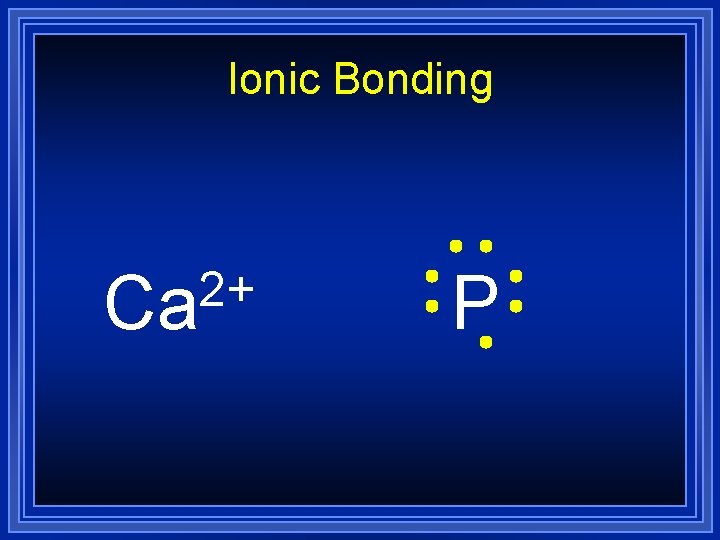
Ionic Bonding 2+ Ca P
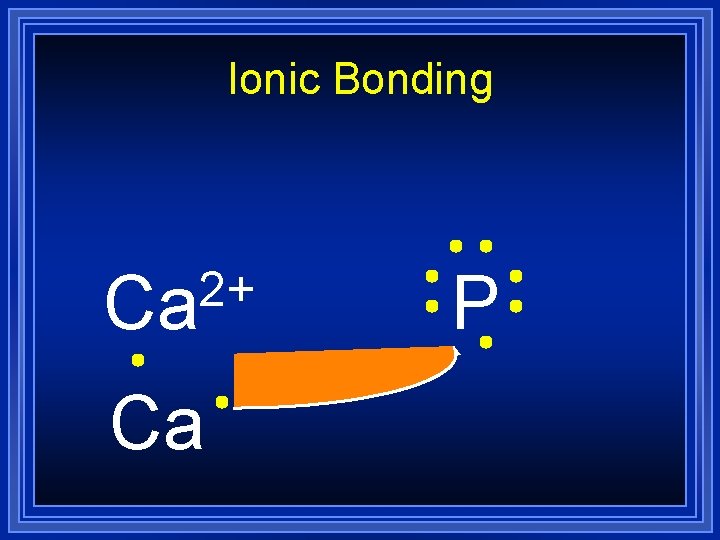
Ionic Bonding 2+ Ca Ca P
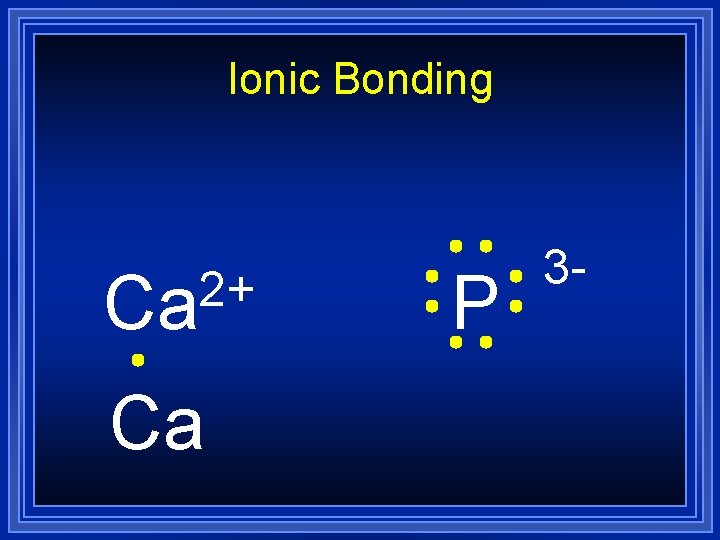
Ionic Bonding 2+ Ca Ca P 3 -
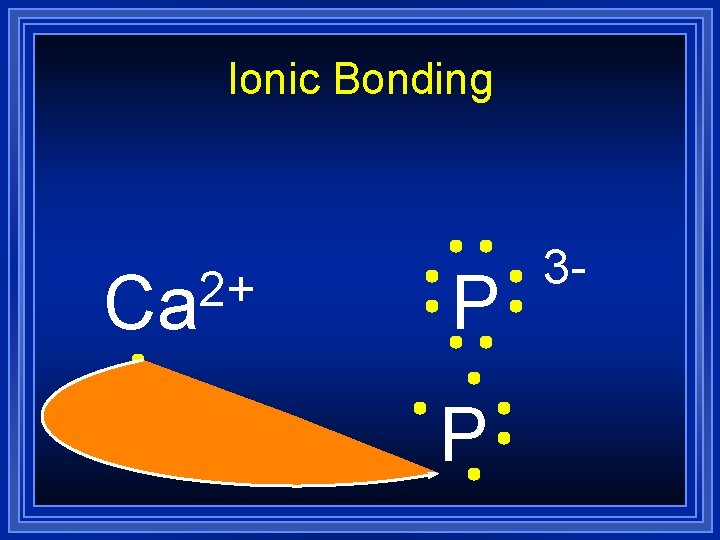
Ionic Bonding 2+ Ca P 3 -
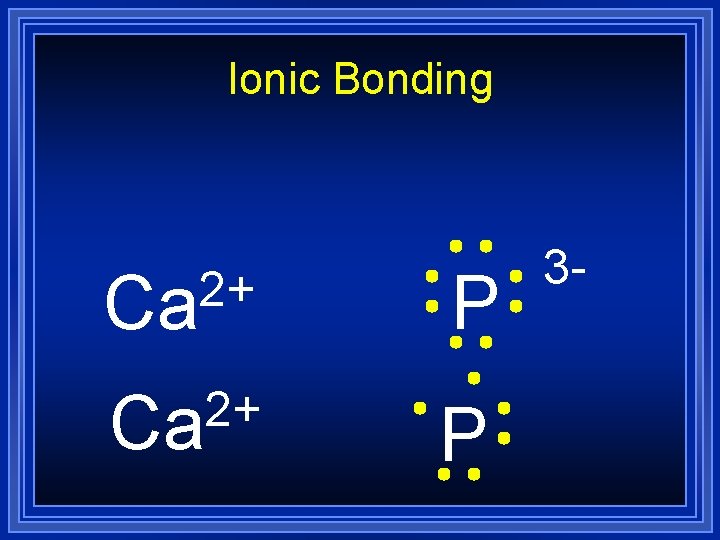
Ionic Bonding 2+ Ca P 3 -
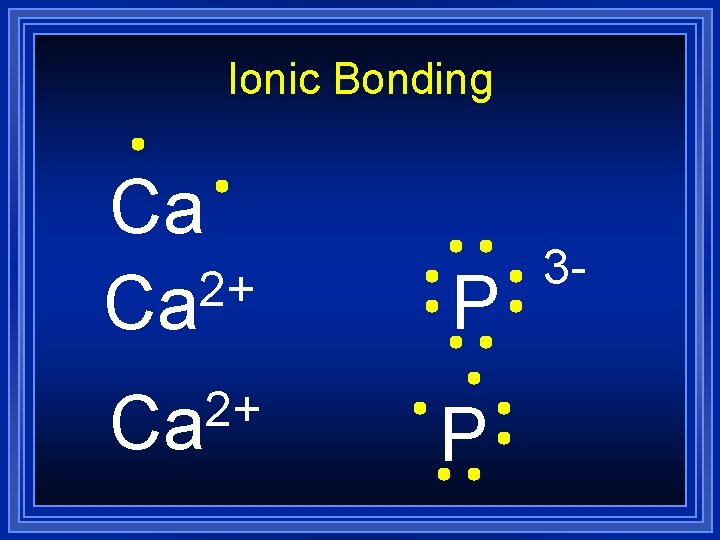
Ionic Bonding Ca 2+ Ca P 3 -
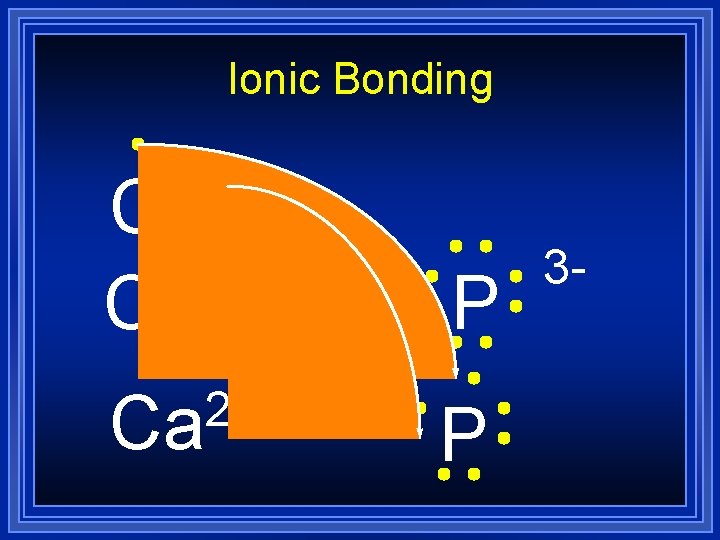
Ionic Bonding Ca 2+ Ca P 3 -
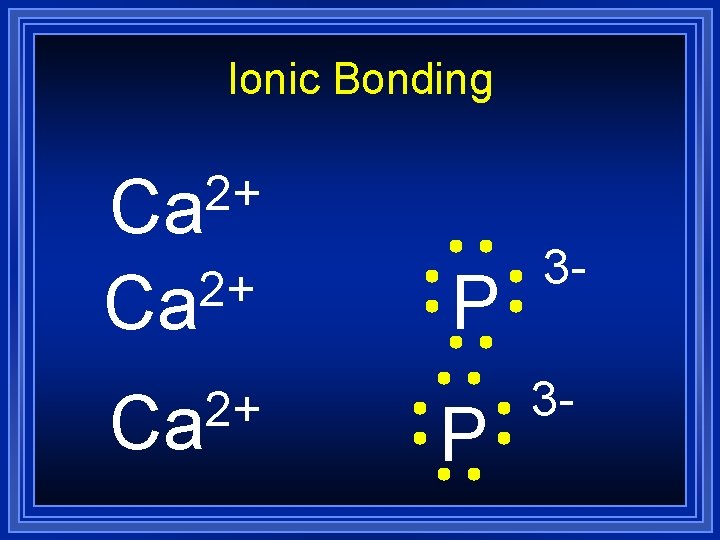
Ionic Bonding 2+ Ca P 3 -
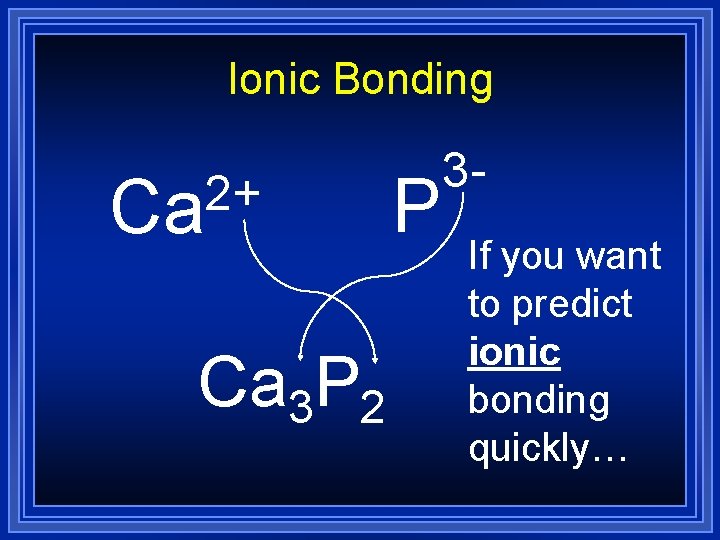
Ionic Bonding 2+ Ca Ca 3 P 2 P 3 If you want to predict ionic bonding quickly…

Ionic Bonding = Ca 3 P 2 Formula Unit This is a chemical formula, which shows the kinds and numbers of atoms in the smallest representative particle of the substance. For an ionic compound, the smallest representative particle is called a: Formula Unit
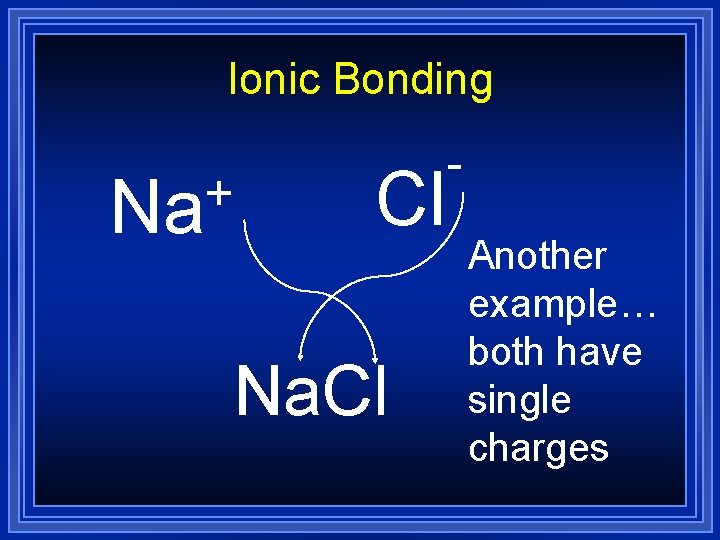
Ionic Bonding + Na Cl Na. Cl Another example… both have single charges
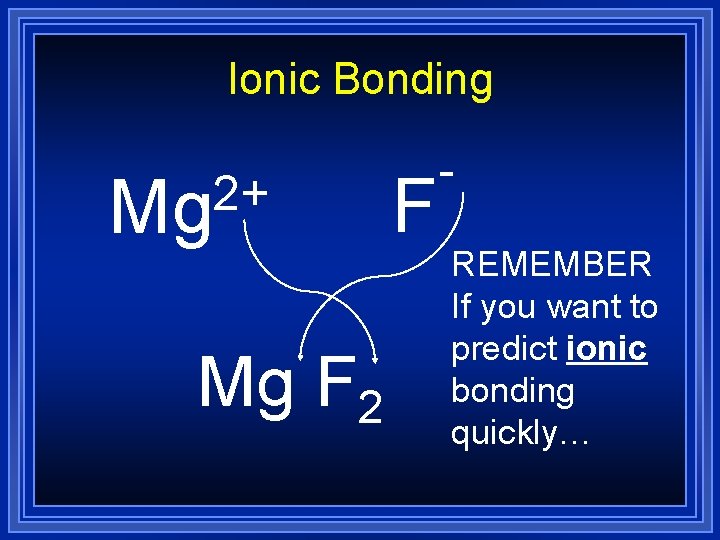
Ionic Bonding 2+ Mg Mg F 2 F REMEMBER If you want to predict ionic bonding quickly…
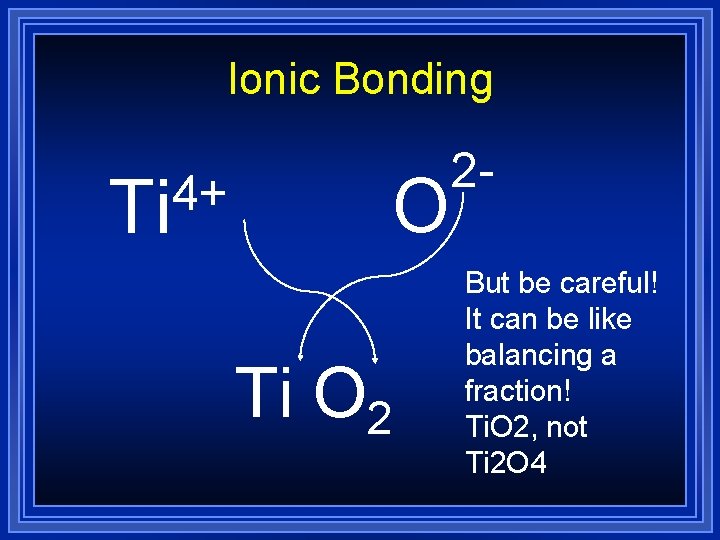
Ionic Bonding 4+ Ti O 2 2 But be careful! It can be like balancing a fraction! Ti. O 2, not Ti 2 O 4

Peer-to-Peer Question #1 l Draw the FORMULA UNIT for the following atoms when they create an ionic bond: – Mg and Cl – Al and O – K and Br – Li and O

Properties of Ionic Compounds 1. Crystalline solids - a regular repeating arrangement of ions in the solid: Fig. 7. 9, page 197 – Ions are strongly bonded together. – Structure is rigid. 2. High melting points l Coordination number- number of ions of opposite charge surrounding it
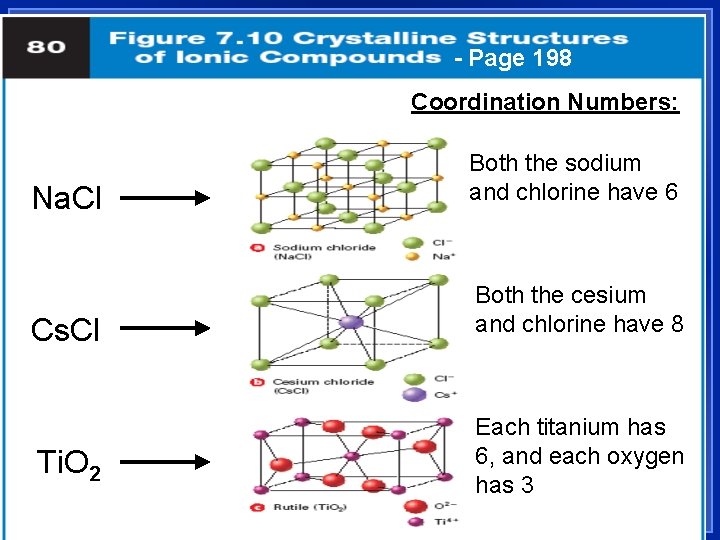
- Page 198 Coordination Numbers: Na. Cl Both the sodium and chlorine have 6 Cs. Cl Both the cesium and chlorine have 8 Ti. O 2 Each titanium has 6, and each oxygen has 3

Do they Conduct? Conducting electricity means allowing charges to move. l In a solid, the ions are locked in place. l Ionic solids are insulators. l When melted, the ions can move around. 3. Melted ionic compounds conduct. – Na. Cl: must get to about 800 ºC. – Dissolved in water, they also conduct (free to move in aqueous solutions) – Ex: Salt water conducts electricity but WATER alone does NOT l

- Page 198 The ions are free to move when they are molten (or in aqueous solution), and thus they are able to conduct the electric current.

Bellringer #58 (Jan 21 st, 2011) l Determine the formula and name for when the following atoms combine to create IONIC COMPOUNDS: – Ca and O – Al and S – K and N

Agenda (Jan 21 st, 2011) Bellringer #58 l Homework Check l 2 nd attempt distribution and discussion l Review for Semester Exam l Homework: Study Guide for EXTRA CREDIT IF YOU PASS!!!!!! – Extra study guide available on homework website…. . VERY helpful! l

Bellringer #59 (Jan 24 th, 2011) l Determine the electron configuration for the following atoms: – Phosphorous – Fluorine – Calcium

Bellringer #1 (Jan 31 st, 2011) l Name three things you know about ionic compounds.

Agenda (Jan 31 st, 2011) Bellringer #1 l Discuss new semester l Objective: Learn how to name ionic compounds from transition metal ions l Work on Chapter 7 Section Assessment: pg. 207 #30 -47 l Hmwk: Cornell Notes 7. 3 l Mini Quiz Tomorrow l
New Semester…. . New Beginnings l l l New Semester: Everyone starts all over again Starting at BR #1: Collecting BR #1 -59 TOMORROW Late Work Policy: accept homework 1 DAY LATE. Work MUST have a note on it as to WHY it is late and personally hand to Ms. Ingham. Don’t wait too late to ask for help. This semester WILL BE harder than last. Homework Board in back of room WILL BE updated.

Cornell Notes l Cornell Notes MUST include the following for COMPLETE credit: – Answer KEY CONCEPT questions. – Define ALL vocabulary in the section AND give a picture and/or example. – Write notes for EVERY RED heading in the section. – All notes MUST end with a brief summary discussing the main points of the section. (If you take good notes, this should not be hard to do. )

Ionic Compounds With Transition Metals l l The charges for transition metals are not as easy to predict as the metals from representative elements. – Ex: Copper can have a charge of 1+ or 2+ (Cu 1+ or Cu 2+) For this reason you MUST be given the charge of the ion from a transition metal if you are going to create a ionic compound – Ex: Ti 4+ and O

Ion Examples Symbol Name Cu+ Copper (I) ion Cu 2+ Fe 3+ Pb 2+ Pb 4+ Sn 2+ Sn 4+ Copper (II) ion Iron (III) ion Lead (IV) ion Tin (II) ion Tin (IV) ion

#1: Complete the table below (turn in at end of class). Symbol Cr 2+ Name Chromium (III) ion Mn 2+ Manganese (III) ion Cobalt (II) ion Co 3+

Predicting Ionic Compounds and Charges of Transition Metals l What Copper ion would create Cu. O? l What Copper ion would create Cu 2 O?

#2: Predict the ion that would create the compound below, then name the compound. Sn. F 2 l Sn. S 2 l Fe 2 O 3 l

What Next? Turn in questions #1 and #2 l Begin work on pg. 207 (unfinished work is homework along with Cornell notes) l Mini Quiz Tomorrow l Bellringers #1 -59 due TOMORROW l

Bellringer #2 l Write THREE things you know about metals.

Agenda (Feb 1 st, 2011) Bellringer #2 l Homework Check l 1 st Semester BR Collection l Write 7. 3 Objectives l 7. 3 Overview l Mini Quiz l Hmwk: 7. 3 Section Assessment #23 -29 and Writing Activity l

Section 7. 3 Bonding in Metals l OBJECTIVES: –Model the valence electrons of metal atoms.

Section 7. 3 Bonding in Metals l OBJECTIVES: –Describe the arrangement of atoms in a metal.

Section 7. 3 Bonding in Metals l OBJECTIVES: –Explain the importance of alloys.

Metallic Bonds are… l How metal atoms are held together in the solid. l Metals hold on to their valence electrons very weakly. l Think of them as positive ions (cations) floating in a sea of electrons: Fig. 7. 12, p. 201

Sea of Electrons l Electrons are free to move through the solid. l Metals conduct electricity. + + +

Metals are Malleable l Hammered into shape (bend). l Also ductile - drawn into wires. l Both malleability and ductility explained in terms of the mobility of the valence electrons
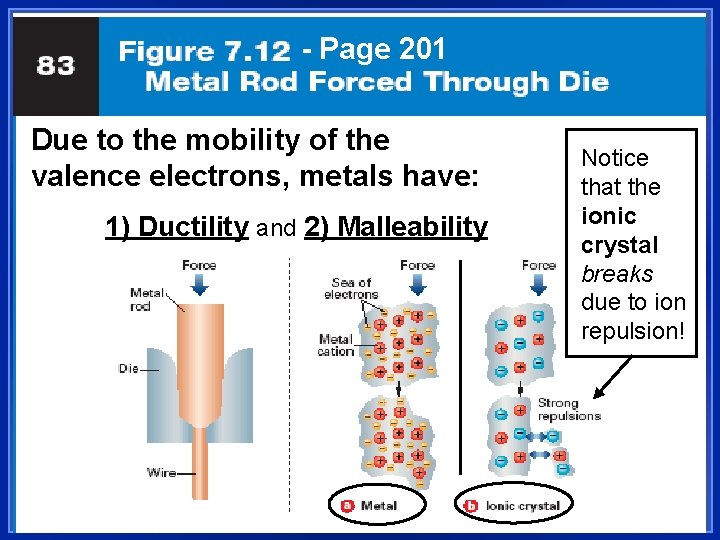
- Page 201 Due to the mobility of the valence electrons, metals have: 1) Ductility and 2) Malleability Notice that the ionic crystal breaks due to ion repulsion!
Malleable Force + + +
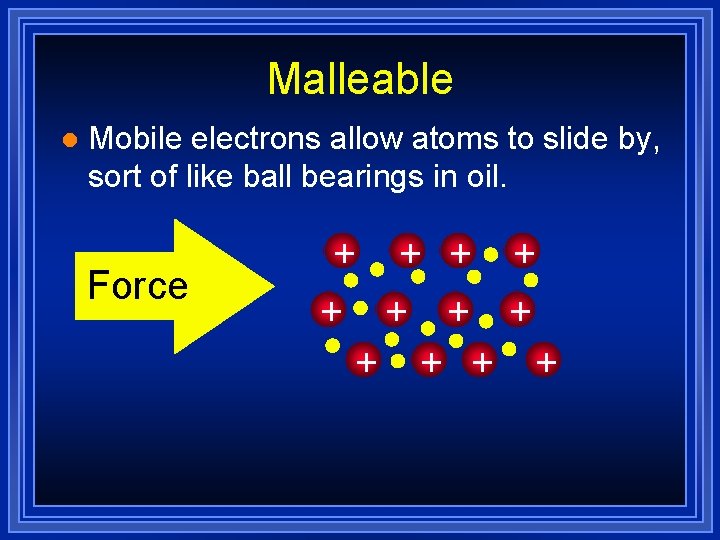
Malleable l Mobile electrons allow atoms to slide by, sort of like ball bearings in oil. Force + + +

Ionic solids are brittle Force + + - + +

Ionic solids are brittle l Strong Repulsion breaks a crystal apart, due to similar ions being next to each other. Force + + - + - + - +

Crystalline structure of metal (pg. 202) l If made of one kind of atom, metals are among the simplest crystals; very compact & orderly l Note Fig. 7. 14, p. 202 for types: 1. Body-centered cubic: –every atom (except those on the surface) has 8 neighbors –Na, K, Fe, Cr, W

Crystalline structure of metal (pg. 202) 2. Face-centered cubic: – every atom has 12 neighbors – Cu, Ag, Au, Al, Pb 3. Hexagonal close-packed – every atom also has 12 neighbors – different pattern due to hexagonal – Mg, Zn, Cd

Alloys l We use lots of metals every day, but few are pure metals l Alloys are mixtures of 2 or more elements, at least 1 is a metal l made by melting a mixture of the ingredients, then cooling l Brass: an alloy of Cu and Zn l Bronze: Cu and Sn

Why use alloys? Properties are often superior to the pure element l Sterling silver (92. 5% Ag, 7. 5% Cu) is harder and more durable than pure Ag, but still soft enough to make jewelry and tableware l Steels are very important alloys – corrosion resistant, ductility, hardness, toughness, cost l

More about Alloys… l Table 7. 3, p. 203 – lists a few alloys l Types? a) substitutional alloy- the atoms in the components are about the same size l b) interstitial alloy- the atomic sizes quite different; smaller atoms fit into the spaces between larger l “Amalgam”- dental use, contains Hg

- Slides: 87