Carotenoid Pyrus communis HPLC 280 nm 640 nm























- Slides: 97




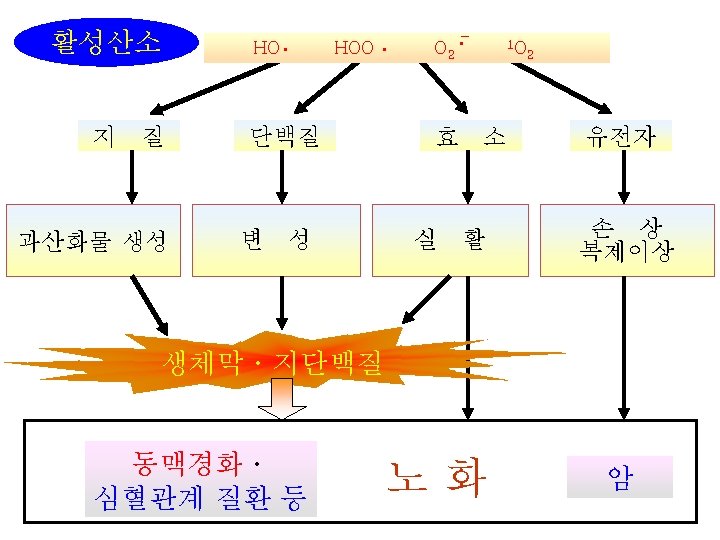
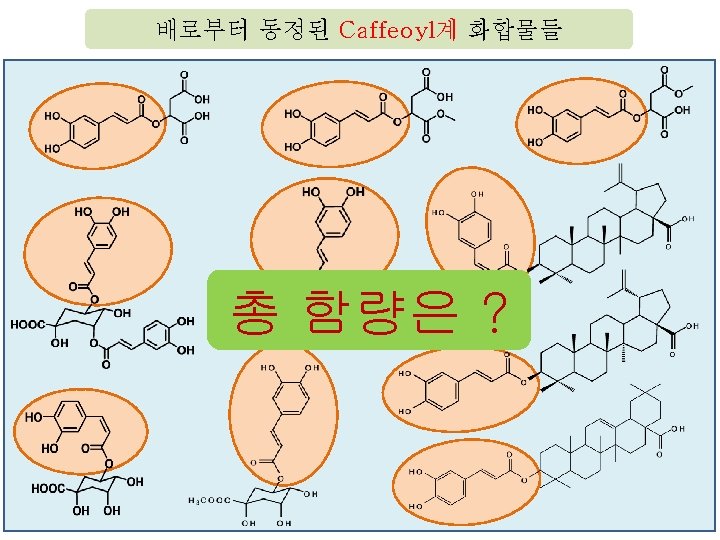
Carotenoid



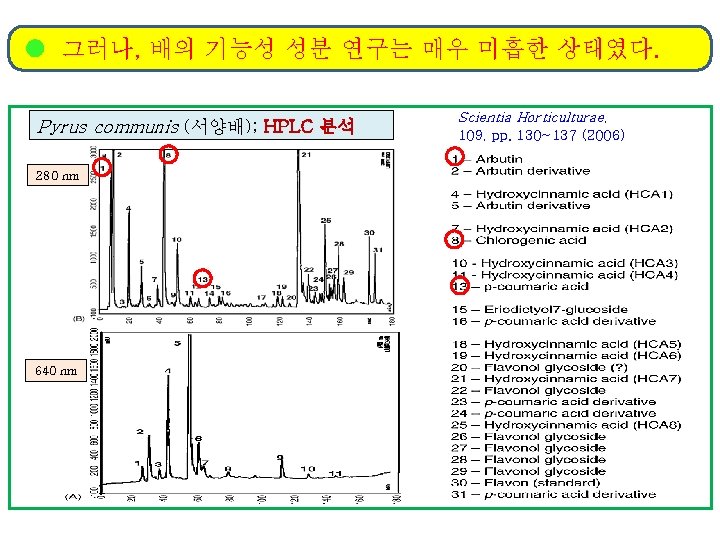
그러나, 배의 기능성 성분 연구는 매우 미흡한 상태였다. Pyrus communis (서양배); HPLC 분석 280 nm 640 nm Scientia Horticulturae, 109, pp. 130~137 (2006)

4핵심 1세부: 배 함유 생리활성 화합물의 탐색 및 분석 Ⅰ. 연구사업 추진 배경 동양배 한국배 서양배 한국배의 기능성 성분 홍보자료 거의 전무 이 유 • 체계적인 한국배의 성분 연구 전무했기 때문 Peak No. Identification 1 Arbutin 3 3 -Caffeoylquinic acid Peak No. • 표준품이 없음 28 A Identification Feruloylmalic acid 30 Isorhamnetin 3 -O-robinioside 5 Chlorogenic acid 31 Isorhamnetin 3 -O-rutinoside 7 4 -Caffeoylquinic acid 33 3, 4 -Dicaffeoylquinic acid Epicatechin 34 Isorhamnetin 3 -O-galactoside Quercetin 3 -O-rutinos 35 Chrysoeriol 7 -O-rutinoside 10 A 20









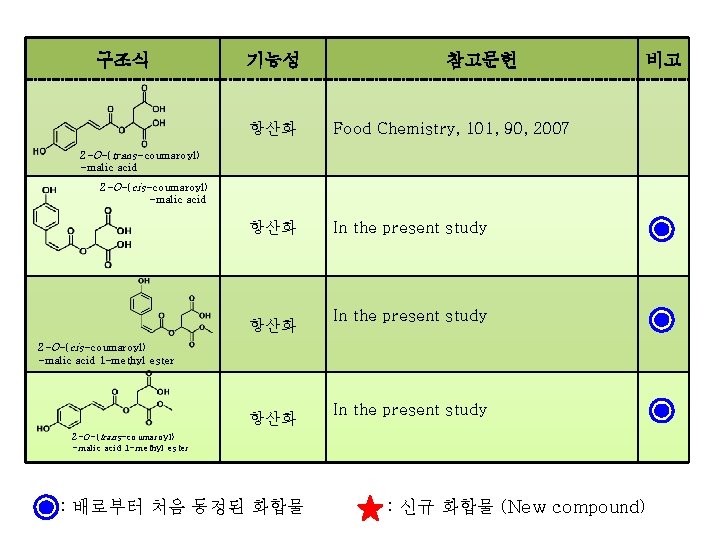
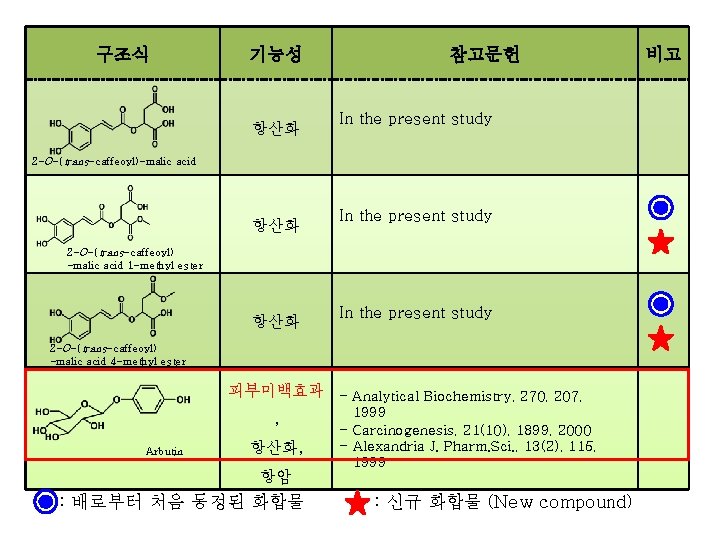
구조식 기능성 참고문헌 항산화 Food Chemistry, 101, 90, 2007 항산화 In the present study 비고 2 -O-(trans-coumaroyl) -malic acid 2 -O-(cis-coumaroyl) -malic acid 항산화 In the present study 2 -O-(cis-coumaroyl) -malic acid 1 -methyl ester 항산화 In the present study 2 -O-(trans-coumaroyl) -malic acid 1 -methyl ester : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound)

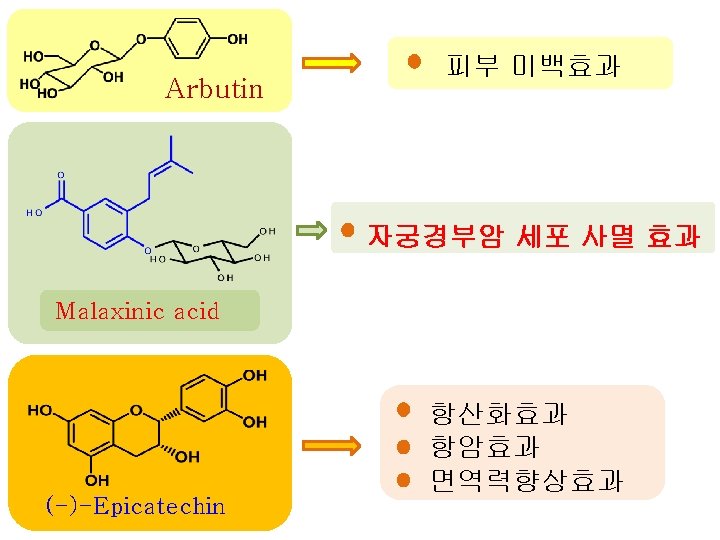
구조식 기능성 항산화 참고문헌 In the present study 2 -O-(trans-caffeoyl)-malic acid 항산화 In the present study 2 -O-(trans-caffeoyl) -malic acid 1 -methyl ester 항산화 In the present study 2 -O-(trans-caffeoyl) -malic acid 4 -methyl ester 피부미백효과 - Analytical Biochemistry, 270, 207, 1999 , Arbutin 항산화, 항암 : 배로부터 처음 동정된 화합물 - Carcinogenesis, 21(10), 1899, 2000 - Alexandria J. Pharm. Sci. , 13(2), 115, 1999 : 신규 화합물 (New compound) 비고

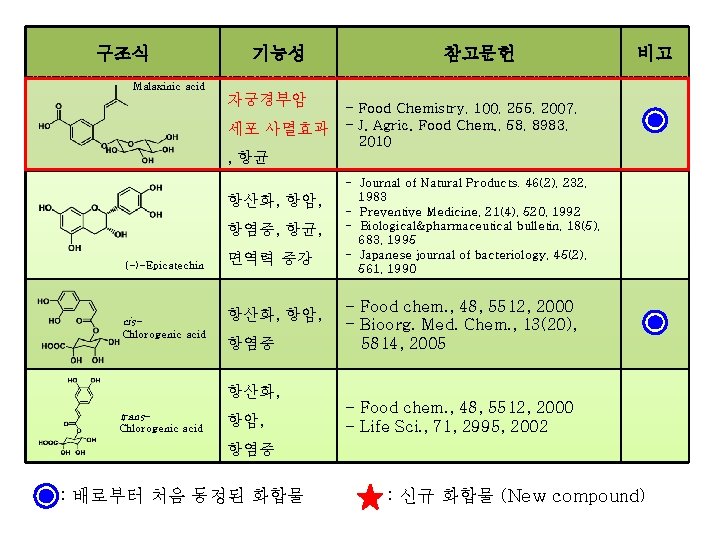
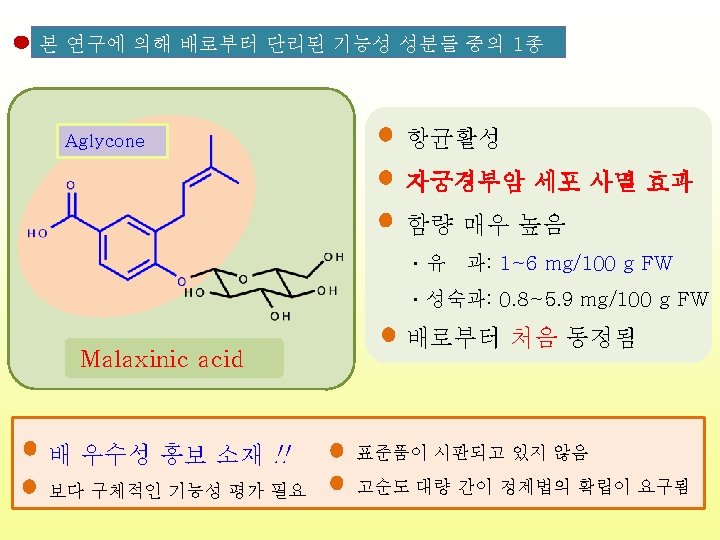
구조식 기능성 참고문헌 비고 Malaxinic acid 자궁경부암 세포 사멸효과 - Food Chemistry, 100, 255, 2007, - J. Agric. Food Chem. , 58, 8983, 2010 , 항균 항산화, 항암, 항염증, 항균, (-)-Epicatechin 면역력 증강 cis- 항산화, 항암, Chlorogenic acid 항염증 항산화, trans. Chlorogenic acid 항암, - Journal of Natural Products. 46(2), 232, 1983 - Preventive Medicine, 21(4), 520, 1992 - Biological&pharmaceutical bulletin, 18(5), 683, 1995 - Japanese journal of bacteriology, 45(2), 561, 1990 - Food chem. , 48, 5512, 2000 - Bioorg. Med. Chem. , 13(20), 5814, 2005 - Food chem. , 48, 5512, 2000 - Life Sci. , 71, 2995, 2002 항염증 : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound)

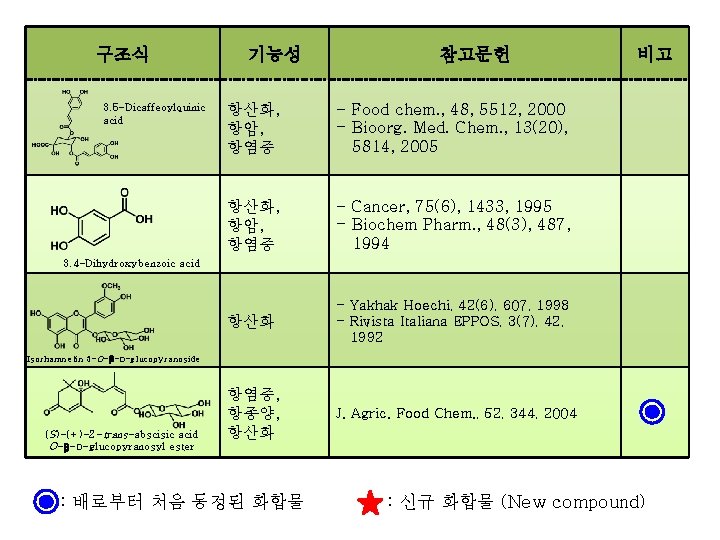
구조식 3, 5 -Dicaffeoylquinic acid 기능성 참고문헌 항산화, 항암, 항염증 - Food chem. , 48, 5512, 2000 - Bioorg. Med. Chem. , 13(20), 5814, 2005 항산화, 항암, 항염증 - Cancer, 75(6), 1433, 1995 - Biochem Pharm. , 48(3), 487, 1994 항산화 - Yakhak Hoechi, 42(6), 607, 1998 - Rivista Italiana EPPOS, 3(7), 42, 1992 항염증, 항종양, 항산화 J. Agric. Food Chem. , 52, 344, 2004 비고 3, 4 -Dihydroxybenzoic acid Isorhamnetin 3 -O- -D-glucopyranoside (S)-(+)-2 -trans-abscisic acid O-β-D-glucopyranosyl ester : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound)

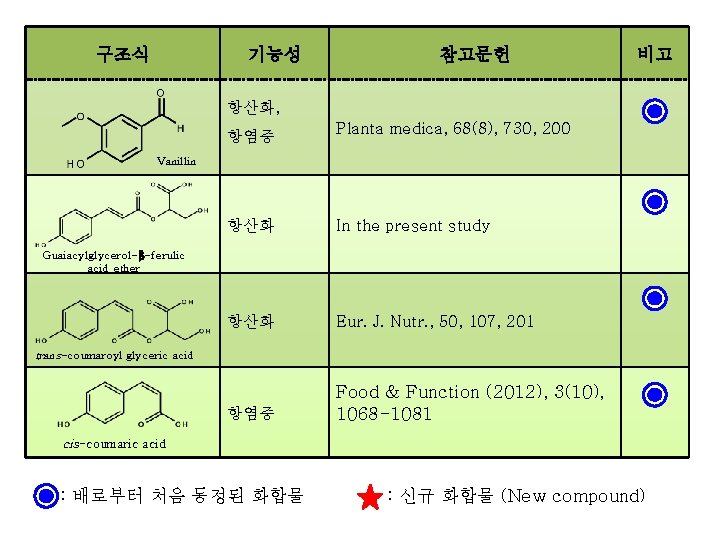
구조식 기능성 참고문헌 비고 항산화, 항염증 Planta medica, 68(8), 730, 200 Vanillin 항산화 In the present study 항산화 Eur. J. Nutr. , 50, 107, 201 항염증 Food & Function (2012), 3(10), 1068 -1081 Guaiacylglycerol-β-ferulic acid ether trans-coumaroyl glyceric acid cis-coumaric acid : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound)

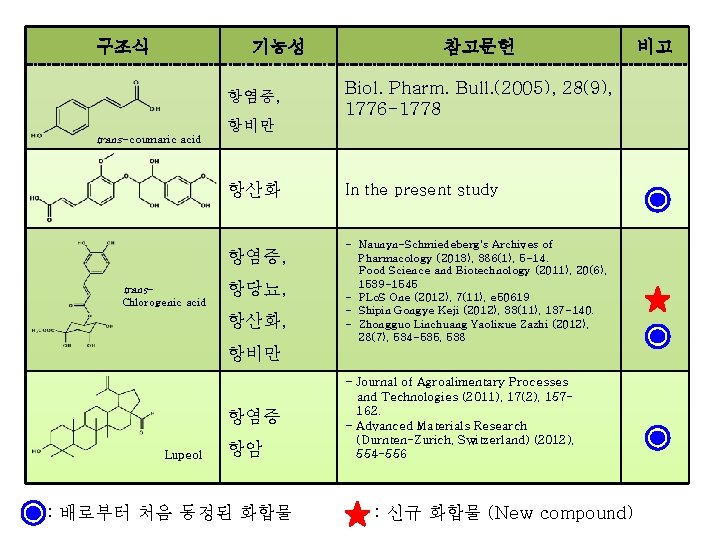
구조식 기능성 항염증, trans-coumaric acid 항비만 항산화 항염증, trans. Chlorogenic acid 항당뇨, 항산화, 참고문헌 Biol. Pharm. Bull. (2005), 28(9), 1776 -1778 In the present study - Naunyn-Schmiedeberg's Archives of Pharmacology (2013), 386(1), 5 -14. Food Science and Biotechnology (2011), 20(6), 1539 -1545 - PLo. S One (2012), 7(11), e 50619 - Shipin Gongye Keji (2012), 33(11), 137 -140. - Zhongguo Linchuang Yaolixue Zazhi (2012), 28(7), 534 -535, 538 항비만 항염증 Lupeol 항암 : 배로부터 처음 동정된 화합물 - Journal of Agroalimentary Processes and Technologies (2011), 17(2), 157162. - Advanced Materials Research (Durnten-Zurich, Switzerland) (2012), 554 -556 : 신규 화합물 (New compound) 비고

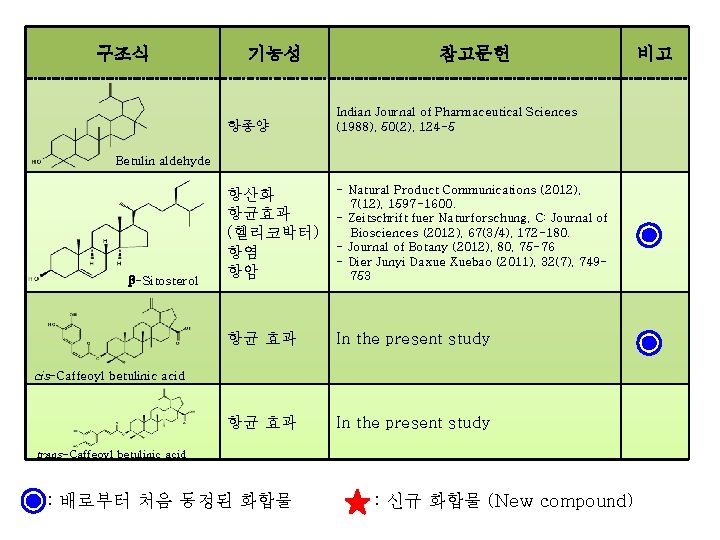
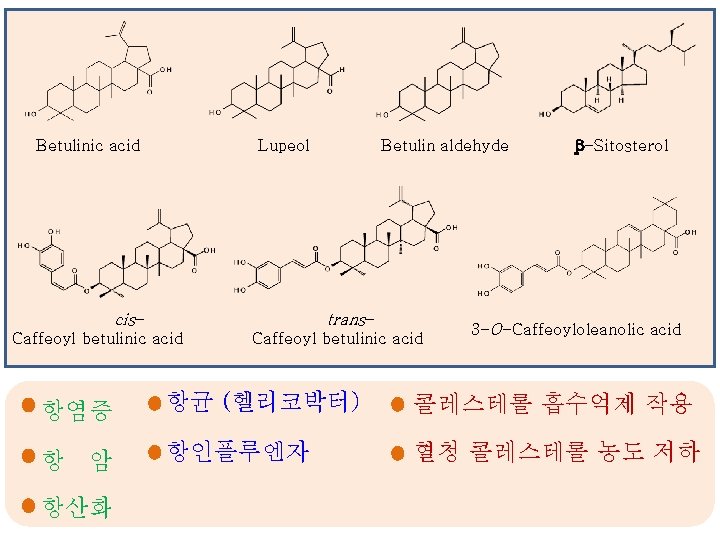
구조식 기능성 참고문헌 항종양 Indian Journal of Pharmaceutical Sciences (1988), 50(2), 124 -5 항산화 항균효과 (헬리코박터) 항염 항암 - Natural Product Communications (2012), 7(12), 1597 -1600. - Zeitschrift fuer Naturforschung, C: Journal of Biosciences (2012), 67(3/4), 172 -180. - Journal of Botany (2012), 80, 75 -76 - Dier Junyi Daxue Xuebao (2011), 32(7), 749753 항균 효과 In the present study Betulin aldehyde β-Sitosterol cis-Caffeoyl betulinic acid trans-Caffeoyl betulinic acid : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound) 비고

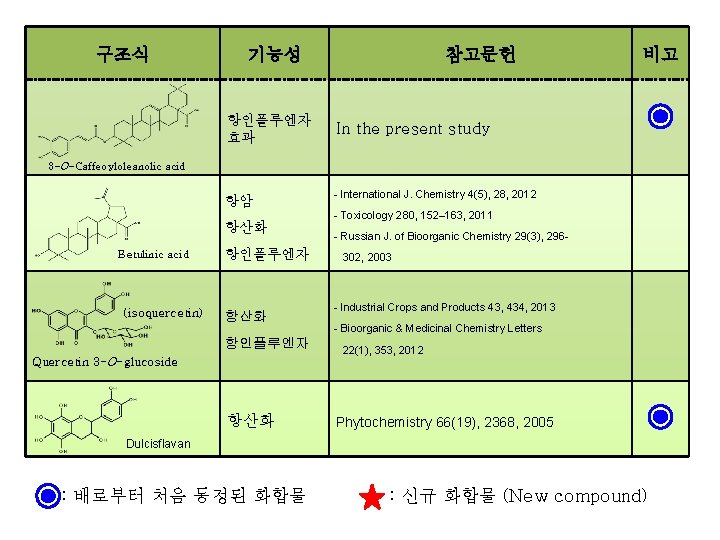
구조식 기능성 항인플루엔자 효과 참고문헌 비고 In the present study 3 -O-Caffeoyloleanolic acid 항암 항산화 Betulinic acid (isoquercetin) 항인플루엔자 항산화 항인플루엔자 Quercetin 3 -O-glucoside 항산화 - International J. Chemistry 4(5), 28, 2012 - Toxicology 280, 152– 163, 2011 - Russian J. of Bioorganic Chemistry 29(3), 296302, 2003 - Industrial Crops and Products 43, 434, 2013 - Bioorganic & Medicinal Chemistry Letters 22(1), 353, 2012 Phytochemistry 66(19), 2368, 2005 Dulcisflavan : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound)

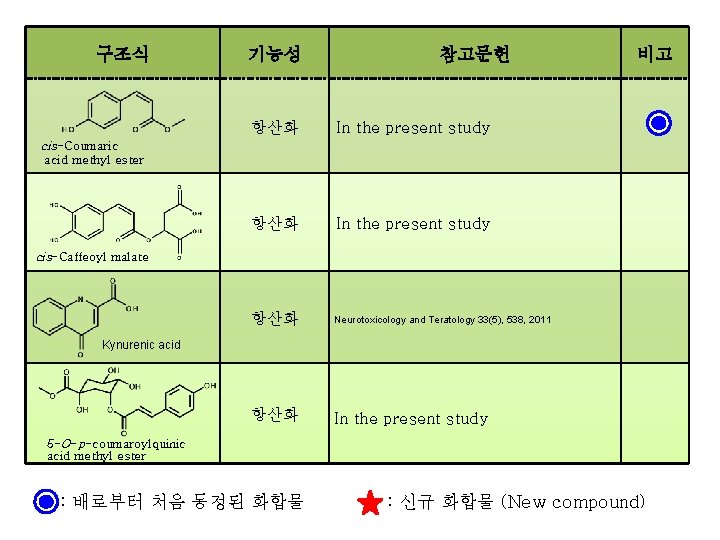
구조식 기능성 참고문헌 항산화 In the present study 항산화 Neurotoxicology and Teratology 33(5), 538, 2011 항산화 In the present study 비고 cis-Coumaric acid methyl ester cis-Caffeoyl malate Kynurenic acid 5 -O-p-coumaroylquinic acid methyl ester : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound)

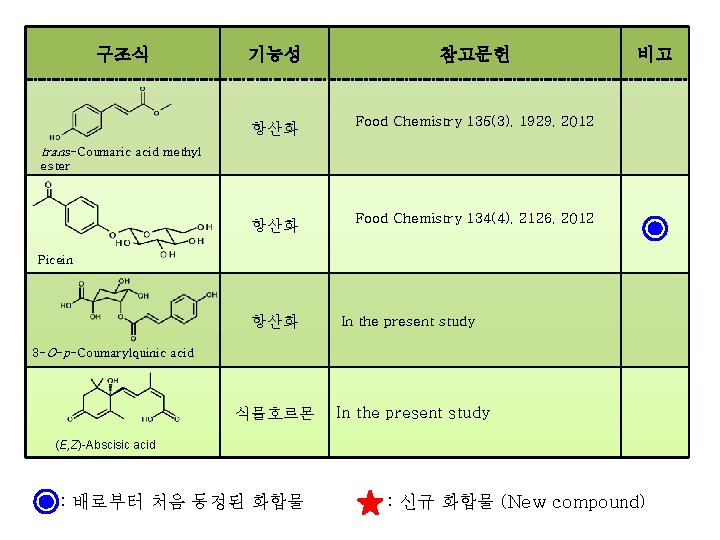
구조식 기능성 참고문헌 항산화 Food Chemistry 135(3), 1929, 2012 항산화 Food Chemistry 134(4), 2126, 2012 비고 trans-Coumaric acid methyl ester Picein 항산화 In the present study 3 -O-p-Coumarylquinic acid 식물호르몬 In the present study (E, Z)-Abscisic acid : 배로부터 처음 동정된 화합물 : 신규 화합물 (New compound)

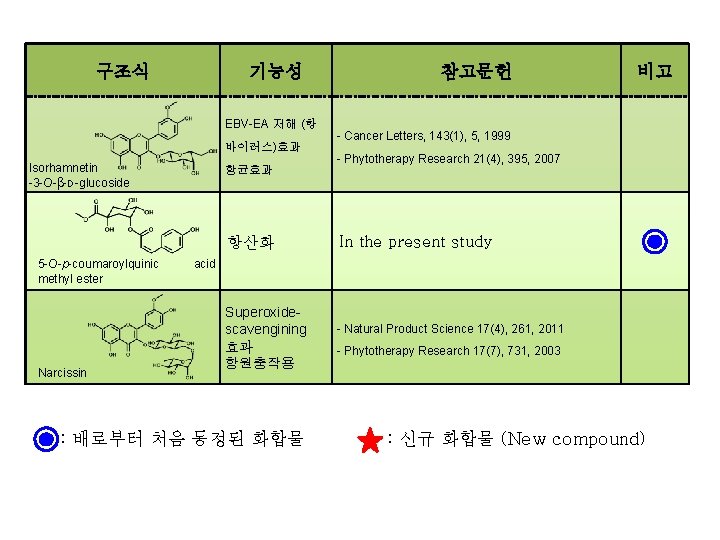
구조식 기능성 EBV-EA 저해 (항 바이러스)효과 Isorhamnetin -3 -O-β-D-glucoside 항균효과 항산화 5 -O-p-coumaroylquinic methyl ester Narcissin 참고문헌 비고 - Cancer Letters, 143(1), 5, 1999 - Phytotherapy Research 21(4), 395, 2007 In the present study acid Superoxidescavengining 효과 항원충작용 : 배로부터 처음 동정된 화합물 - Natural Product Science 17(4), 261, 2011 - Phytotherapy Research 17(7), 731, 2003 : 신규 화합물 (New compound)

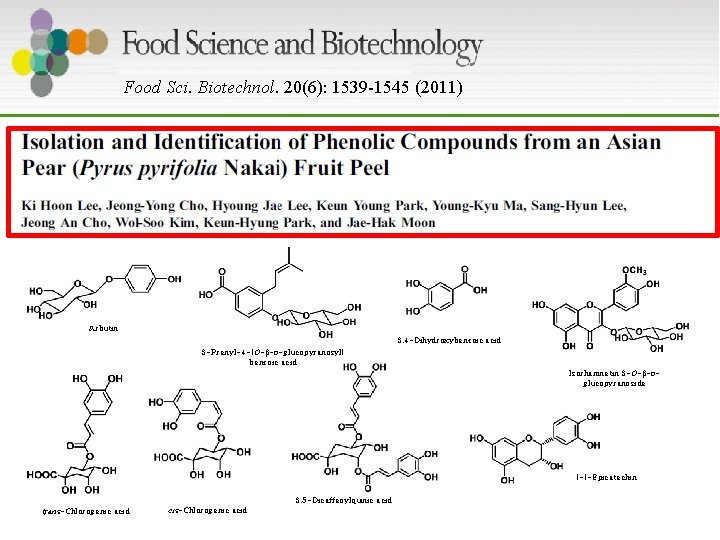
Food Sci. Biotechnol. 20(6): 1539 -1545 (2011) Arbutin 3, 4 -Dihydroxybenzoic acid 3 -Prenyl-4 -(O- -D-glucopyranosyl) benzoic acid Isorhamnetin 3 -O- -Dglucopyranoside (-)-Epicatechin 3, 5 -Dicaffeoylquinic acid trans-Chlorogenic acid cis-Chlorogenic acid

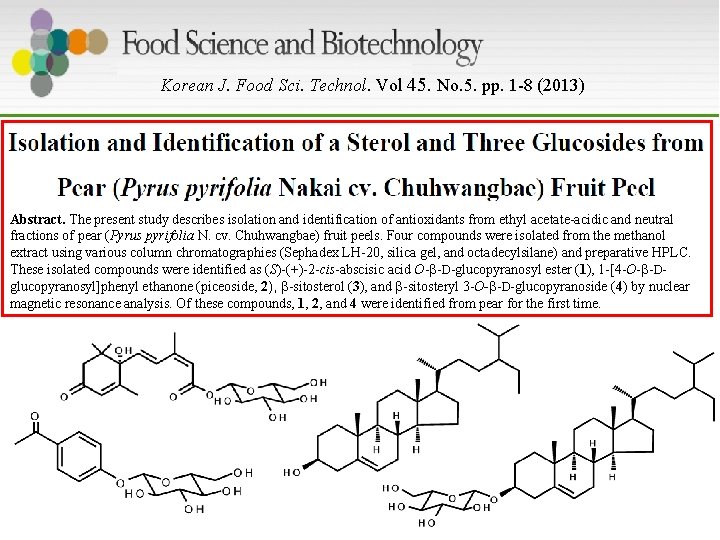
Korean J. Food Sci. Technol. Vol 45. No. 5. pp. 1 -8 (2013) Abstract. The present study describes isolation and identification of antioxidants from ethyl acetate-acidic and neutral fractions of pear (Pyrus pyrifolia N. cv. Chuhwangbae) fruit peels. Four compounds were isolated from the methanol extract using various column chromatographies (Sephadex LH-20, silica gel, and octadecylsilane) and preparative HPLC. These isolated compounds were identified as (S)-(+)-2 -cis-abscisic acid O-β-D-glucopyranosyl ester (1), 1 -[4 -O-β-Dglucopyranosyl]phenyl ethanone (piceoside, 2), β-sitosterol (3), and β-sitosteryl 3 -O-β-D-glucopyranoside (4) by nuclear magnetic resonance analysis. Of these compounds, 1, 2, and 4 were identified from pear for the first time.

Korean J. Food Sci. Technol. Vol. 45. No. 2. pp. 174 -179 (2013) Isolation and Identification of 3 Low-Molecular Compounds from Pear (Pyrus pyrifolia Nakai cv. Chuhwangbae) Fruit Peel Abstract Three low-molecular compounds were isolated from methanol extracts of pear (Pyrus pyrifolia N. cv. Chuhwangbae) fruit peels using solvent fractionation, various types of column chromatogrphy (Diaion HP-20, Sephadex LH-20, and silica gel), and high performance liquid chromatography with an assay guided by 1, 1 -diphenyl-2 picrylhydrazyl radical-scavenging activity. The isolated compounds were identified as 2 -carboxyl-4(1 H)-quinolinone (kynurenic acid, 1) from Bu. OH fraction, cis-p-coumaric acid (2) from Et. OAc-acidic fraction, and vanillin (3) from the Et. OAc-phenolic fraction, respectively. These isolated compounds were confirmed on the basis of the spectroscopic data of electrospray ionization mass spectrometry and nuclear magnetic resonance. This is the first time that compounds 1 -3 were isolated and identified in pear.

J. Agric. Food Chem. 2011, 59, 10124 -10128

Food Sci. Biotechnol. 22(3): 803 -810 (2013)

J. Agric. Food Chem. 2013, 61, 4563− 4569


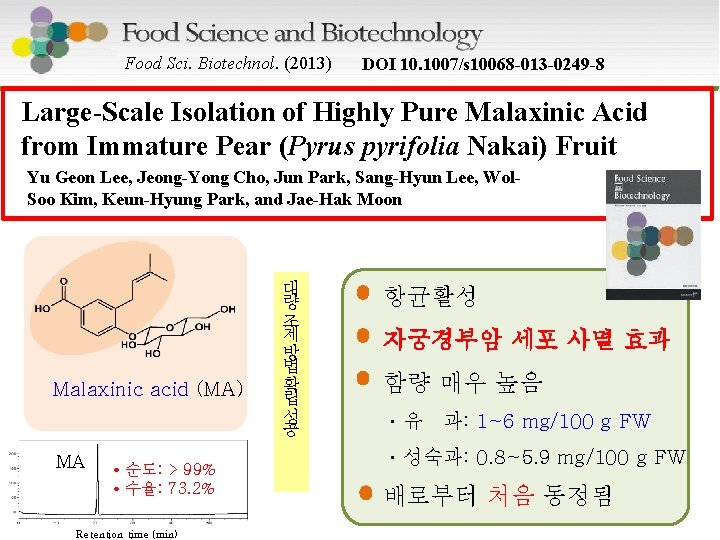
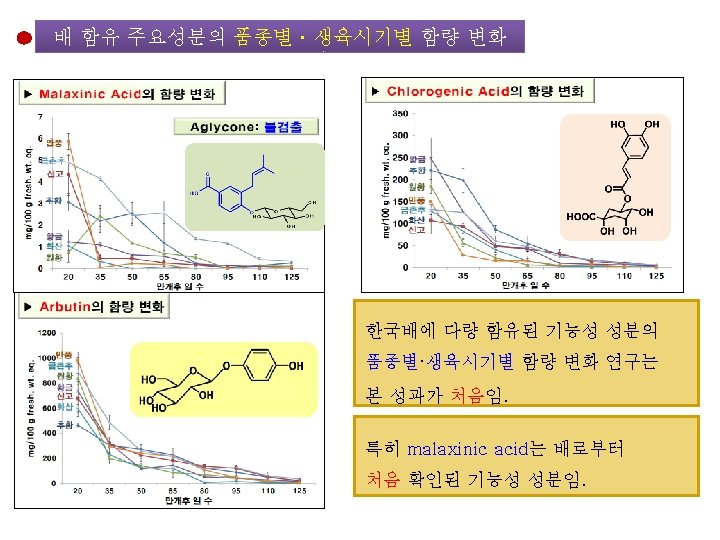
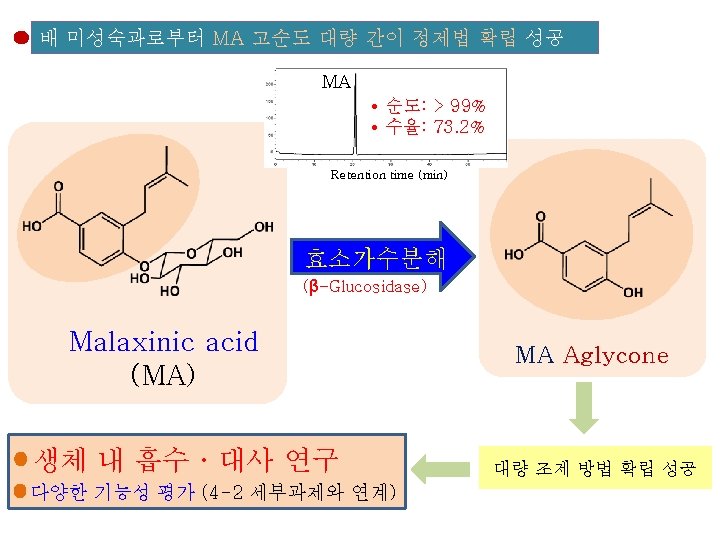
Food Sci. Biotechnol. (2013) DOI 10. 1007/s 10068 -013 -0249 -8 Large-Scale Isolation of Highly Pure Malaxinic Acid from Immature Pear (Pyrus pyrifolia Nakai) Fruit Yu Geon Lee, Jeong-Yong Cho, Jun Park, Sang-Hyun Lee, Wol. Soo Kim, Keun-Hyung Park, and Jae-Hak Moon Malaxinic acid (MA) MA • 순도: > 99% • 수율: 73. 2% Retention time (min) 대 량 조 제 방 법 확 립 성 공 항균활성 자궁경부암 세포 사멸 효과 함량 매우 높음 ㆍ유 과: 1~6 mg/100 g FW ㆍ성숙과: 0. 8~5. 9 mg/100 g FW 배로부터 처음 동정됨

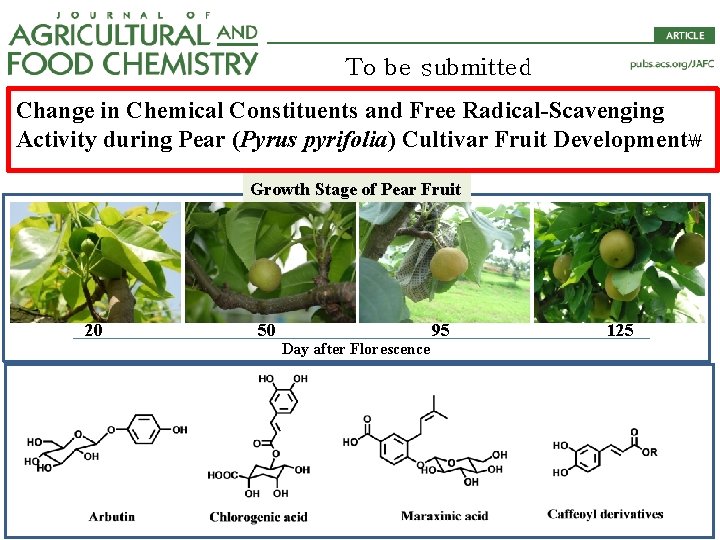
To be submitted Change in Chemical Constituents and Free Radical-Scavenging Activity during Pear (Pyrus pyrifolia) Cultivar Fruit Development Growth Stage of Pear Fruit 20 50 Day after Florescence 95 125

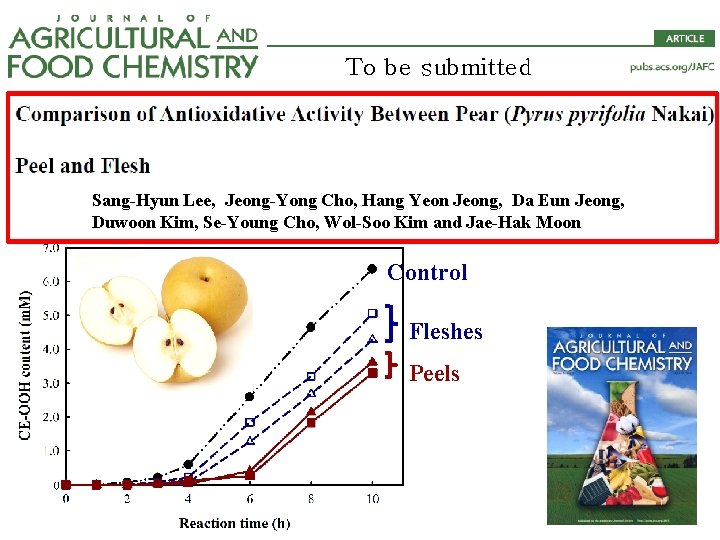
To be submitted Sang-Hyun Lee, Jeong-Yong Cho, Hang Yeon Jeong, Da Eun Jeong, Duwoon Kim, Se-Young Cho, Wol-Soo Kim and Jae-Hak Moon Control Fleshes Peels

Food Sci. Biotechnol. 23(1): 1 -7 (2014) DOI 10. 1007/s 10068 -014 -0

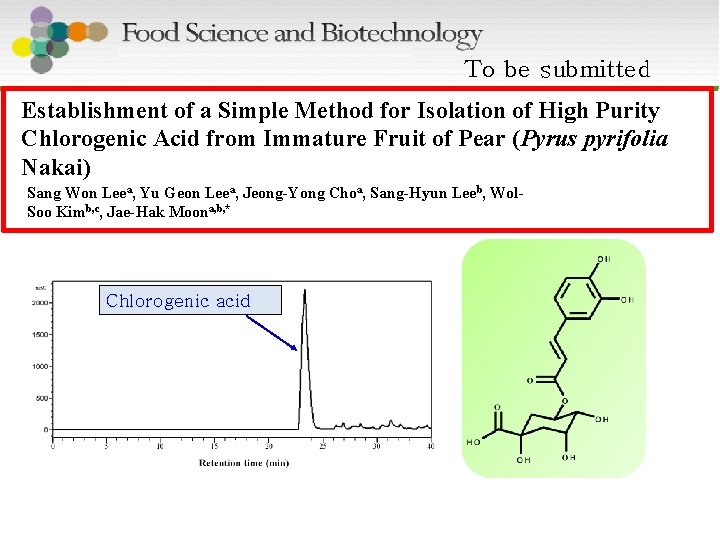
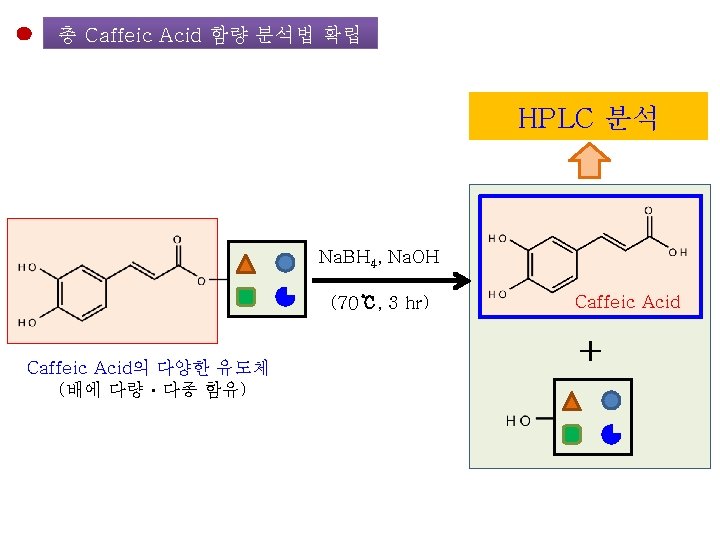
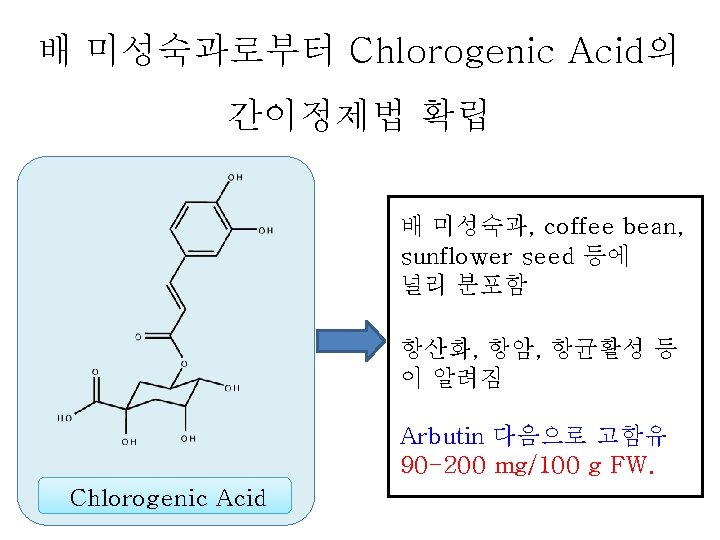
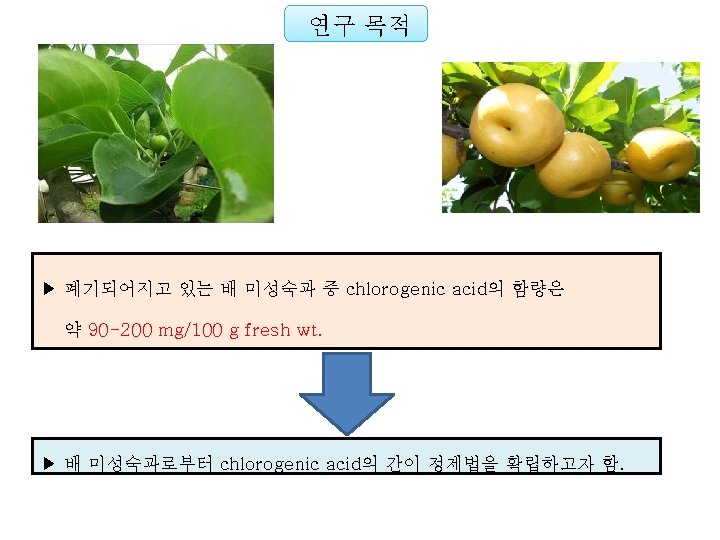
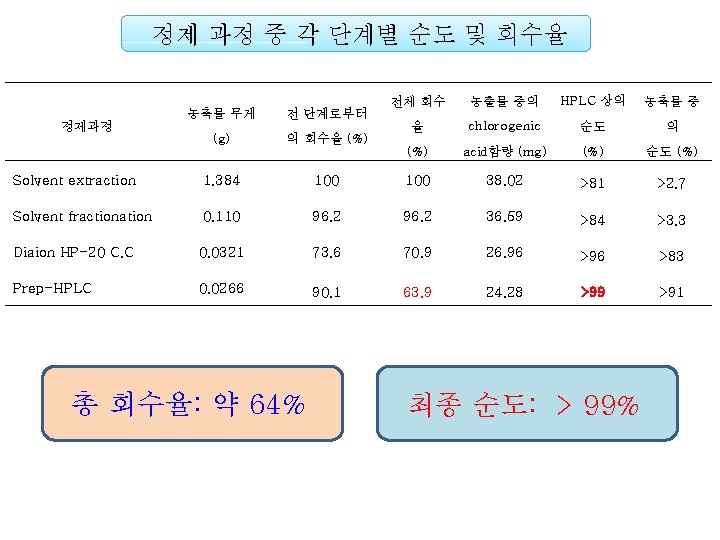
To be submitted Establishment of a Simple Method for Isolation of High Purity Chlorogenic Acid from Immature Fruit of Pear (Pyrus pyrifolia Nakai) Sang Won Leea, Yu Geon Leea, Jeong-Yong Choa, Sang-Hyun Leeb, Wol. Soo Kimb, c, Jae-Hak Moona, b, * Chlorogenic acid

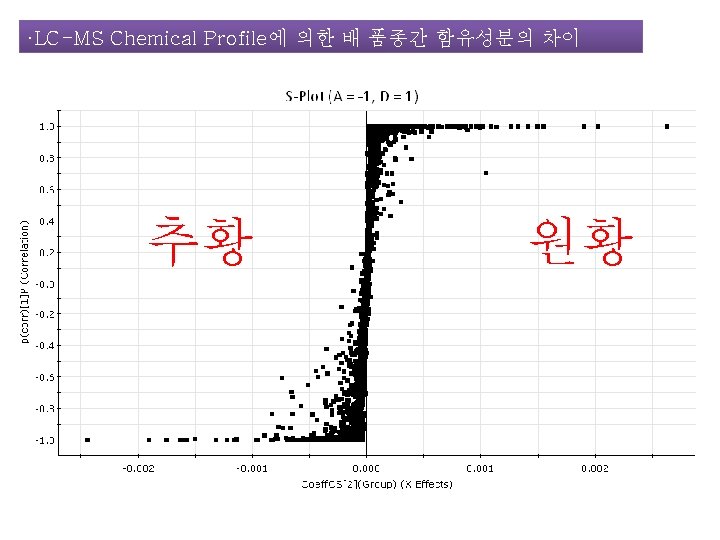
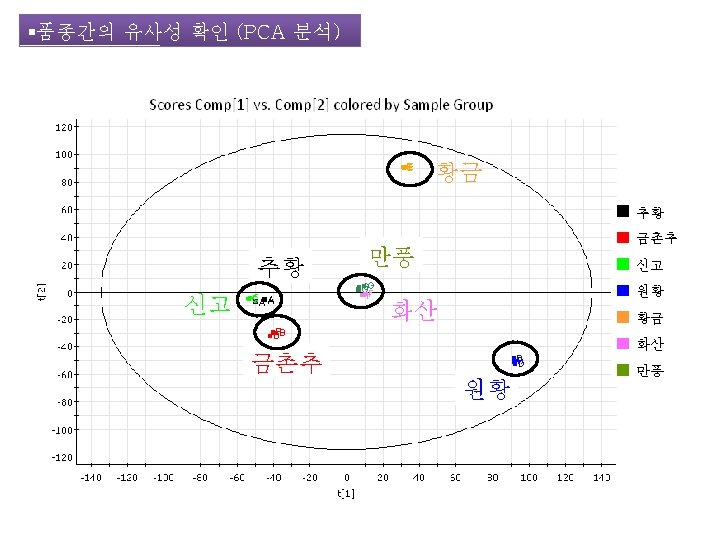
LC-MS를 이용한 배 함유성분의 Chemical Profile 작성 The Analysis of Sample Using UPLC/SYNAPT G 2 -HDMS Q-TOF 2013 한국식품과학회 포스터 발표 J. Agric. Food Chem. 작성중



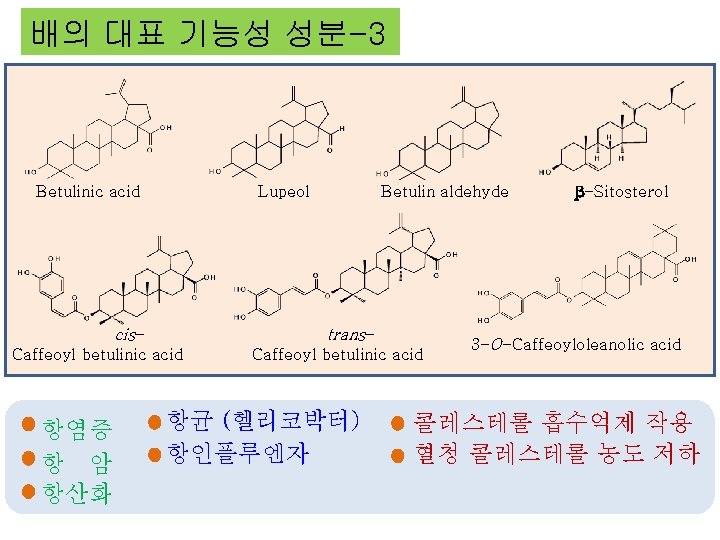
배의 대표 기능성 성분-3 Betulinic acid Lupeol cis- trans- Caffeoyl betulinic acid 항염증 항 암 항산화 Betulin aldehyde Caffeoyl betulinic acid 항균 (헬리코박터) 항인플루엔자 β-Sitosterol 3 -O-Caffeoyloleanolic acid 콜레스테롤 흡수억제 작용 혈청 콜레스테롤 농도 저하



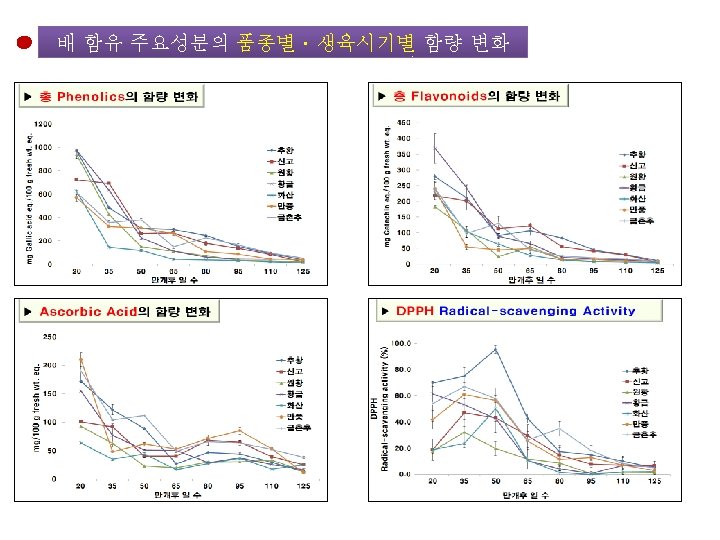
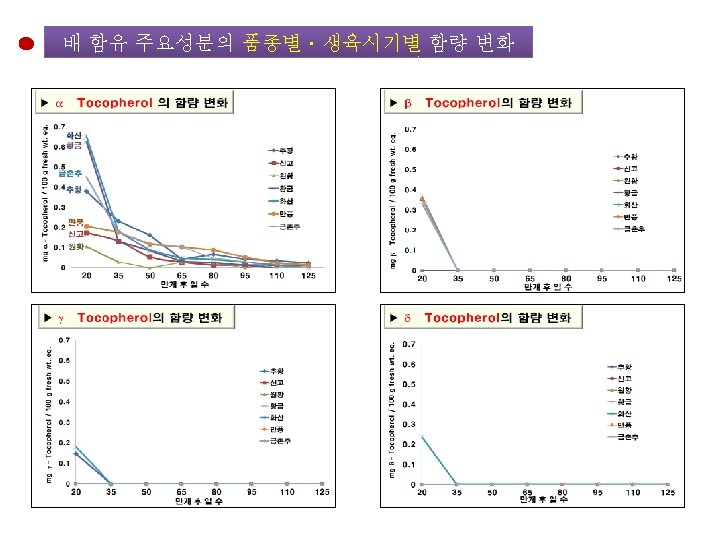
배 품종별ㆍ생육시기별 주요 성분의 함량 변화 Growth Stage of Pear Fruit 20 50 95 Day after Florescence 125 <분석 대상 항목> • • • Chlorogenic acid Malaxinic acid 및 그 Aglycone Arbutin Ascorbic acid Tocopherol Total Phenolics Total Flavonoids Total Caffeic acid DPPH radical-scavenging 활성






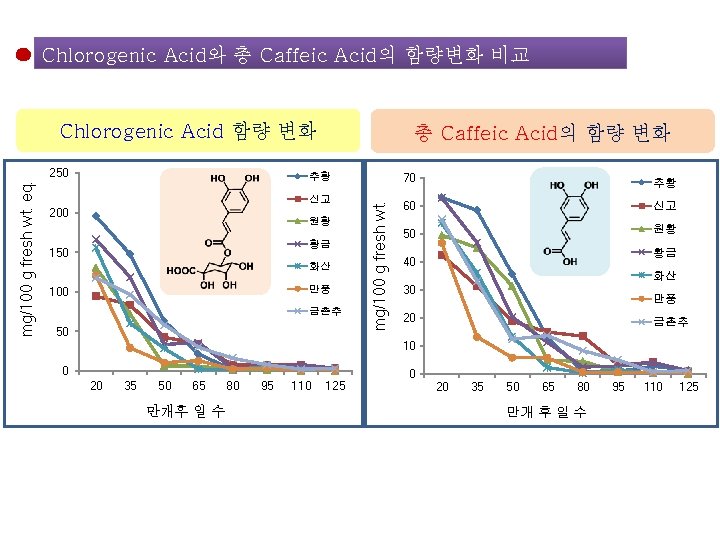
Chlorogenic Acid와 총 Caffeic Acid의 함량변화 비교 Chlorogenic Acid 함량 변화 추황 신고 200 원황 황금 150 화산 만풍 100 금촌추 50 mg/100 g fresh wt. eq. 250 총 Caffeic Acid의 함량 변화 70 추황 60 신고 50 원황 황금 40 화산 30 만풍 20 금촌추 10 0 20 35 50 65 만개후 일 수 80 95 110 125 0 20 35 50 65 80 만개 후 일 수 95 110 125



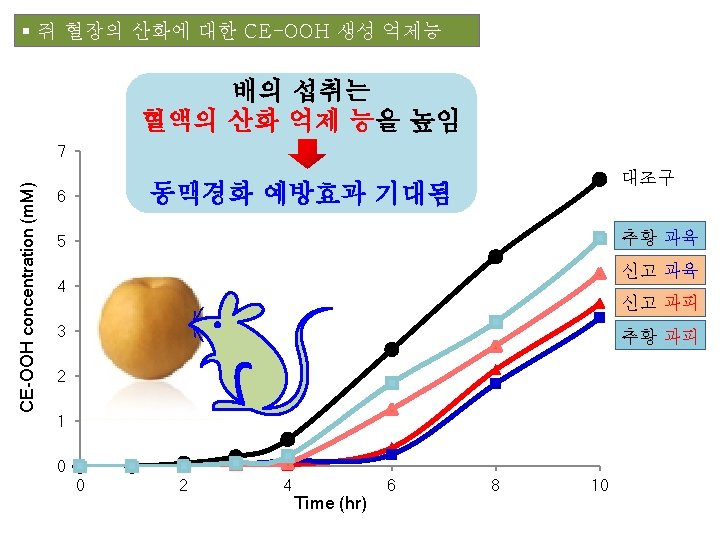
▣ 생리활성 평가 - Total Phenolics Total Flavonoids DPPH radical scavenging activity ABTS+ radical scavenging activity Nitrite scavenging activity Reducing power 산화적 스트레스에 의한 세포생존율 평가 활성 산소종 생성 억제능 경구 투여 혈장 내 CE-OOH 생성 억제능

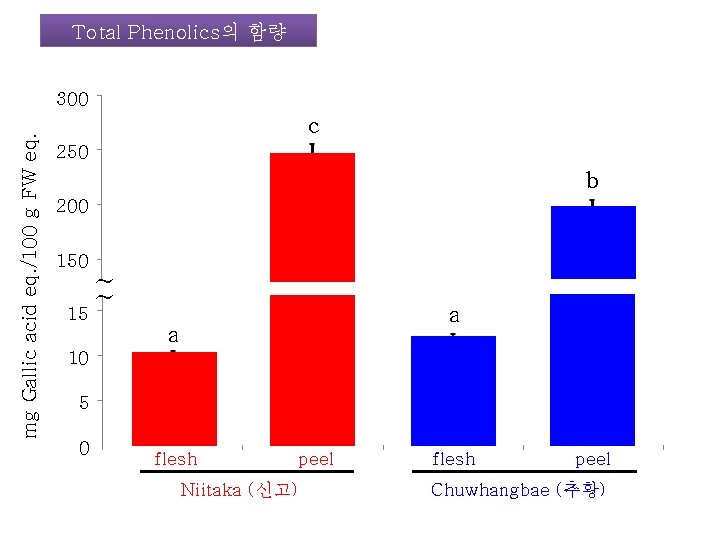
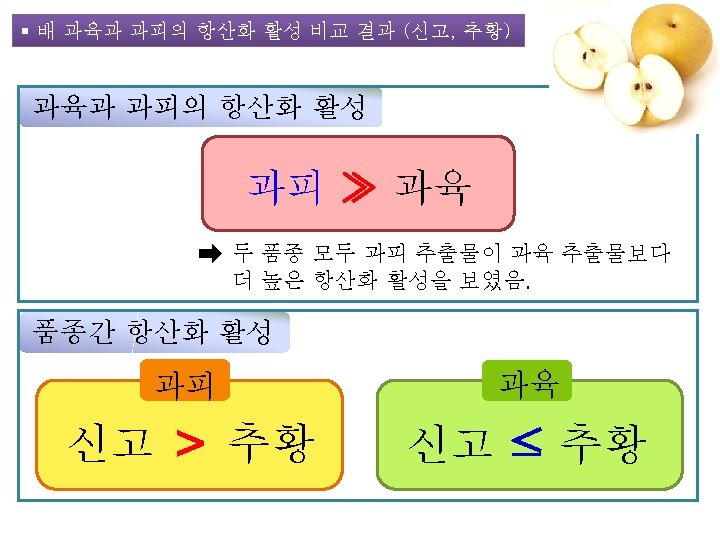
Total Phenolics의 함량 mg Gallic acid eq. /100 g FW eq. 300 c 250 b 200 150 ~ ~ 15 a a 10 5 0 flesh Niitaka (신고) peel flesh peel Chuwhangbae (추황)

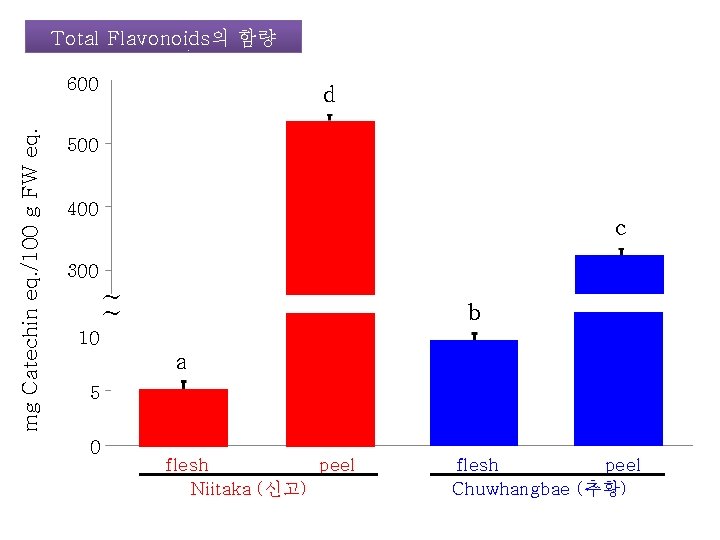
Total Flavonoids의 함량 mg Catechin eq. /100 g FW eq. 600 d 500 400 c 300 ~ ~ b 10 a 5 0 flesh peel Niitaka (신고) flesh peel Chuwhangbae (추황)

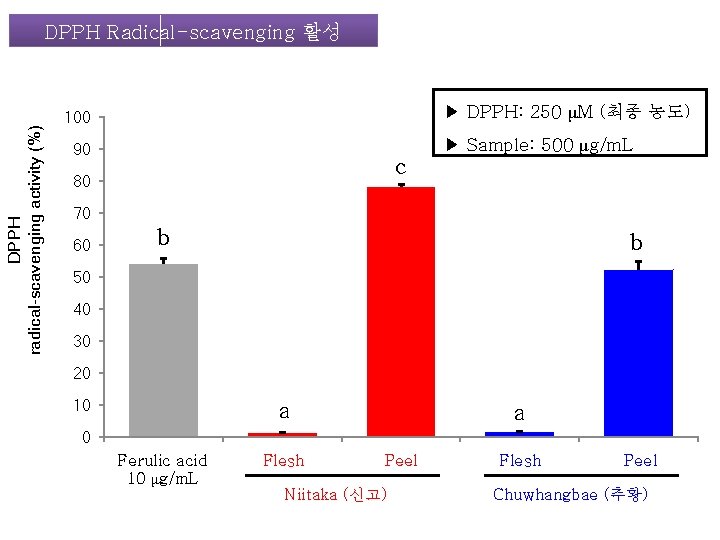
DPPH radical-scavenging activity (%) DPPH Radical-scavenging 활성 ▶ DPPH: 250 μM (최종 농도) 100 90 c 80 ▶ Sample: 500 μg/m. L 70 60 b b 50 40 30 20 10 a a 0 Ferulic acid 10 μg/m. L Flesh Peel Niitaka (신고) Flesh Peel Chuwhangbae (추황)

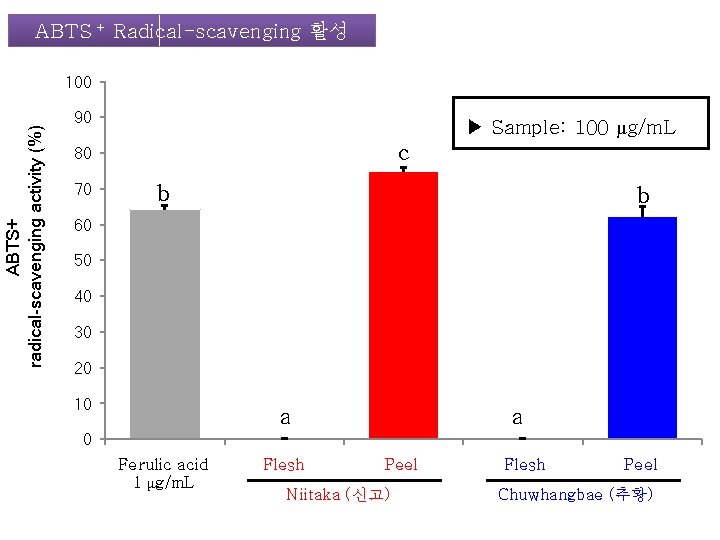
ABTS + Radical-scavenging 활성 ABTS+ radical-scavenging activity (%) 100 90 ▶ Sample: 100 μg/m. L c 80 70 b b 60 50 40 30 20 10 a a 0 Ferulic acid 1 μg/m. L Flesh Peel Niitaka (신고) Flesh Peel Chuwhangbae (추황)

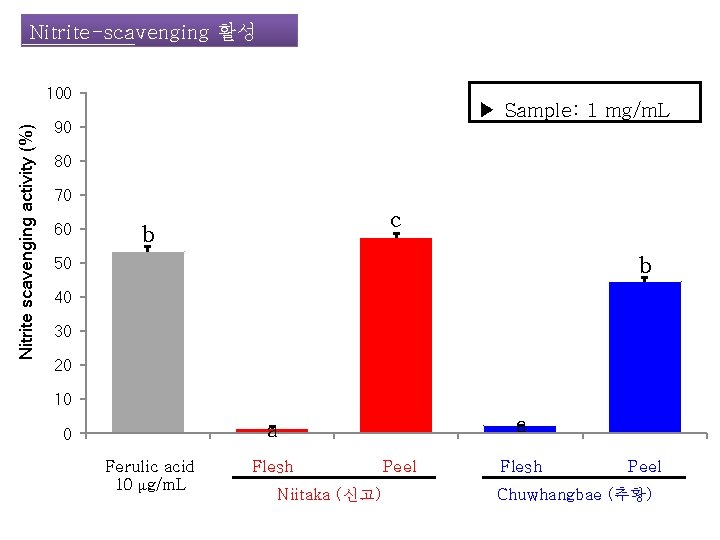
Nitrite-scavenging 활성 Nitrite scavenging activity (%) 100 ▶ Sample: 1 mg/m. L 90 80 70 60 c b b 50 40 30 20 10 a a 0 Ferulic acid 10 μg/m. L Flesh Niitaka (신고) Peel Flesh Peel Chuwhangbae (추황)

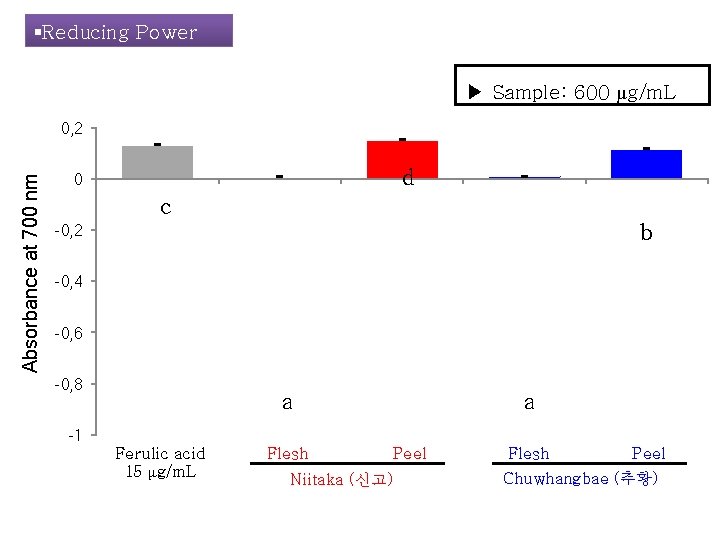
§Reducing Power ▶ Sample: 600 μg/m. L Absorbance at 700 nm 0, 2 d 0 c b -0, 2 -0, 4 -0, 6 -0, 8 a a -1 Ferulic acid 15 μg/m. L Flesh Peel Niitaka (신고) Flesh Peel Chuwhangbae (추황)

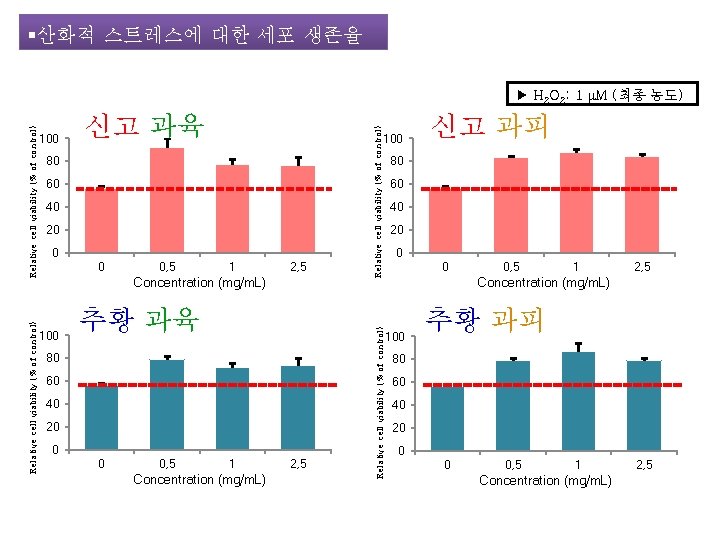
§산화적 스트레스에 대한 세포 생존율 신고 과육 80 60 40 20 0 0 0, 5 1 2, 5 Concentration (mg/m. L) 100 추황 과육 80 60 40 20 0 0 0, 5 1 Concentration (mg/m. L) 2, 5 Relative cell viability (% of control) 100 Relative cell viability (% of control) ▶ H 2 O 2: 1 μM (최종 농도) 신고 과피 80 60 40 20 0 0 0, 5 1 2, 5 Concentration (mg/m. L) 100 추황 과피 80 60 40 20 0 0 0, 5 1 Concentration (mg/m. L) 2, 5

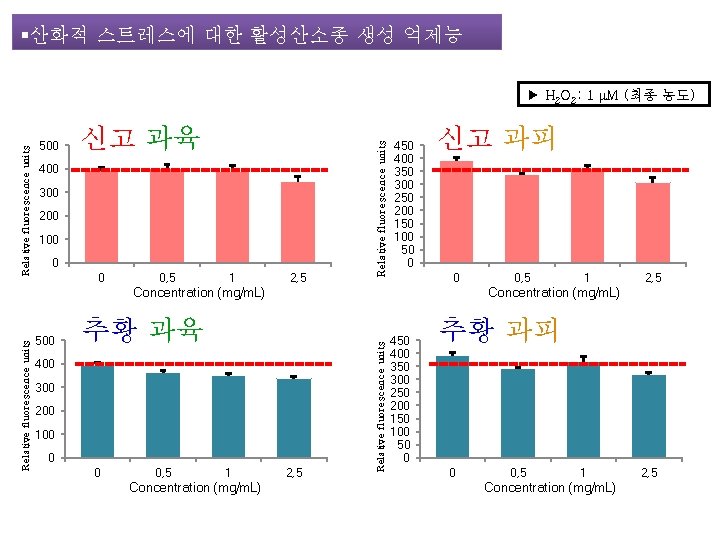
§산화적 스트레스에 대한 활성산소종 생성 억제능 500 신고 과육 400 300 200 100 0 0 0, 5 1 2, 5 Relative fluorescence units ▶ H 2 O 2: 1 μM (최종 농도) 450 400 350 300 250 200 150 100 50 0 신고 과피 0 500 추황 과육 400 300 200 100 0 0 0, 5 1 Concentration (mg/m. L) 1 2, 5 Concentration (mg/m. L) 2, 5 Relative fluorescence units Concentration (mg/m. L) 0, 5 450 400 350 300 250 200 150 100 50 0 추황 과피 0 0, 5 1 Concentration (mg/m. L) 2, 5



LC-MS를 이용한 배 함유성분의 Chemical Profile 작성 The Analysis of Sample Using UPLC/SYNAPT G 2 -HDMS Q-TOF










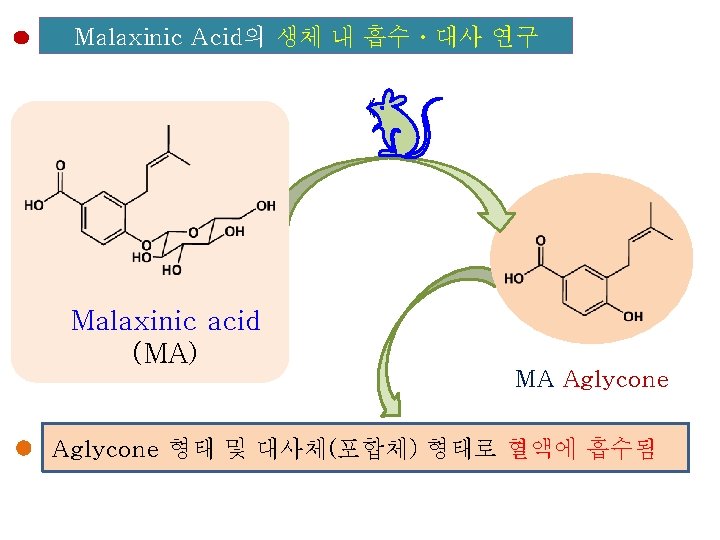
Malaxinic Acid의 흡수·대사



Malaxinic Acid의 생체 내 흡수ㆍ대사 연구 Malaxinic acid (MA) MA Aglycone

Malaxinic Acid 및 그 Aglycone의 합성

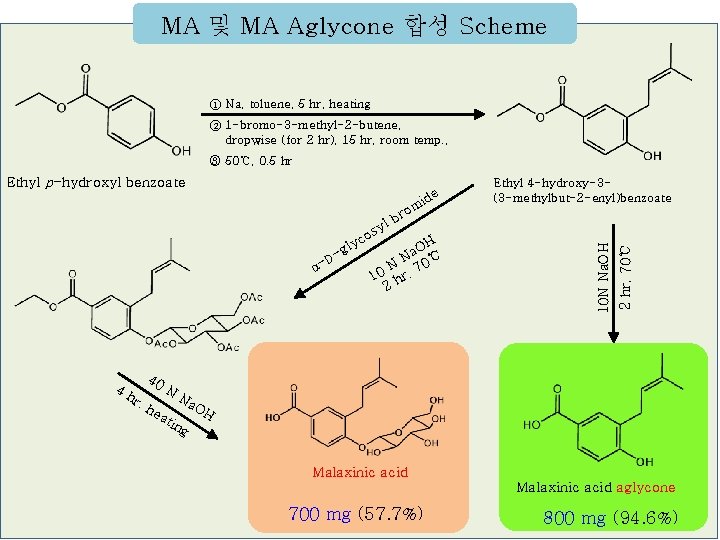
MA 및 MA Aglycone 합성 Scheme ① Na, toluene, 5 hr, heating ② 1 -bromo-3 -methyl-2 -butene, dropwise (for 2 hr), 15 hr, room temp. , ③ 50℃, 0. 5 hr Ethyl p-hydroxyl benzoate om 40 4 h r, he N ati y OH a N 0℃ N 7 10 hr, 2 2 hr, 70℃ D α- Ethyl 4 -hydroxy-3(3 -methylbut-2 -enyl)benzoate 10 N Na. OH -g os lyc r lb ide Na ng OH Malaxinic acid aglycone 700 mg (57. 7%) 800 mg (94. 6%)


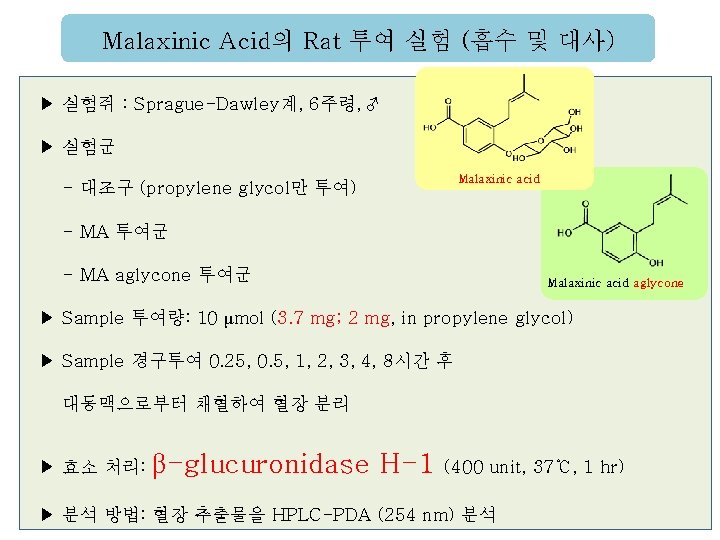
Malaxinic Acid의 Rat 투여 실험 (흡수 및 대사) ▶ 실험쥐 : Sprague-Dawley계, 6주령, ♂ ▶ 실험군 Malaxinic acid - 대조구 (propylene glycol만 투여) - MA 투여군 - MA aglycone 투여군 Malaxinic acid aglycone ▶ Sample 투여량: 10 μmol (3. 7 mg; 2 mg, in propylene glycol) ▶ Sample 경구투여 0. 25, 0. 5, 1, 2, 3, 4, 8시간 후 대동맥으로부터 채혈하여 혈장 분리 ▶ 효소 처리: β-glucuronidase H-1 (400 unit, 37℃, 1 hr) ▶ 분석 방법: 혈장 추출물을 HPLC-PDA (254 nm) 분석


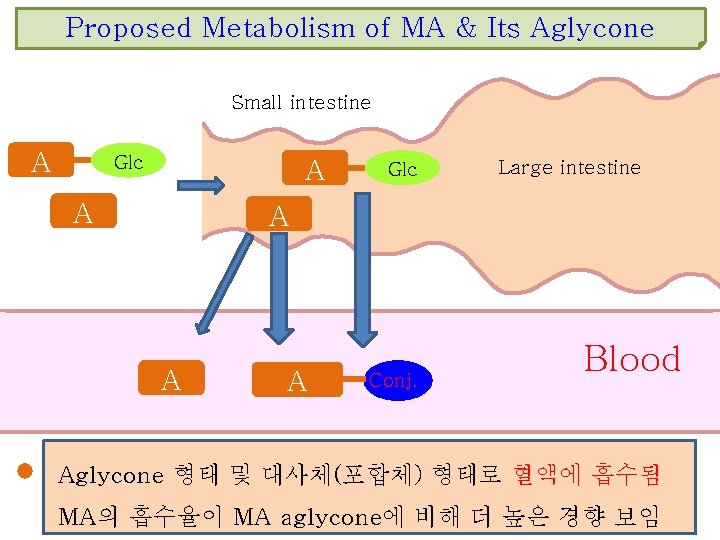
Proposed Metabolism of MA & Its Aglycone Small intestine A Glc A A Glc Large intestine A A A Conj. Blood Aglycone 형태 및 대사체(포합체) 형태로 혈액에 흡수됨 MA의 흡수율이 MA aglycone에 비해 더 높은 경향 보임

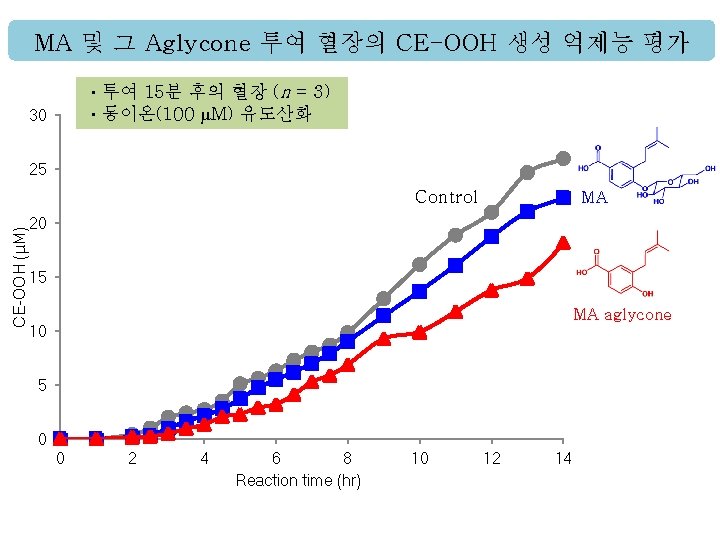
MA 및 그 Aglycone 투여 혈장의 CE-OOH 생성 억제능 평가 ㆍ투여 15분 후의 혈장 (n = 3) ㆍ동이온(100 µM) 유도산화 30 채 25 CE-OOH (μM) Control MA 20 15 MA aglycone 10 5 0 0 2 4 6 8 Reaction time (hr) 10 12 14






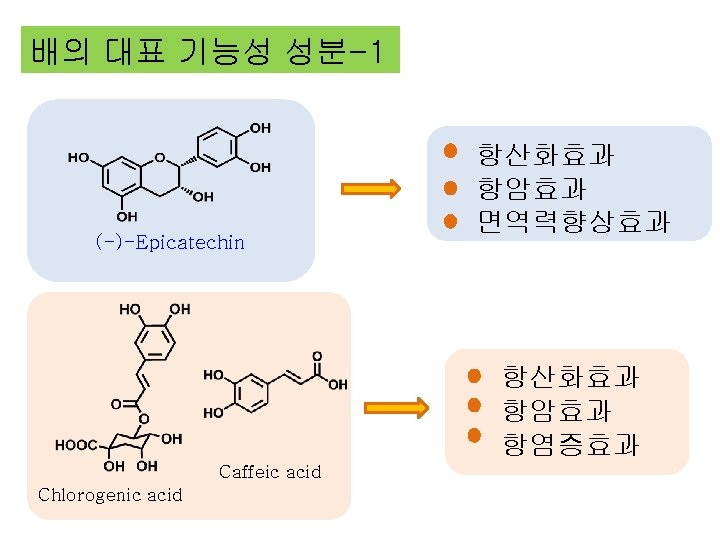
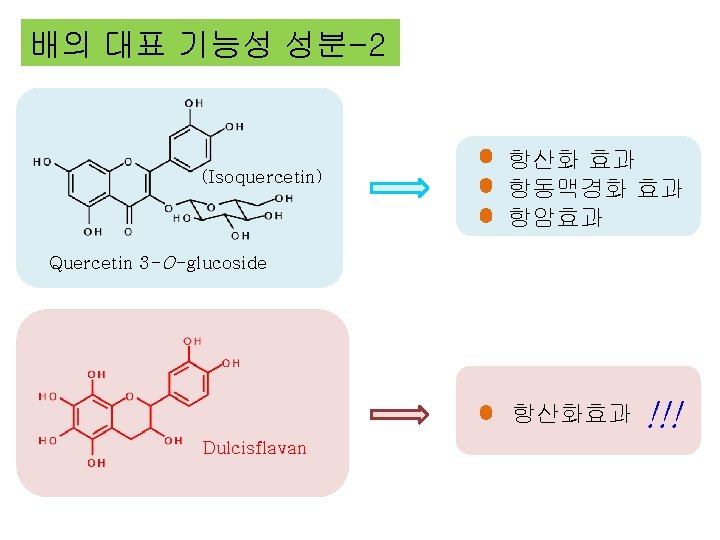
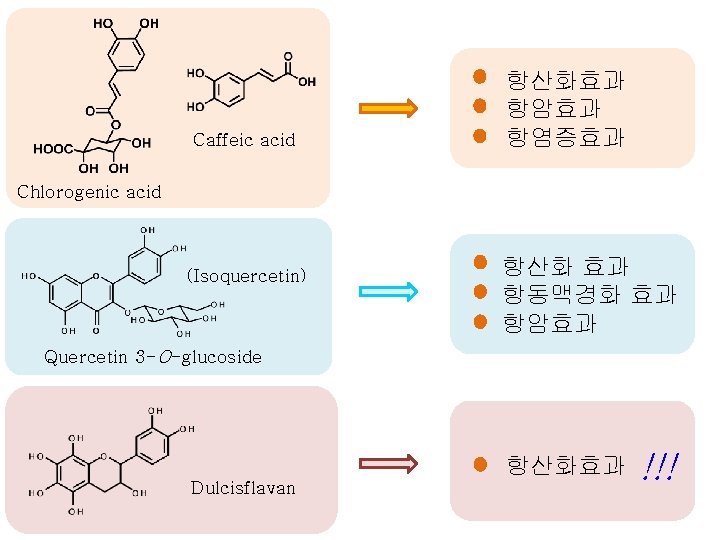
Caffeic acid 항산화효과 항암효과 항염증효과 Chlorogenic acid (Isoquercetin) 항산화 효과 항동맥경화 효과 항암효과 Quercetin 3 -O-glucoside 항산화효과 Dulcisflavan !!!

Betulinic acid Lupeol cis- Betulin aldehyde trans- Caffeoyl betulinic acid β-Sitosterol 3 -O-Caffeoyloleanolic acid 항염증 항균 (헬리코박터) 콜레스테롤 흡수억제 작용 항 암 항인플루엔자 혈청 콜레스테롤 농도 저하 항산화



