PTT 155 Aldehydes and Ketones JOHAN ARIFF MOHTAR

















- Slides: 49

PTT 155 Aldehydes and Ketones JOHAN ARIFF MOHTAR johanariff@unimap. edu. my

Course Outcomes Involved n Ability to APPLY the structure, nomenclature and naming of aldehydes and ketones n Ability to ILLUSTRATE and PROPOSE the reaction of aldehydes and ketones n Ability to ANALYZE the knowledge or organic chemistry in the chemical process industry especially in aldehydes and ketones 2

Outlines n Naming of Aldehydes and Ketones n Relative Reactivity of Carbonyl Group n Nucleophilc Addition Reaction n n n n Water H-Y HCN Gignard Reagent Hydride Amines Hydrazines Alcohols Phosphorus Ylid n Oxidation Reaction of Aldehydes and Ketones 3

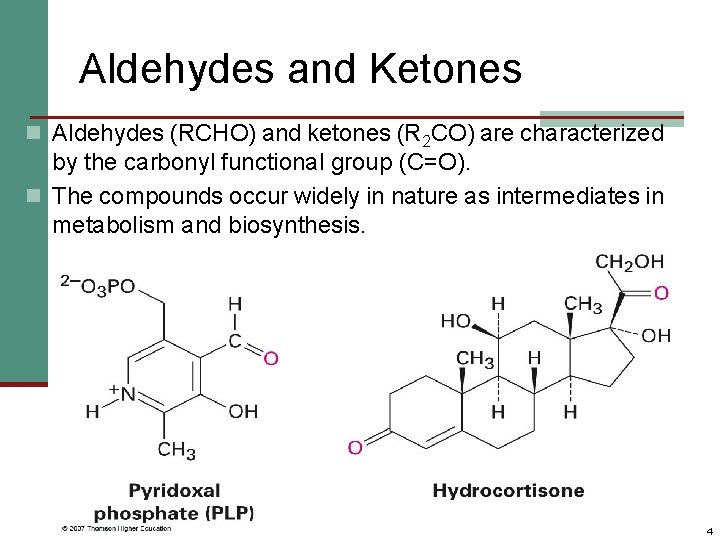
Aldehydes and Ketones n Aldehydes (RCHO) and ketones (R 2 CO) are characterized by the carbonyl functional group (C=O). n The compounds occur widely in nature as intermediates in metabolism and biosynthesis. 4

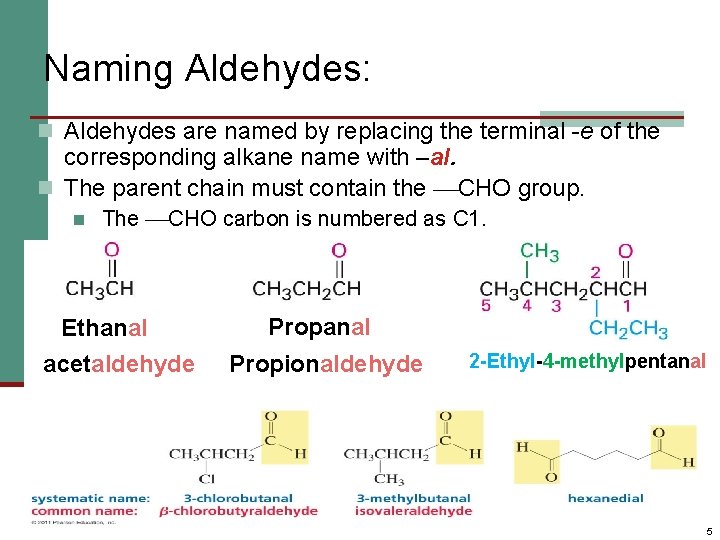
Naming Aldehydes: n Aldehydes are named by replacing the terminal -e of the corresponding alkane name with –al. n The parent chain must contain the CHO group. n The CHO carbon is numbered as C 1. Ethanal acetaldehyde Propanal Propionaldehyde 2 -Ethyl-4 -methylpentanal 5

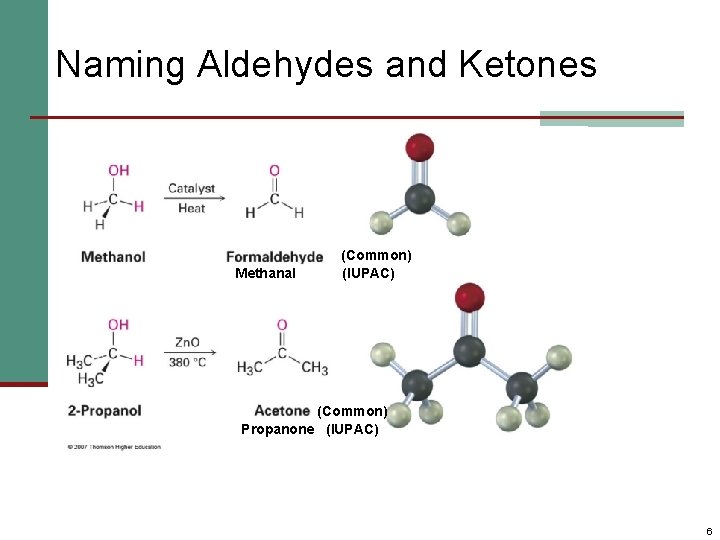
Naming Aldehydes and Ketones Methanal (Common) (IUPAC) (Common) Propanone (IUPAC) 6

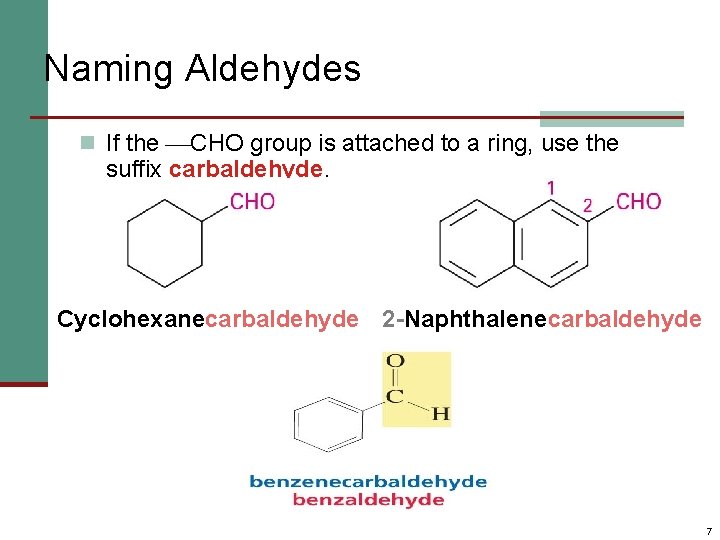
Naming Aldehydes n If the CHO group is attached to a ring, use the suffix carbaldehyde. Cyclohexanecarbaldehyde 2 -Naphthalenecarbaldehyde 7

Naming Aldehydes: n Common Names end in aldehyde 8

Naming Ketones n Replace the terminal -e of the alkane name with –one. n Parent chain is the longest one that contains the ketone group. n Numbering begins at the end nearer the carbonyl carbon (smaller number). 9

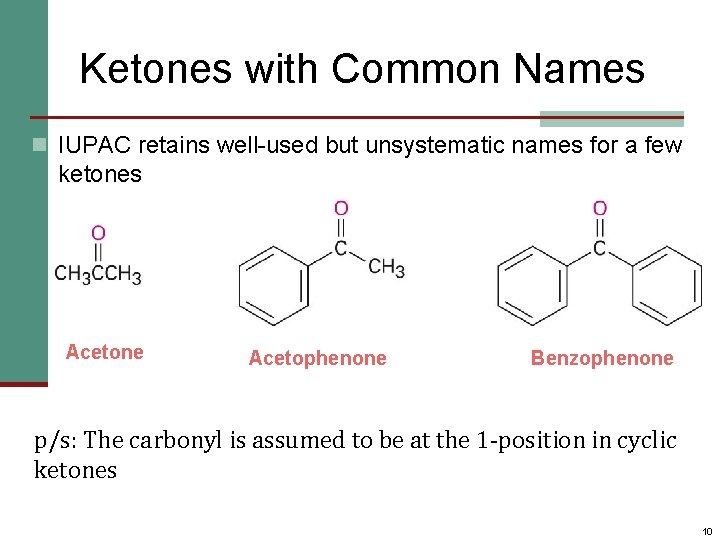
Ketones with Common Names n IUPAC retains well-used but unsystematic names for a few ketones Acetone Acetophenone Benzophenone p/s: The carbonyl is assumed to be at the 1 -position in cyclic ketones 10

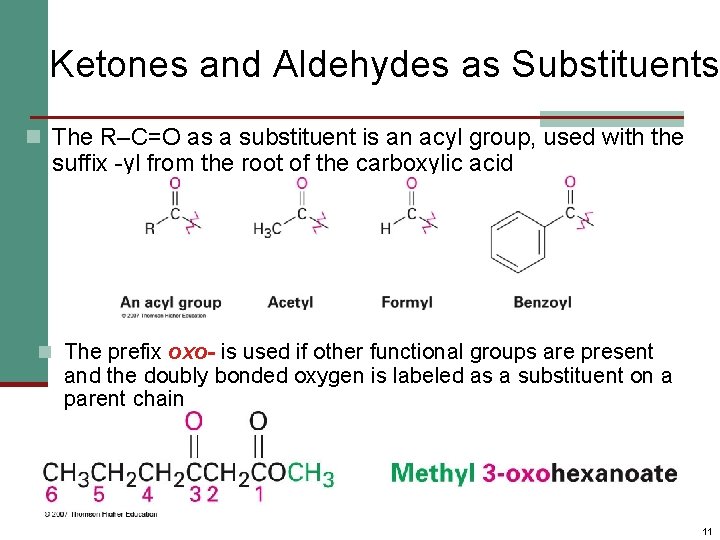
Ketones and Aldehydes as Substituents n The R–C=O as a substituent is an acyl group, used with the suffix -yl from the root of the carboxylic acid n The prefix oxo- is used if other functional groups are present and the doubly bonded oxygen is labeled as a substituent on a parent chain 11

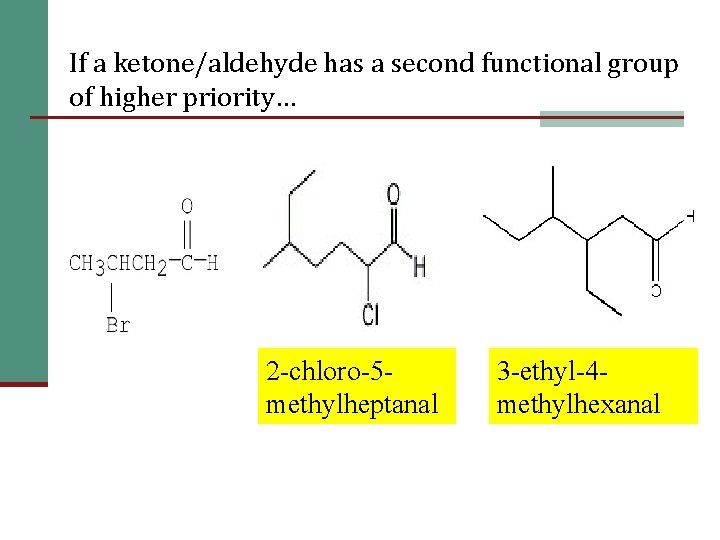
12 If a ketone/aldehyde has a second functional group of higher priority… 2 -chloro-5 methylheptanal 3 -ethyl-4 methylhexanal

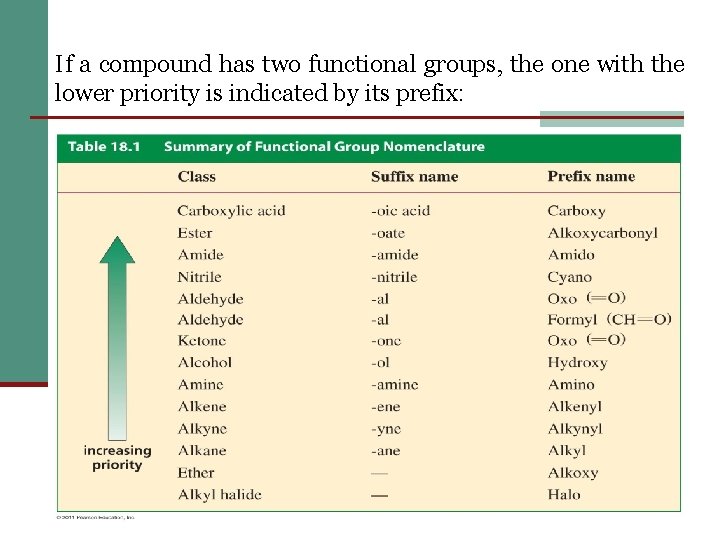
13 If a compound has two functional groups, the one with the lower priority is indicated by its prefix:

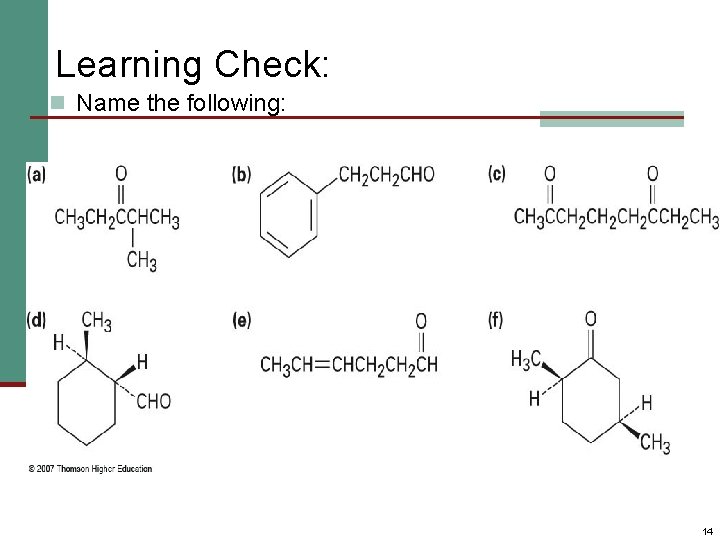
Learning Check: n Name the following: 14

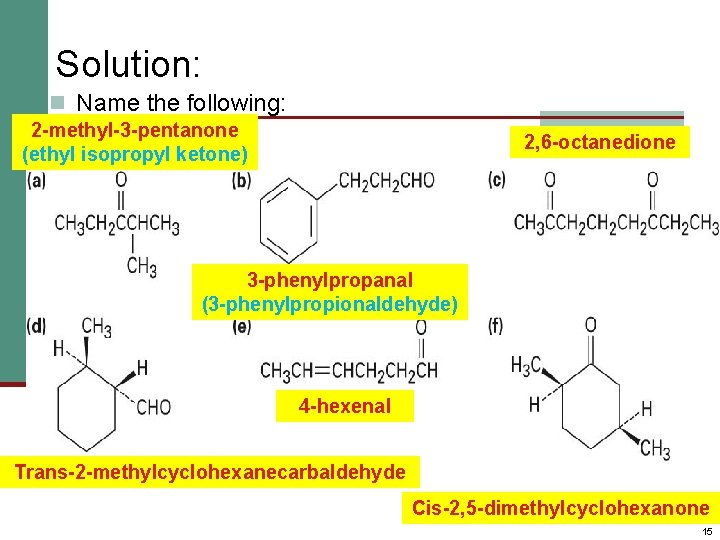
Solution: n Name the following: 2 -methyl-3 -pentanone (ethyl isopropyl ketone) 2, 6 -octanedione 3 -phenylpropanal (3 -phenylpropionaldehyde) 4 -hexenal Trans-2 -methylcyclohexanecarbaldehyde Cis-2, 5 -dimethylcyclohexanone 15

Physical Properties Have higher boiling points than hydrocarbon because they are more polar and the forces between molecules are stronger. They have lower boiling point than alcohols? , why? They are more soluble than hydrocarbons but less soluble than alcohols in water.

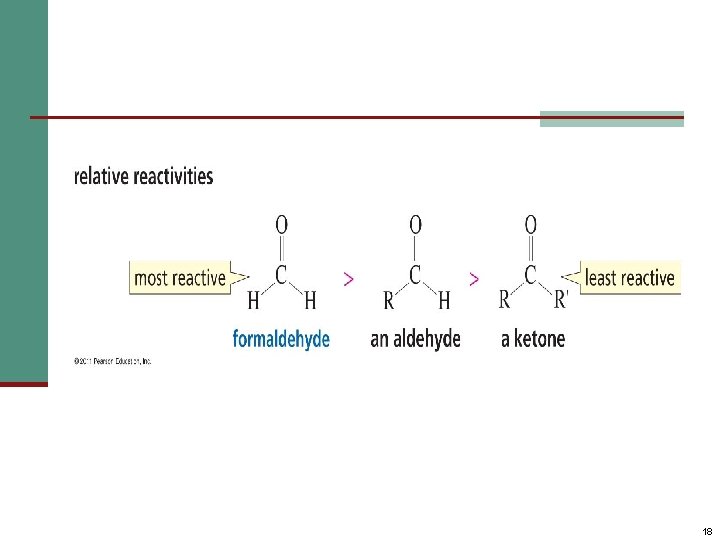
The Relative Reactivity of Carbonyl Group n The partial positive charge on the carbonyl carbon causes that carbon to be attacked by nucleophiles: n An aldehyde has a greater partial positive charge on its carbonyl carbon than does a ketone: 17

18

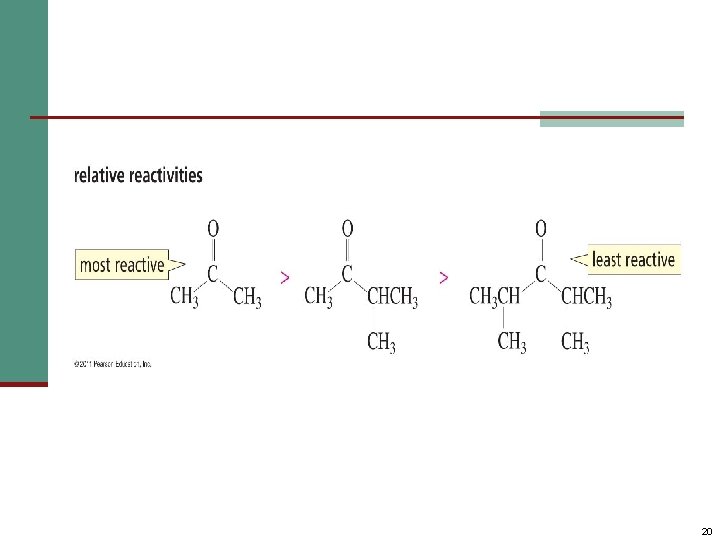
Aldehydes Are More Reactive Than Ketones n Steric factors contribute to the reactivity of an aldehyde. • The carbonyl carbon of an aldehyde is more accessible to the nucleophile. • Ketones have greater steric crowding in their transition states, so they have less stable transition states. 19

20

n The reactivity of carbonyl compounds is also related to the basicity of Y–: 21

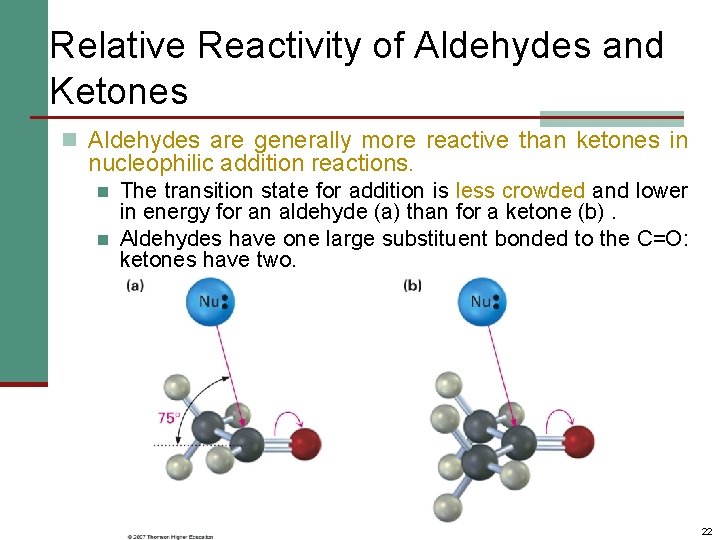
Relative Reactivity of Aldehydes and Ketones n Aldehydes are generally more reactive than ketones in nucleophilic addition reactions. n n The transition state for addition is less crowded and lower in energy for an aldehyde (a) than for a ketone (b). Aldehydes have one large substituent bonded to the C=O: ketones have two. 22

Relative Reactivity of Aldehydes and Ketones n Aldehydes are generally more reactive than ketones in nucleophilic addition reactions. n n Aldehyde C=O is more polarized than ketone C=O. Ketone has more electron donation alkyl groups, stabilizing the C=O carbon inductively. 23

How aldehydes and ketones react? 24

Nucleophilic Addition Reactions of Aldehydes and Ketones n Nu- approaches 75° to the plane of C=O and adds to C n A tetrahedral alkoxide ion intermediateis produced 25

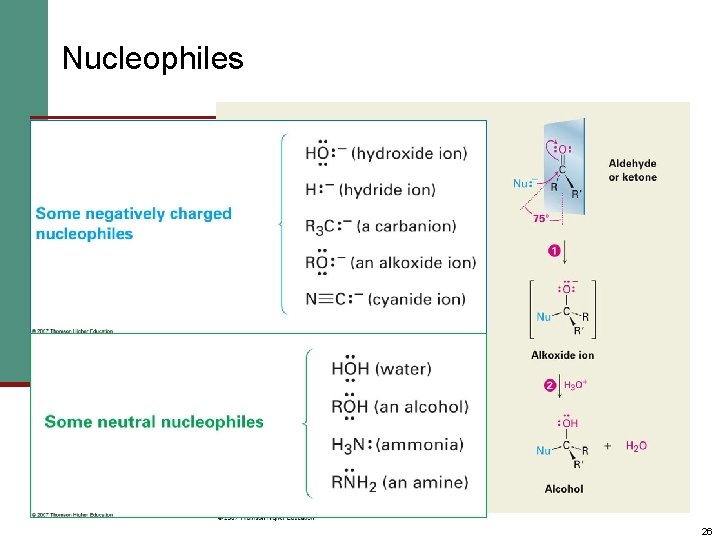
Nucleophiles 26

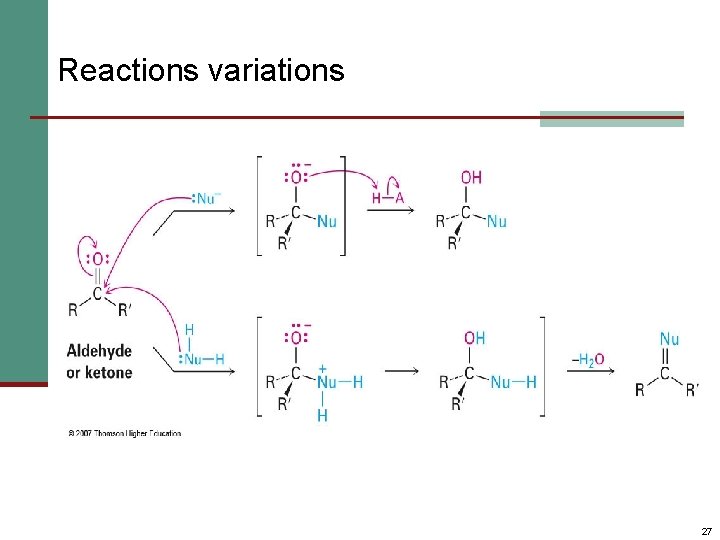
Reactions variations 27

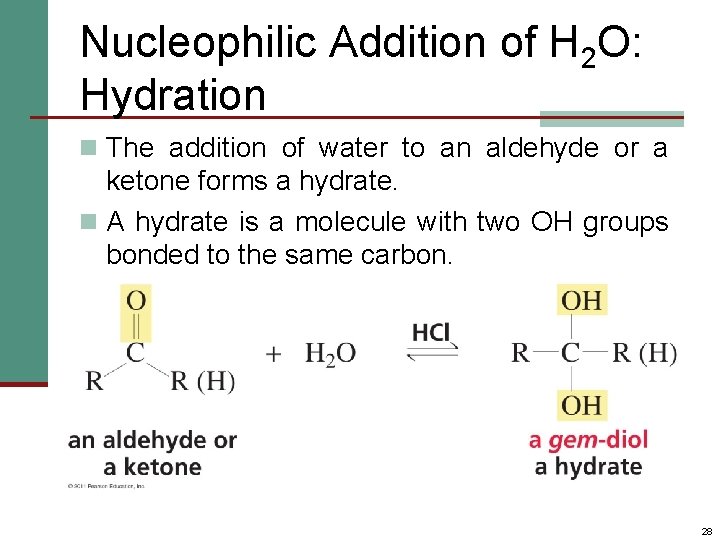
Nucleophilic Addition of H 2 O: Hydration n The addition of water to an aldehyde or a ketone forms a hydrate. n A hydrate is a molecule with two OH groups bonded to the same carbon. 28

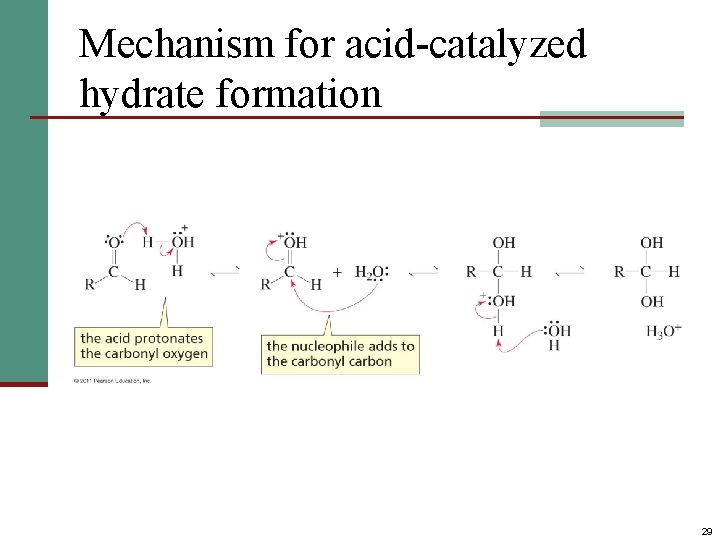
Mechanism for acid-catalyzed hydrate formation 29

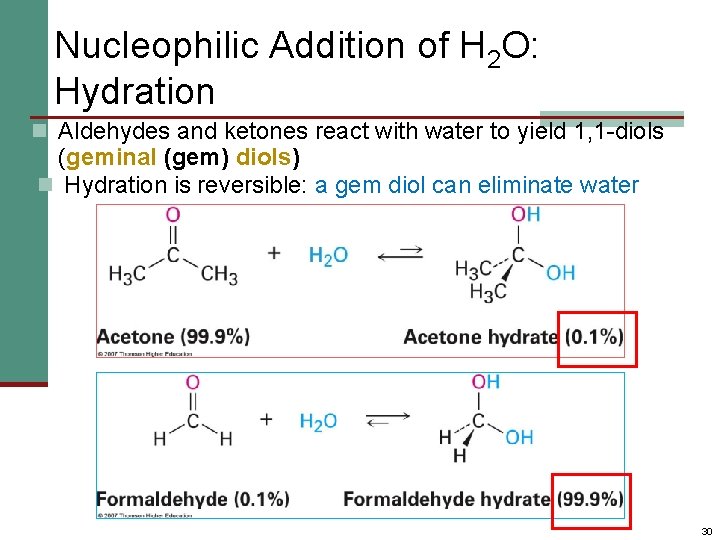
Nucleophilic Addition of H 2 O: Hydration n Aldehydes and ketones react with water to yield 1, 1 -diols (geminal (gem) diols) n Hydration is reversible: a gem diol can eliminate water 30

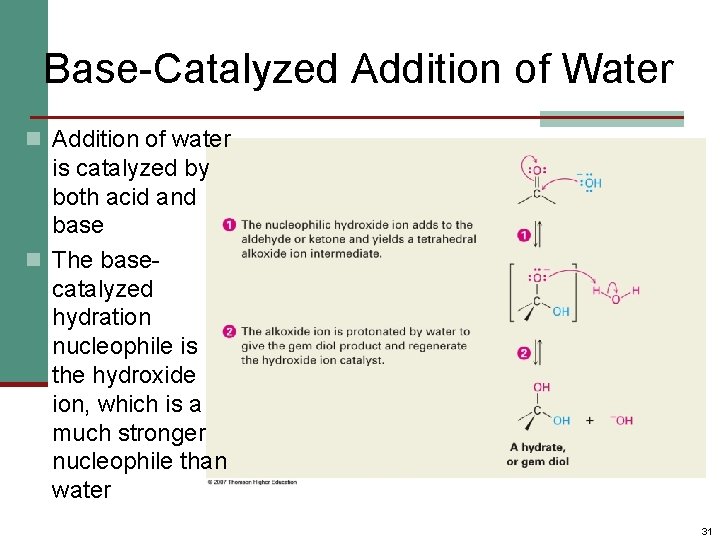
Base-Catalyzed Addition of Water n Addition of water is catalyzed by both acid and base n The basecatalyzed hydration nucleophile is the hydroxide ion, which is a much stronger nucleophile than water 31

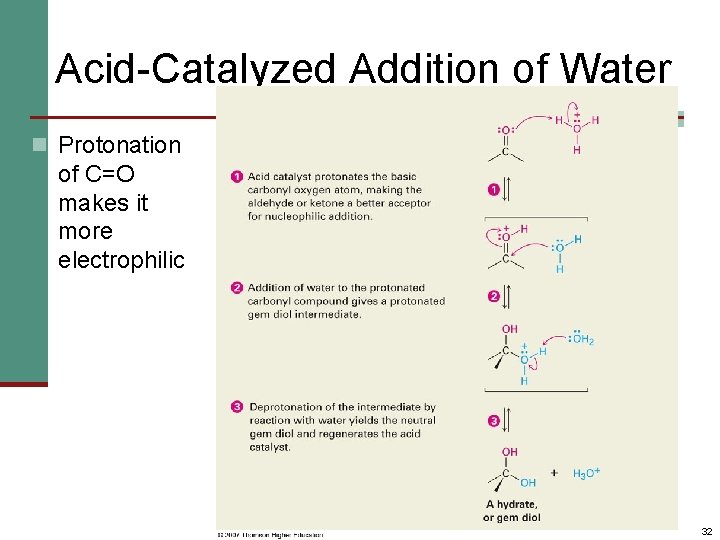
Acid-Catalyzed Addition of Water n Protonation of C=O makes it more electrophilic 32

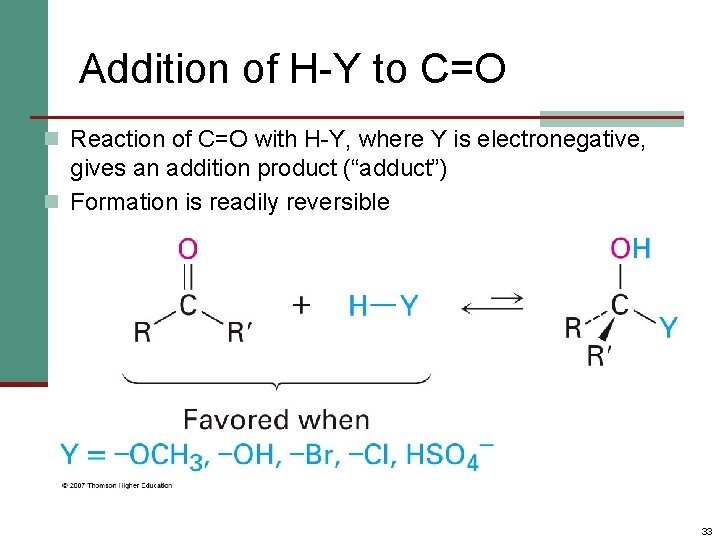
Addition of H-Y to C=O n Reaction of C=O with H-Y, where Y is electronegative, gives an addition product (“adduct”) n Formation is readily reversible 33

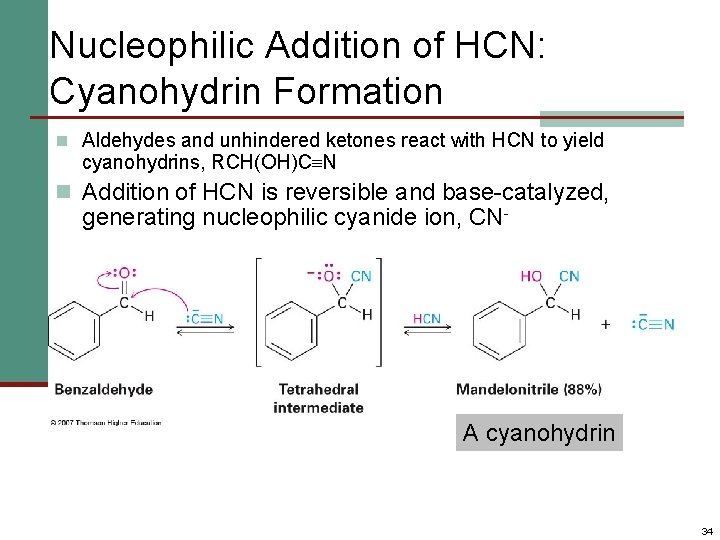
Nucleophilic Addition of HCN: Cyanohydrin Formation n Aldehydes and unhindered ketones react with HCN to yield cyanohydrins, RCH(OH)C N n Addition of HCN is reversible and base-catalyzed, generating nucleophilic cyanide ion, CN- A cyanohydrin 34

Nucleophilic Addition of Grignard Reagents and Hydride Reagents: Alcohol Formation n Grignard reagents react with aldehydes, ketones, and carboxylic acid derivatives. 35

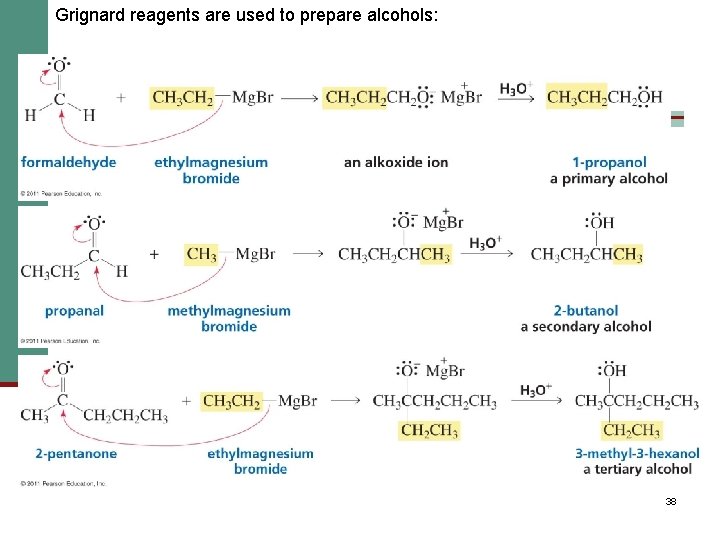
Nucleophilic Addition of Grignard Reagents and Hydride Reagents: Alcohol Formation n Treatment of aldehydes or ketones with Grignard reagents yields an alcohol n Nucleophilic addition of the equivalent of a carbon anion, or carbanion. A carbon–magnesium bond is strongly polarized, so a Grignard reagent reacts for all practical purposes as R : Mg. X +. 36

Mechanism of Addition of Grignard Reagents n Complexation of C=O by Mg 2+, Nucleophilic addition of R : , protonation by dilute acid yields the neutral alcohol n Grignard additions are irreversible because a carbanion is not a leaving group 37

Grignard reagents are used to prepare alcohols: 38

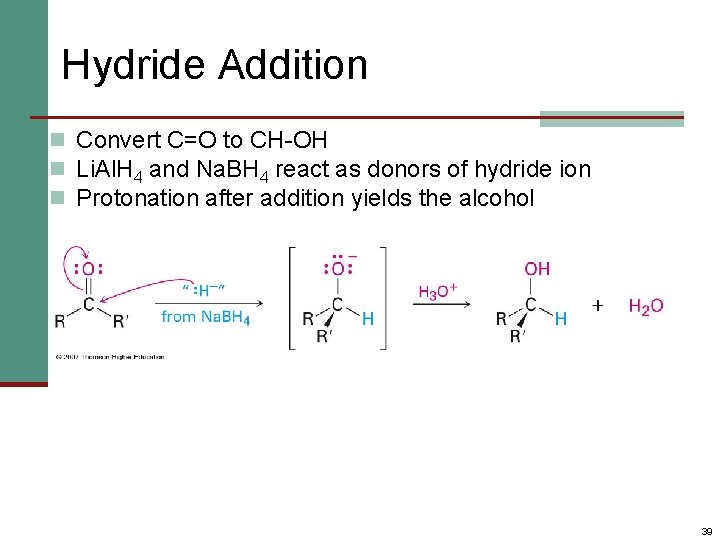
Hydride Addition n Convert C=O to CH-OH n Li. Al. H 4 and Na. BH 4 react as donors of hydride ion n Protonation after addition yields the alcohol 39

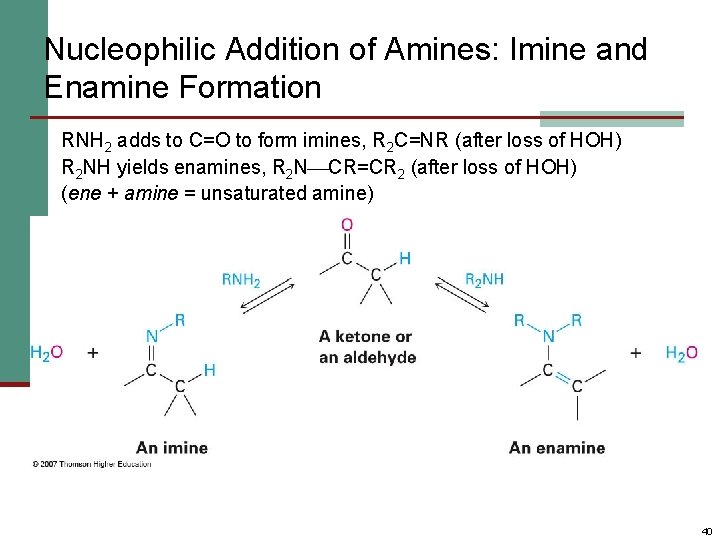
Nucleophilic Addition of Amines: Imine and Enamine Formation RNH 2 adds to C=O to form imines, R 2 C=NR (after loss of HOH) R 2 NH yields enamines, R 2 N CR=CR 2 (after loss of HOH) (ene + amine = unsaturated amine) 40

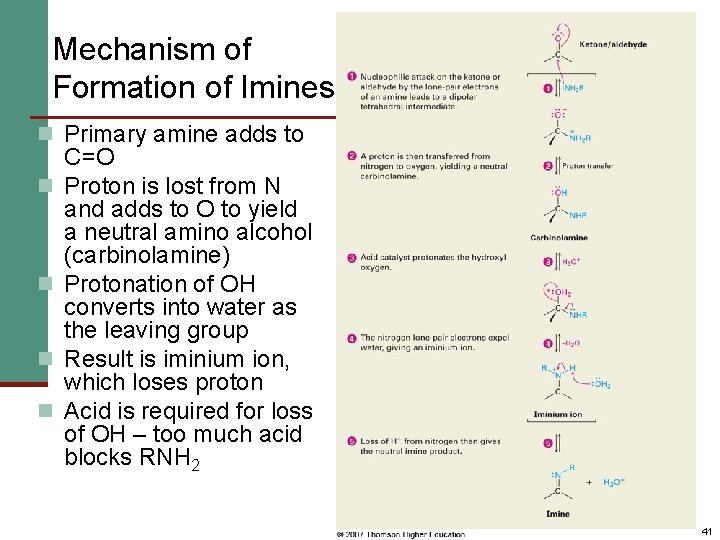
Mechanism of Formation of Imines n Primary amine adds to n n C=O Proton is lost from N and adds to O to yield a neutral amino alcohol (carbinolamine) Protonation of OH converts into water as the leaving group Result is iminium ion, which loses proton Acid is required for loss of OH – too much acid blocks RNH 2 41

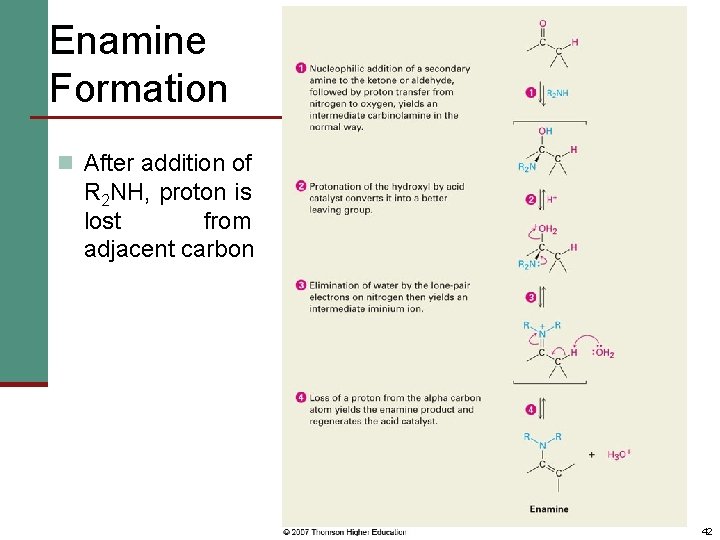
Enamine Formation n After addition of R 2 NH, proton is lost from adjacent carbon 42

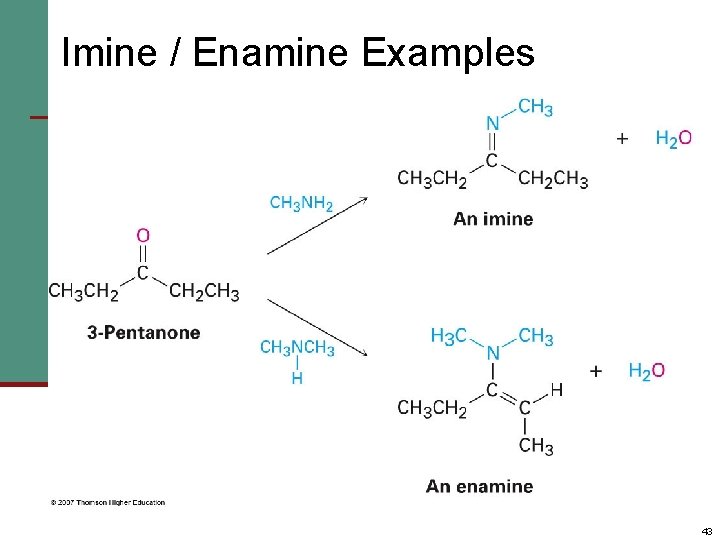
Imine / Enamine Examples 43

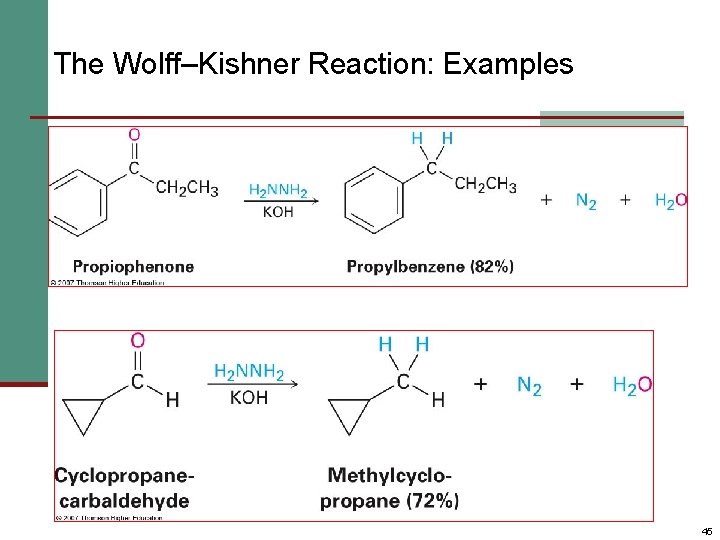
Nucleophilic Addition of Hydrazine: The Wolff –Kishner Reaction n Treatment of an aldehyde or ketone with hydrazine, H 2 NNH 2 and KOH converts the compound to an alkane n Originally carried out at high temperatures but with dimethyl sulfoxide as solvent takes place near room temperature 44

The Wolff–Kishner Reaction: Examples 45

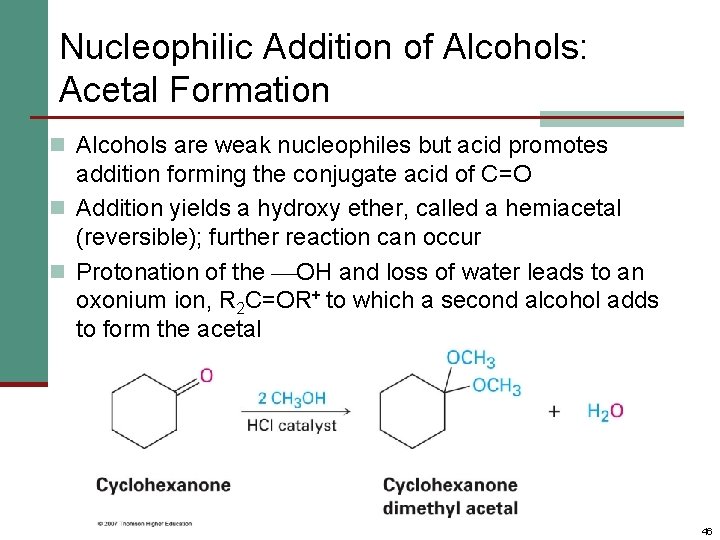
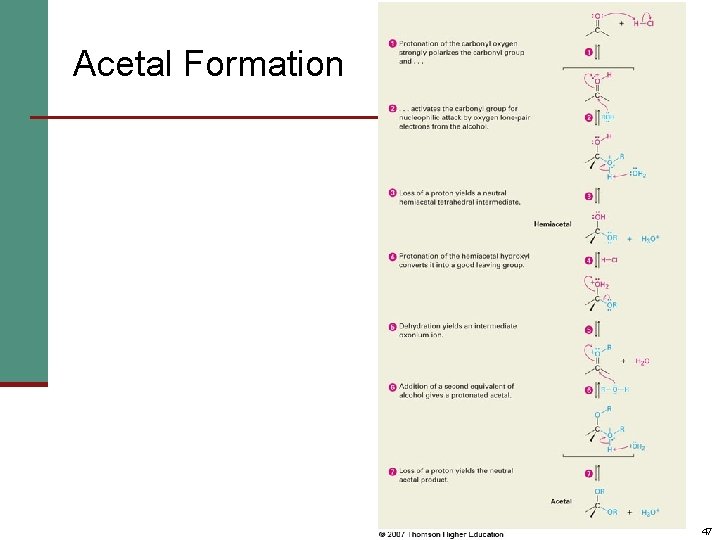
Nucleophilic Addition of Alcohols: Acetal Formation n Alcohols are weak nucleophiles but acid promotes addition forming the conjugate acid of C=O n Addition yields a hydroxy ether, called a hemiacetal (reversible); further reaction can occur n Protonation of the OH and loss of water leads to an oxonium ion, R 2 C=OR+ to which a second alcohol adds to form the acetal 46

Acetal Formation 47

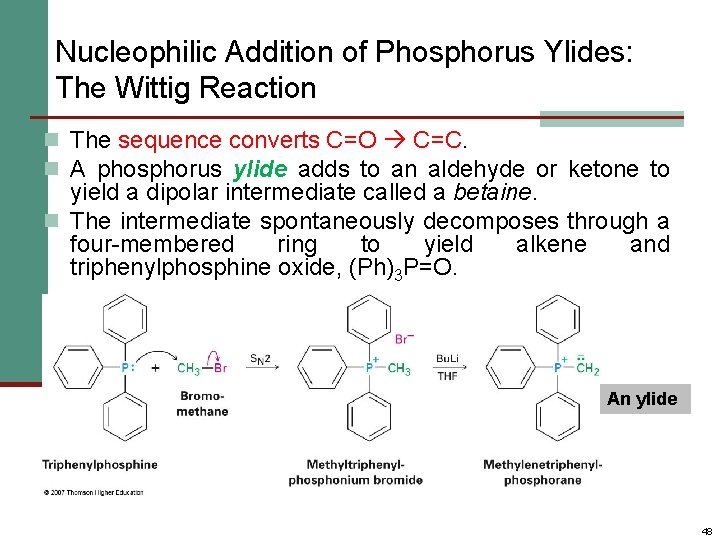
Nucleophilic Addition of Phosphorus Ylides: The Wittig Reaction n The sequence converts C=O C=C. n A phosphorus ylide adds to an aldehyde or ketone to yield a dipolar intermediate called a betaine. n The intermediate spontaneously decomposes through a four-membered ring to yield alkene and triphenylphosphine oxide, (Ph)3 P=O. An ylide 48

Mechanism of the Wittig Reaction 49