Heat and Temperature Heat is a form of



























- Slides: 35

Heat and Temperature.

Heat is a form of energy and is measured in Joules.

Scientists use the word "thermal" to mean "heat". It does not matter whether you say "heat energy" or thermal energy“ they both mean the same thing.

• Temperature is a measure of how hot something is and is measured in °C(degree Celsius). • Heat is related to temperature but the two are not the same.

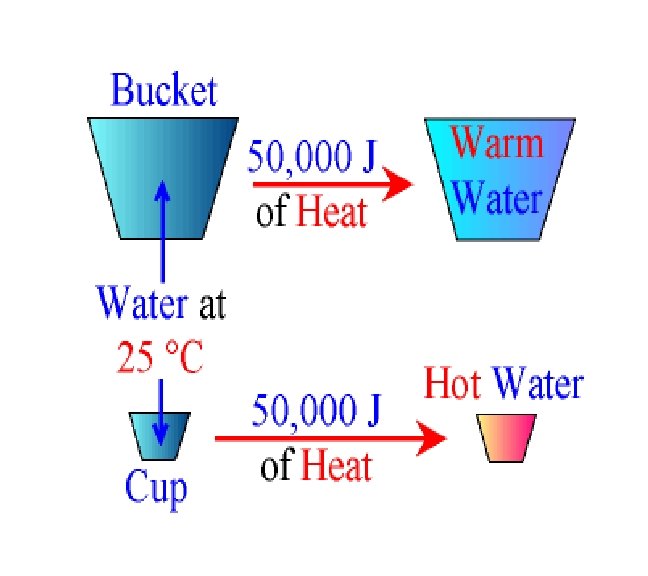
Imagine that you have a bucket full of water and a cup full of water both at 25 °C. If you add the same amount of heat energy (for example 50, 000 J) to both You would find that the temperature of the cup of water increases by much more than the temperature of the bucket of water.


Temperature is a measure of the kinetic energy of the particles (how fast they are going). Temperature does not depend on the mass of the substance (how many particles there are).

The amount of heat energy which a substance has does depend on its mass. If you double the mass, you must double the heat energy to heat it to the same temperature.

Heat Transfer. Heat is transferred naturally from a substance with a higher temperature to a substance with a lower temperature.

Heat can be transferred in three ways. 1. Conduction – by a substance which does not move (solids). 2. Convection – by a substance which moves (liquids and gasses). 3. Radiation – infra-red radiation is exchanged between all substances.

Heat - Conduction. • Heat can be transferred by conduction only in solids. • If one end of a solid is heated, the particles of the solid gain kinetic energy. • This means that they move faster.

Heat - Conduction. • In a solid the particles are held together by strong forces of attraction. The only way in which the particles can move is to vibrate forwards and backwards.

Heat - Conduction. When the solid is heated, the amount by which the particles vibrate is increased. This is what is meant by saying that the particles of the solid have gained kinetic energy. The increase in energy (heat) is passed on to the next particle, which in turn starts to vibrate more.

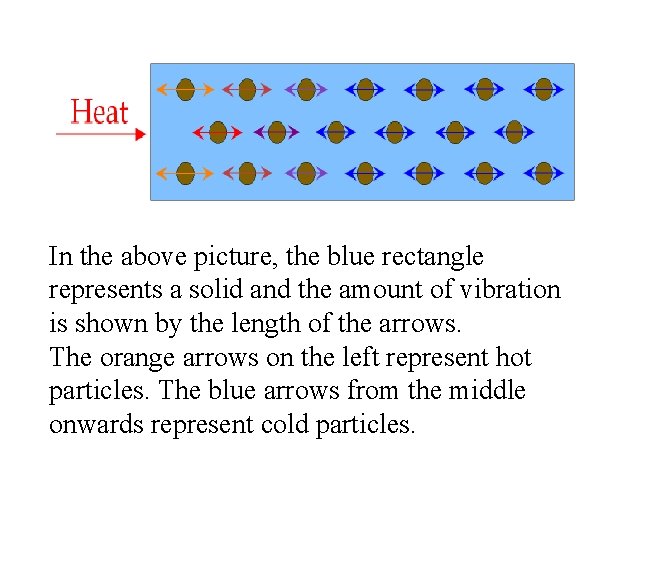
In the above picture, the blue rectangle represents a solid and the amount of vibration is shown by the length of the arrows. The orange arrows on the left represent hot particles. The blue arrows from the middle onwards represent cold particles.

In non-metals the process is slow. It takes a long time for the particles to pass on their heat. Non-metals are not good conductors. They are good insulators We have seen that heat conduction in non-metals is slow. Metals conduct heat quickly.

Metals have ions which are surrounded by free electrons. These free electrons can travel quickly and easily throughout the structure. The electrons transfer the heat energy by colliding with other atoms and electrons in the metal.

In the above picture, the blue lines represent free electrons and the red balls are metal atoms. It is the presence of free electrons which makes metals good conductors of electricity as well as heat.

Heat - Convection. • Heat can be transferred by convection in liquids and gases. Particles in the liquid or gas collide with a substance which has a high temperature and gain kinetic energy (heat).

The particles move to a region which has substance at a lower temperature and transfer heat by colliding with the colder substance. In this way, the high temperature substance has heat taken away from it and gets colder. The low temperature substance has heat given to it and gets warmer.

The liquid or gas which transfers the heat can circulate round and round between the hot and cold regions. The flow of liquid or gas is called a convection current. Convection currents are caused by changes in density

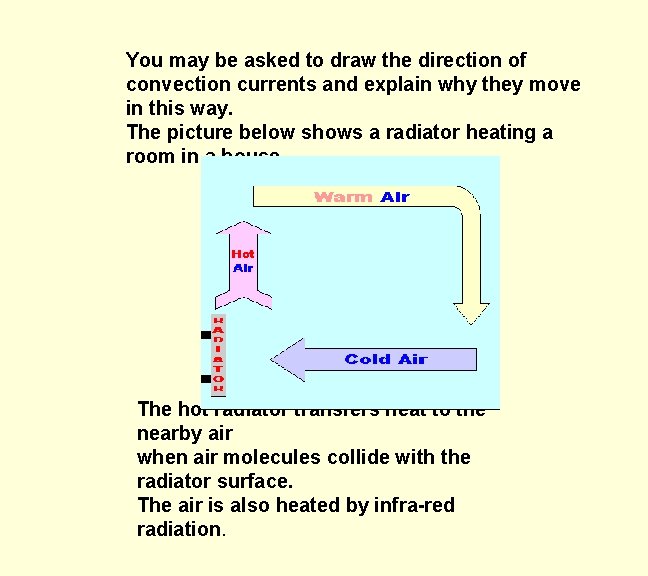
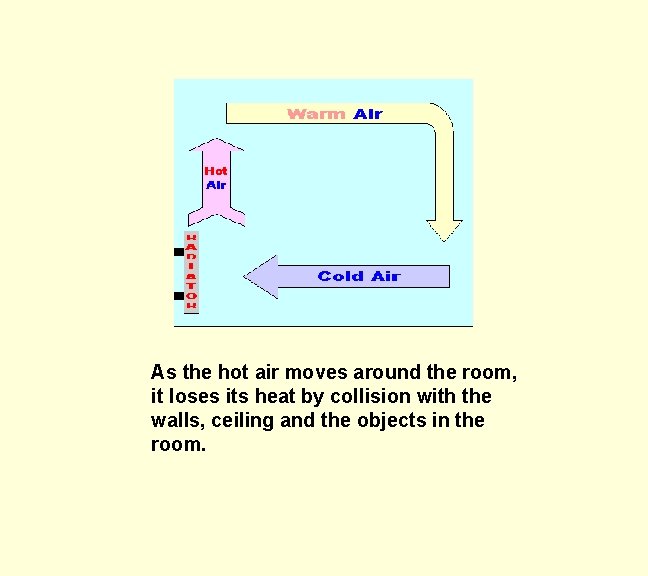
You may be asked to draw the direction of convection currents and explain why they move in this way. The picture below shows a radiator heating a room in a house. The hot radiator transfers heat to the nearby air when air molecules collide with the radiator surface. The air is also heated by infra-red radiation.

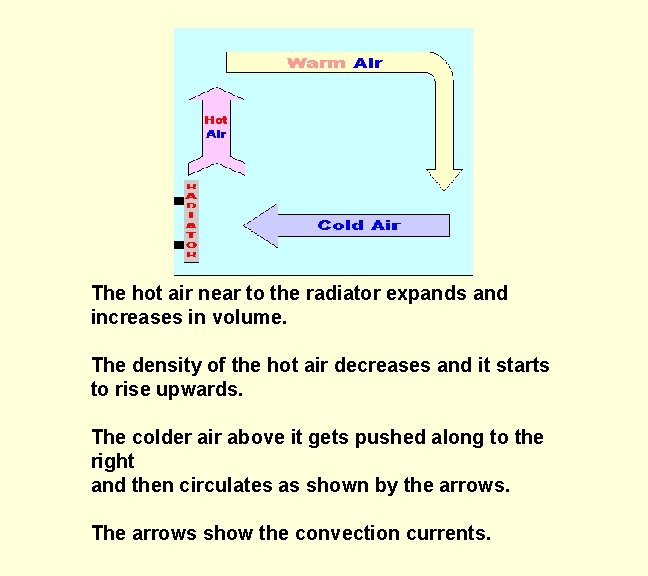
The hot air near to the radiator expands and increases in volume. The density of the hot air decreases and it starts to rise upwards. The colder air above it gets pushed along to the right and then circulates as shown by the arrows. The arrows show the convection currents.

As the hot air moves around the room, it loses its heat by collision with the walls, ceiling and the objects in the room.

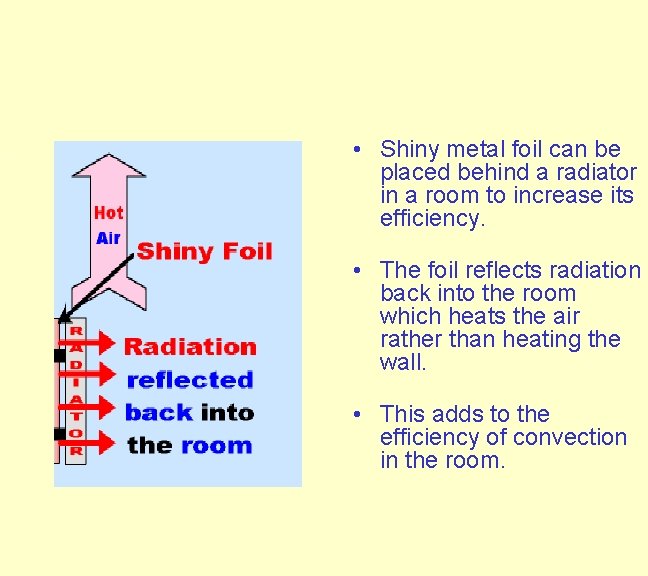
Finally the colder air circulates near to the radiator where it is heated and the whole process repeats itself. The efficiency of convection can be improved by placing shiny metal foil behind the radiator.

Heat - Radiation.

Infra-red radiation transfers heat between all objects. Infra-red radiation is an electromagnetic wave and can travel through a vacuum. Heat from the Sun reaches us through the vacuum of space by traveling as infrared radiation.

An object can absorb (take in), emit (give out) and reflect radiation. A hot object will emit more infra-red radiation than it absorbs. A cold object will absorb more infra-red radiation than it emits. In this way heat is transferred from hotter to colder objects. An object whose temperature does not change will emit infra-red radiation at the same rate as it is absorbed.

Objects which are at the same temperature as each other will absorb, emit and reflect infra-red radiation at different rates depending on the type of surface which the object has.

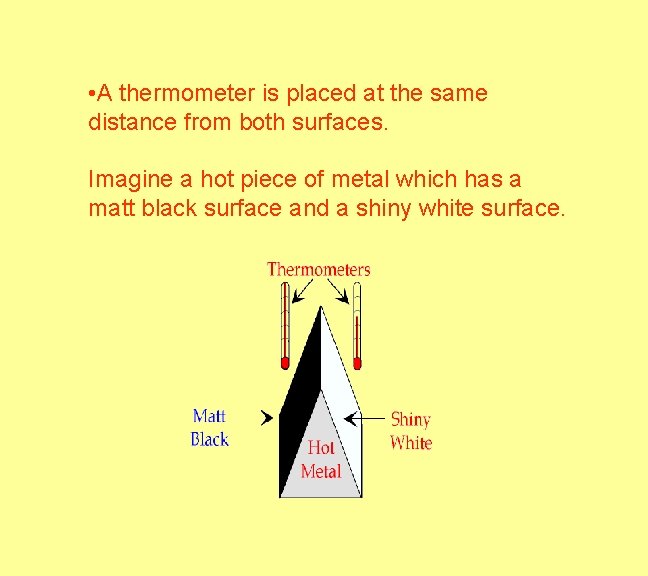
Heat - Radiation - Surfaces Absorption and Emission An object with a matt (dull) surface will absorb and emit infra-red radiation at a faster rate than an object with a shiny surface. An object with a dark surface will absorb and emit infra-red radiation at a faster rate than an object with a light surface. Radiation is also reflected at the surface.

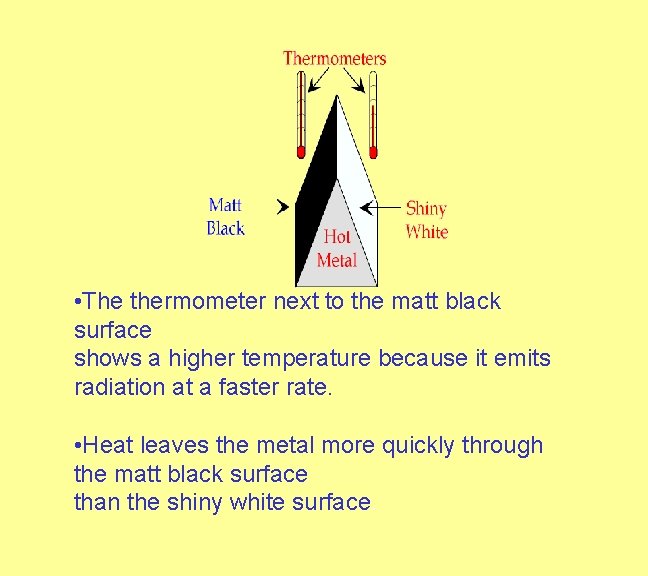
• A thermometer is placed at the same distance from both surfaces. Imagine a hot piece of metal which has a matt black surface and a shiny white surface.

• The thermometer next to the matt black surface shows a higher temperature because it emits radiation at a faster rate. • Heat leaves the metal more quickly through the matt black surface than the shiny white surface.

• Part of the space shuttle is covered in matt black tiles. These help the craft to lose heat from its surface • when it re-enters the Earth's atmosphere on returning from space. The space shuttle gets very hot in the Earth's atmosphere in the same way that meteors do.

Heat - Radiation - Surfaces - Reflection • When an amount of infra-red radiation falls on an object, some will be reflected and some will be absorbed. • The greater the proportion of radiation which is reflected, the less will be absorbed.

• An object with a matt or dark surface will be a poor reflector of infra-red radiation. • An object with a shiny or light surface will be a good reflector of infra-red radiation.

. • Shiny metal foil can be placed behind a radiator in a room to increase its efficiency. • The foil reflects radiation back into the room which heats the air rather than heating the wall. • This adds to the efficiency of convection in the room.