Temperature and Heat Objectives Temperature Gas Laws Heat

Temperature and Heat Objectives: • • Temperature Gas Laws Heat and Heat Transfer Thermal Expansion

Temperature Related to how fast atoms/molecules are moving (Kinetic Energy) Fahrenheit (F) Celsius (C) Centigrade Kelvin (K)
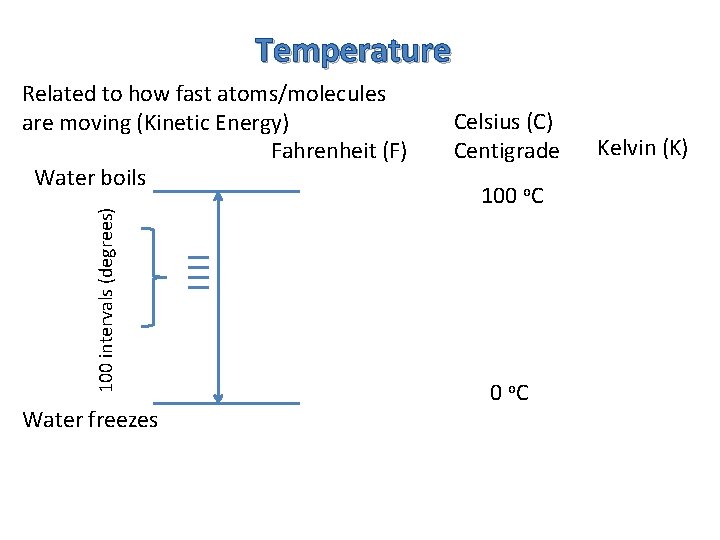
Temperature 100 intervals (degrees) Related to how fast atoms/molecules are moving (Kinetic Energy) Fahrenheit (F) Water boils Water freezes Celsius (C) Centigrade 100 o. C Kelvin (K)
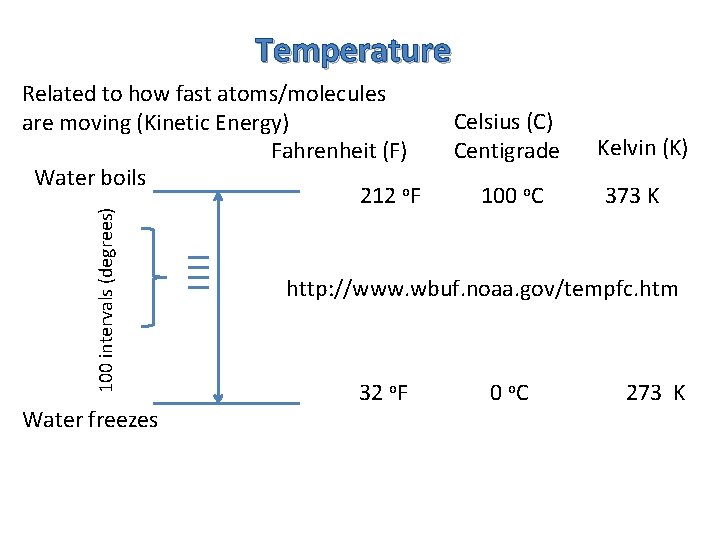
Temperature 100 intervals (degrees) Related to how fast atoms/molecules are moving (Kinetic Energy) Fahrenheit (F) Water boils 212 o. F Water freezes Celsius (C) Centigrade 100 o. C Kelvin (K) 373 K http: //www. wbuf. noaa. gov/tempfc. htm 32 o. F 0 o. C 273 K
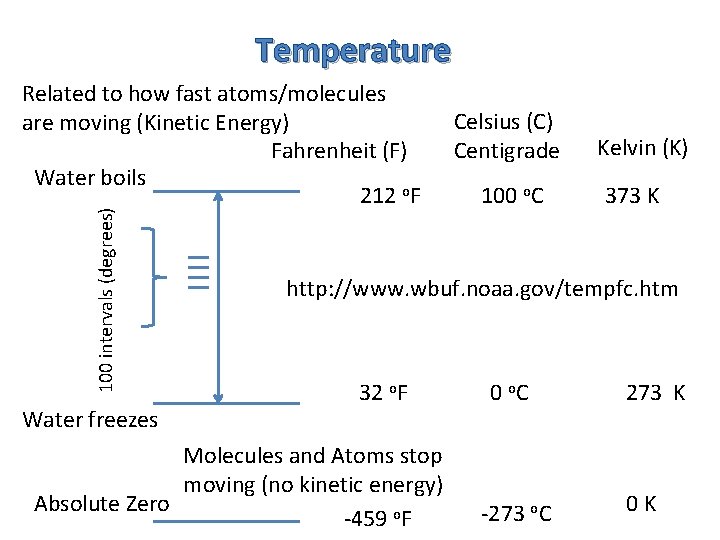
Temperature 100 intervals (degrees) Related to how fast atoms/molecules are moving (Kinetic Energy) Fahrenheit (F) Water boils 212 o. F Water freezes Celsius (C) Centigrade 100 o. C Kelvin (K) 373 K http: //www. wbuf. noaa. gov/tempfc. htm 32 o. F Molecules and Atoms stop moving (no kinetic energy) Absolute Zero -459 o. F 0 o. C -273 o. C 273 K 0 K

Boyle’s Law Temperature remains constant! (Pressure)1 (Volume)1= (Pressure)2 (Volume)2 Q 1. How can we maintain constant temperature?

Boyle’s Law Temperature remains constant! (Pressure)1 (Volume)1= (Pressure)2 (Volume)2 Q 1. How can we maintain constant temperature? Ans. Something happens slow! It exchanges heat with the surrounding environment and maintains that temperature Q 2. You measure the volume of a gas to be 12 m. L at 0. 01 PSI. Then you let the gas expand slowly to 18 m. L. What would be the pressure? (Pressure)1 (Volume)1= (Pressure)2 (Volume)2

Gay Lussac Law Volume remains constant! (Pressure)1 (Temperature)1 = (Pressure)2 (Temperature)2 Q 3. How can we maintain constant volume?

Charles’ Law Charle’s Law Volume remains constant! (Pressure)1 (Temperature)1 (Pressure)2 = (Temperature)2 Q 3. How can we maintain constant volume? Ans. Sealed container Charle’s Law Requires Kelvin (no Celsius! No Fahrenheit!) Q 4. The pressure at 300 K is 0. 02 PSI. What is the pressure at 600 K? (Pressure)2 (Pressure)1 = (Temperature)1 (Temperature)2

Gay Lussac Law Charle’s Law Volume remains constant! (Pressure)1 (Temperature)1 = (Pressure)2 (Temperature)2 Q 3. How can we maintain constant volume? Ans. Sealed container Charle’s Law Requires Kelvin (no Celsius! No Fahrenheit!) Q 4. The pressure at 300 K is 0. 02 PSI. What is the pressure at 600 K? (Pressure)2 (0. 02 PSI)1 = (300 K)1 (600 K)2

Temperature and Gas Laws P 1. What is 70 F in Centigrade? Ans. P 2. You seal a gas in a container. If you double the temperature of the gas, what happens to the pressure inside? Ans. P 3. In the Boyle’s Law (keeping temperature constant), if you triple the pressure, what happens to the volume of the gas. Ans. P 4. For a gas sealed in a container, you measure its temperature and pressure to be 300 K and 0. 2 PSI. What happens to the pressure if you increase the temperature to 350 K? Ans. 20

Heat and Heat Transfer 1 BTU Heat required to warm 1 lb water by 1 F at sea level. 1 cal Heat required to warm 1 g water by 1 o. C 1 Cal Heat required to warm 1000 g water by 1 o. C Q 5. How many calories are necessary to heat 50 g of water by 1 o. C

Heat and Heat Transfer 1 BTU Heat required to warm 1 lb water by 1 F at sea level. 1 cal Heat required to warm 1 g water by 1 o. C 1 Cal Heat required to warm 1000 g water by 1 o. C Q 5. How many calories are necessary to heat 50 g of water by 1 o. C (Heat) = (50 g) (1 cal) Q 6. How many calories are necessary to heat 50 g of water by 10 o. C?

Heat and Heat Transfer 1 BTU Heat required to warm 1 lb water by 1 F at sea level. 1 cal Heat required to warm 1 g water by 1 o. C 1 Cal Heat required to warm 1000 g water by 1 o. C Q 5. How many calories are necessary to heat 50 g of water by 1 o. C (Heat) = (50 g) (1 cal) Q 6. How many calories are necessary to heat 50 g of water by 10 o. C? (Heat) = (50 g) (1 cal) (10 o. C)

Heat and Heat Transfer 1 BTU Heat required to warm 1 lb water by 1 F at sea level. 1 cal Heat required to warm 1 g water by 1 o. C 1 Cal Heat required to warm 1000 g water by 1 o. C Q 6. How many calories are necessary to heat 50 g of water by 10 o. C (Heat) = (50 g) (1 cal) (10 o. C) Q 7. How many calories are necessary to heat 50 g of iron by 10 o. C? (Heat) = (50 g) (? ? ? cal) (10 o. C)

Heat and Heat Transfer 1 BTU Heat required to warm 1 lb water by 1 F at sea level. 1 cal Heat required to warm 1 g water by 1 o. C 1 Cal Heat required to warm 1000 g water by 1 o. C Q 6. How many calories are necessary to heat 50 g of water by 10 o. C (Heat) = (50 g) (1 cal) (10 o. C) Q 7. How many calories are necessary to heat 50 g of iron by 10 o. C? (Heat) = (50 g) (? ? ? cal) (10 o. C) Specific heat 0. 108 cal/g

Specific Heat (Heat) = (mass) (specific heat) (temperature change) Q 8. How many Btus are required to heat up 5. 0 lb of iron by 10 F?

Specific Heat (Heat) = (mass) (specific heat) (temperature change) Q 8. How many Btus are required to heat up 5. 0 lb of iron by 10 F? (Heat) = (5. 0 lb) (0. 11 Btu/g. F) (10 o. F)
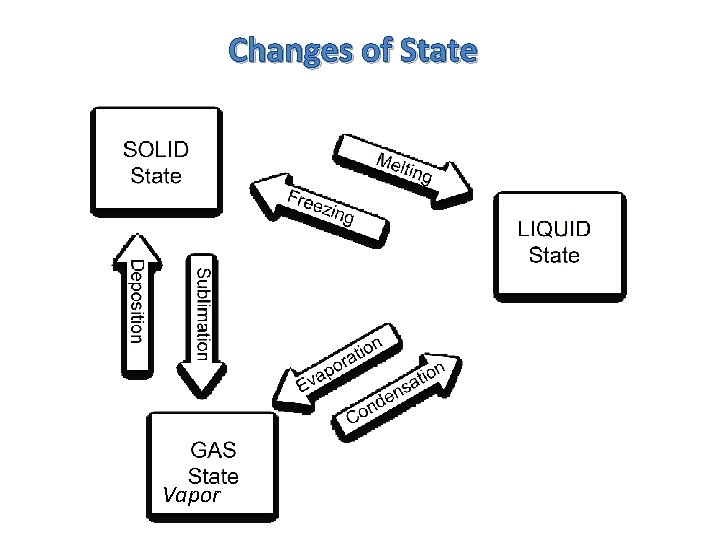
Changes of State Vapor
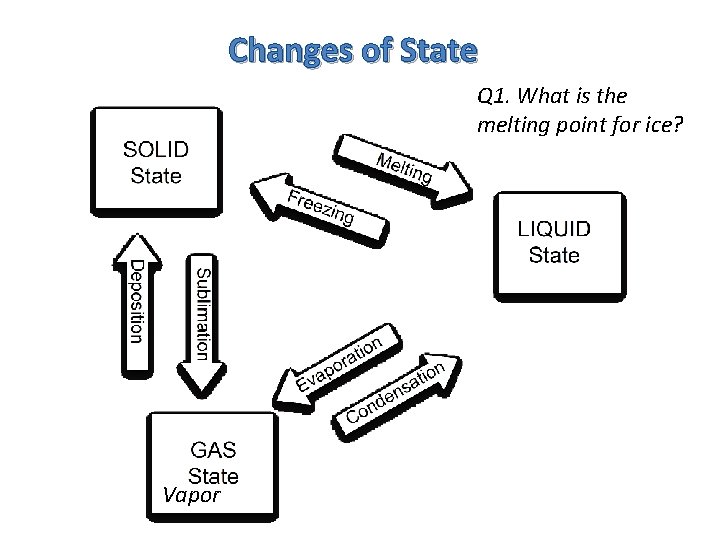
Changes of State Q 1. What is the melting point for ice? Vapor
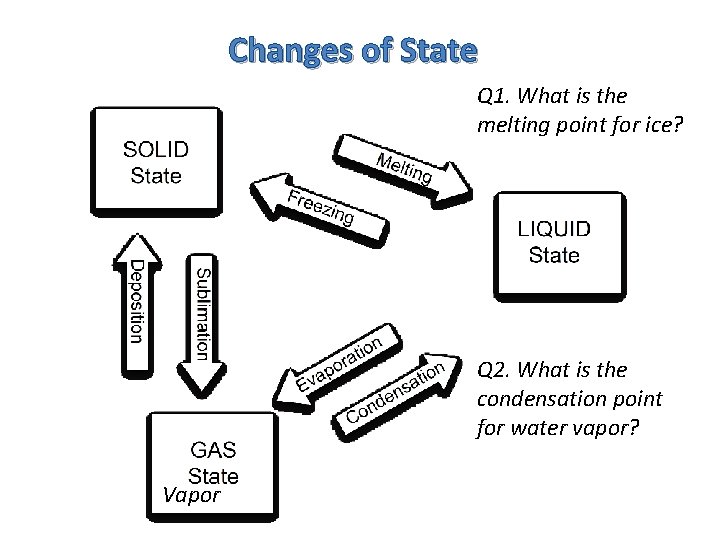
Changes of State Q 1. What is the melting point for ice? Q 2. What is the condensation point for water vapor? Vapor

Heat Transfer Mixing (requires air or Convection other mixing particles) Examples: warming soup on stove top

Heat Transfer Mixing (requires air or Convection other mixing particles) Examples: warming soup on stove top Conduction Direct Contact Examples: tip of a needle over flame

Heat Transfer Mixing (requires air or Convection other mixing particles) Examples: warming soup on stove top Conduction Direct Contact Examples: tip of a needle over flame Radiation No substance required Examples: warming soup in the microwave oven, heat coming from the sun

Thermal Expansion As temperature rises, dimensions expand Linear Railroad tracks, Bridge metal rods Volume Gasoline inside tank Q 9. As you heat a rectangular plate, what happens to its dimensions?

Thermal Expansion As temperature rises, dimensions expand Linear Railroad tracks, Bridge metal rods Volume Gasoline inside tank Q 9. As you heat a rectangular plate, what happens to its dimensions? Q 10. If the plate has a hole, what happens to the dimensions of the hole?

Heat and Heat Transfer 20 P 1. How many Btus are required to heat 2. 0 lb of water by 20 F? Ans. P 2. How many cal are required to heat 200 g of iron by 5 o. C ? Ans. P 3. You place an ice cube on the stove top and wait until it melts. (a) Ice absorbed heat and turned into water (b) Ice released heat and turned into water (c) Ice changed into water without any heat transfer. P 4. When does your car tank contain more gasoline? (a) summer (b) winter (c) It doesn’t matter
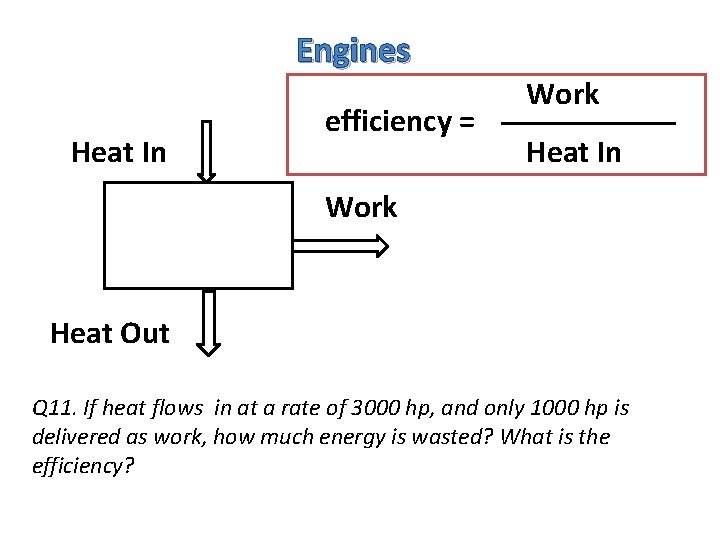
Engines Heat In efficiency = Work Heat In Work Heat Out Q 11. If heat flows in at a rate of 3000 hp, and only 1000 hp is delivered as work, how much energy is wasted? What is the efficiency?
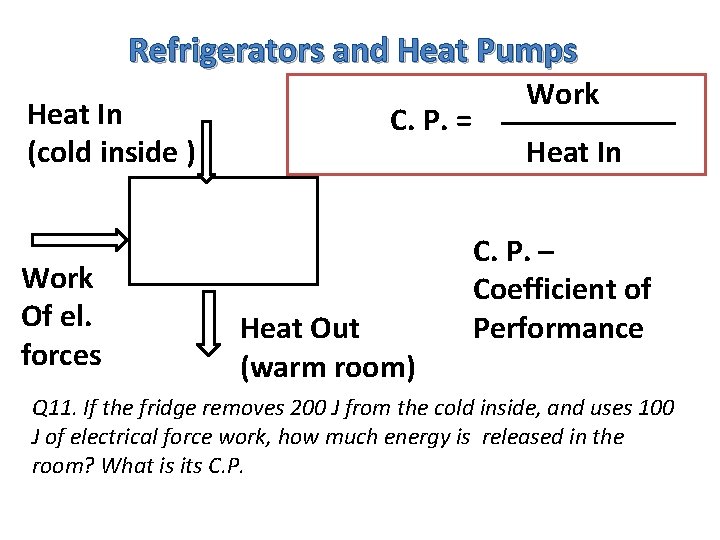
Refrigerators and Heat Pumps Heat In (cold inside ) Work Of el. forces C. P. = Heat Out (warm room) Work Heat In C. P. – Coefficient of Performance Q 11. If the fridge removes 200 J from the cold inside, and uses 100 J of electrical force work, how much energy is released in the room? What is its C. P.
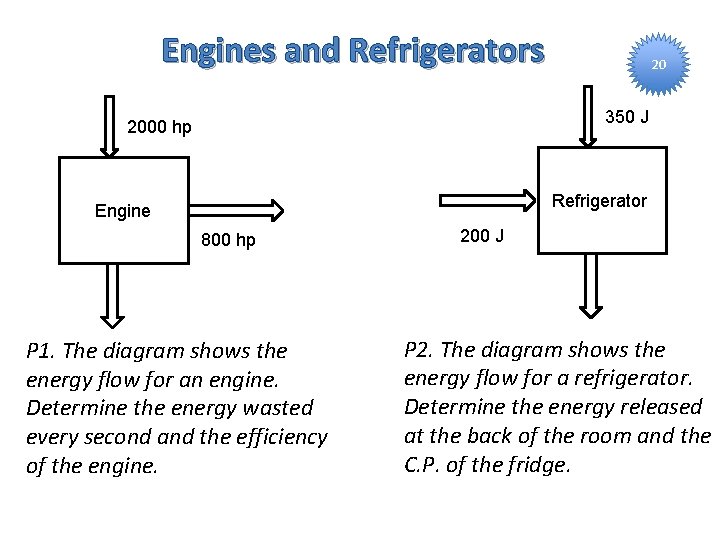
Engines and Refrigerators 20 350 J 2000 hp Refrigerator Engine 800 hp P 1. The diagram shows the energy flow for an engine. Determine the energy wasted every second and the efficiency of the engine. 200 J P 2. The diagram shows the energy flow for a refrigerator. Determine the energy released at the back of the room and the C. P. of the fridge.
- Slides: 30